Abstract
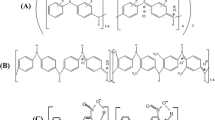
The development of the sphere of organic electronics contributes to the emergence of new materials used in this field. An important direction in the creation of new materials is the modification of already known functional organic compounds, such as polyaniline (PANI). The introduction of a substituent into the PANI aromatic ring is one of the most popular methods for polymer modification. Therefore, the study of the influence of various structural factors on the physicochemical properties of substituted PANI derivatives seems to be an important direction in the study of polymers. In this regard, in this paper, PANI was modified by introducing alkenyl substituents into the ortho-position of the aromatic ring of the monomer with a double bond in the allyl and vinyl positions. Based on the synthesized monomers, (co)polymers with aniline in various molar ratios were obtained by classical oxidative polymerization in an acidic medium and under the influence of (NH4)2S2O8. Solubility, morphology, thermal stability, humidity sensing, optical properties, and spectral characteristics were studied for all samples. The shift of the double bond from allyl to the vinyl position led to a decrease in the quantum yield from 0.13 to 0.03. However, the thermal stability of vinyl-substituted polyaniline derivatives was higher than allyl-substituted polymer, probably due to the formation of more cross-links in the polymer. Additionally, it was reported on the performance of a humidity sensor based on polyaniline copolymers, showing sensitivity to moisture at room temperature with a response time of 2–3 s.








Similar content being viewed by others
References
Li G, Chang WH, Yang Y (2017) Low-bandgap conjugated polymers enabling solution-processable tandem solar cells. Nat Rev Mater 2:1. https://doi.org/10.1038/natrevmats.2017.43
Inzelt G (2012) Conducting polymers: a new era in electrochemistry. Springer Science & Business Media
Andriianova AN, Biglova YN, Mustafin AG (2020) Effect of structural factors on the physicochemical properties of functionalized polyanilines. RSC Adv 10:7468. https://doi.org/10.1039/C9RA08644G
Chaudhary V, Kaushik AK, Furukawa H, Khosla A (2022) Towards 5th generation ai and iot driven sustainable intelligent sensors based on 2d mxenes and borophene. ECS Sensors Plus 1:013601. https://doi.org/10.1149/2754-2726/ac5ac6
Chaudhary V, Chavali M (2021) Novel methyl-orange assisted core-shell polyaniline-silver nanosheets for highly sensitive ammonia chemiresistors. J Appl Polym Sci 138:51288. https://doi.org/10.1002/app.51288
Chaudhary V, Ashraf N, Khalid M, Walvekar R, Yang Y, Kaushik A, Mishra YK (2022) Emergence of MXene–polymer hybrid nanocomposites as high-performance next-generation chemiresistors for efficient air quality monitoring. Adv Funct Mater 32:2112913. https://doi.org/10.1002/adfm.202112913
Chaudhary V (2022) Charge carrier dynamics of electrochemically synthesized poly (Aniline Co-Pyrrole) nanospheres based sulfur dioxide chemiresistor. Polym Plast Technol Eng 61:107. https://doi.org/10.1080/25740881.2021.1959932
Khamngoen K, Paradee N, Sirivat A (2016) Chemical oxidation polymerization and characterization of poly ortho-anisidine nanoparticles. J Polym Res 23:172. https://doi.org/10.1007/s10965-016-1073-7
Mustafin AG, Latypova LR, Andriianova AN, Usmanova GS (2021) Poly[N-(2-chloroprop-2-en-1-yl)aniline]s: synthesis, polymer analogous reaction, and physicochemical properties. Polym Chem 12:5650. https://doi.org/10.1039/D1PY00940K
Tian Y, Qu K, Zeng X (2017) Investigation into the ring-substituted polyanilines and their application for the detection and adsorption of sulfur dioxide. Sens Actuators B Chem 249:423. https://doi.org/10.1016/j.snb.2017.04.057
Andriianova A, Shigapova A, Biglova Y, Salikhov R, Abdrakhmanov I, Mustafin A (2019) Synthesis and physico-chemical properties of (co)polymers of 2-[(2E)-1-methyl-2-buten-1-yl] aniline and aniline. Chin J Polym Sci 37:774. https://doi.org/10.1007/s10118-019-2261-9
Jaymand M (2013) Recent progress in chemical modification of polyaniline. Prog Polym Sci 38:1287. https://doi.org/10.1016/j.progpolymsci.2013.05.015
Thota A, Arukula R, Narayan R, Rao CR, Raju KVSN (2015) Energy storage and surface protection properties of dianiline co-polymers. RSC Adv 5:106523. https://doi.org/10.1039/C5RA11731C
Mustafin A, Latypova L, Andriianova A, Salikhov Sh, Sattarova A, Mullagaliev I, Salikhov R, Abdrakhmanov I (2020) Synthesis and physico-chemical properties of poly(2-Ethyl-3-Methylindole). Macromolecules 53:8050. https://doi.org/10.1021/acs.macromol.0c01308
Andriianova AN, Gribko DE, Petrov IS, Mullagaliev I, Sattarova AF, Salikhov RB, Abdrakhmanov IB, Mustafina AG (2021) Synthesis and physicochemical properties of poly[2-(cyclohex-2-en-1-yl)aniline] as a new polyaniline derivative. New J Chem 45:6356. https://doi.org/10.1039/D1NJ00596K
Latypova LR, Andriianova AN, Mullagaliev IN, Salikhov ShM, Salikhov RB, Abdrakhmanov IB, Mustafin AG (2021) Polymerization of new aniline derivatives: synthesis, characterization and application as sensors. RSC Adv 11:21006. https://doi.org/10.1039/D1RA02474D
Andriianova AN, Sadykov TT, Mustafin AG (2021) Synthesis and physicochemical properties of poly-2-[(2E)-1-methyl-1-buten-1-yl]aniline and its copolymers. ChemistrySelect 6:8942. https://doi.org/10.1002/slct.202102184
Chen Y, Barkley MD (1998) Toward understanding tryptophan fluorescence in proteins. Biochemistry 37:9976. https://doi.org/10.1021/bi980274n
Kulkarni MV, Viswanath AK, Khanna PK (2006) Synthesis and characterization of poly (o-anisidine) doped with polymeric acids. Int J Polym Mater Polym Biomater 55:501. https://doi.org/10.1080/00914030500210358
Trchová M, Šeděnková I, Tobolková E, Stejskal J (2004) FTIR spectroscopic and conductivity study of the thermal degradation of polyaniline films. Polym Degrad Stab 86:179. https://doi.org/10.1016/j.polymdegradstab.2004.04.011
Bhadra S, Kim NH, Lee JH (2010) Synthesis of water-soluble sulfonated polyaniline and determination of crystal structure. J Appl Polym Sci 117:2025. https://doi.org/10.1002/app.32152
Ahlatcıoğlu E, Senkal BF, Okutan M, Gursel Y (2014) Preparation and polymerization of chalcone substituted aniline and investigation of impedance properties. Mater Sci Semicond Process 28:103. https://doi.org/10.1016/j.mssp.2014.07.018
Movahedifar F, Modarresi-Alam AR (2016) The effect of initiators and oxidants on the morphology of poly[(±)-2-(sec-butyl) aniline] a chiral bulky substituted polyaniline derivative. Polym Adv Technol 27:131. https://doi.org/10.1002/pat.3614
Stejskal J, Trchová M, Prokeš J, Sapurina I (2001) Brominated polyaniline. Chem Mater 13:4083. https://doi.org/10.1021/cm011059n
Wei Y, Hariharan R, Patel SA (1990) Chemical and electrochemical copolymerization of aniline with alkyl ring-substituted anilines. Macromolecules 23:758. https://doi.org/10.1021/ma00205a011
Lindfors T, Ivaska A (2002) pH sensitivity of polyaniline and its substituted derivatives. J Electroanal Chem 531:43. https://doi.org/10.1016/S0022-0728(02)01005-7
Yang S, Liao F (2012) Poly (p-phenylenediamine) nanofibers having conjugated structures, a novel, simple and highly selective fluorescent probe for l-cysteine. Synth Met 162:1343. https://doi.org/10.1016/j.synthmet.2012.05.019
Zhang T, Yang S, Sun J, Li X, He L, Yan S, Liao F (2013) Poly (p-phenylenediamine) fluorescent nanosphere: a ultra-sensitive fluorescent probe for caffeine. Synth Met 181:86. https://doi.org/10.1016/j.synthmet.2013.08.008
Tian J, Li H, Luo Y, Wang L, Zhang Y, Sun X (2011) Poly(o-phenylenediamine) colloid-quenched fluorescent oligonucleotide as a probe for fluorescence-enhanced nucleic acid detection. Langmuir 27:874. https://doi.org/10.1021/la103799e
Salavagione HJ, Acevedo DF, Miras MC, Motheo AJ, Barbero CA (2004) Comparative study of 2-amino and 3-aminobenzoic acid copolymerization with aniline synthesis and copolymer properties. J Polym Sci A Polym Chem 42:5587. https://doi.org/10.1002/pola.20409
Shimano JY, MacDiarmid AG (2001) Polyaniline, a dynamic block copolymer: key to attaining its intrinsic conductivity? Synth Met 123:251. https://doi.org/10.1016/S0379-6779(01)00293-4
Parker CA (1969) Photoluminescence of solutions with applications to photochemistry and analytical chemistry. Elsevier, New York
Liao F, Yang S, Li X, Yang L, Xie Z, Hu C, Ren T (2014) Poly(o-phenylenediamine) and benzeneselenol copolymer fluorescent nanorod: an ultra-sensitive fluorescent probe and a fluorescent switch triggered by redox procedure. Synth Met 189:135. https://doi.org/10.1016/j.synthmet.2014.01.020
Arias-Pardilla J, Salavagione HJ, Barbero C, Morallon E, Vazquez JL (2006) Study of the chemical copolymerization of 2-aminoterephthalic acid and aniline: synthesis and copolymer properties. Eur Polym J 42:1521. https://doi.org/10.1016/j.eurpolymj.2006.02.003
Tran HD, Norris I, D’Arcy JM, Tsang H, Wang Y, Mattes BR, Kaner RB (2008) Substituted polyaniline nanofibers produced via rapid initiated polymerization. Macromolecules 41:7405. https://doi.org/10.1021/ma800122d
Huang J, Kaner RB (2006) The intrinsic nanofibrillar morphology of polyaniline. Chem Comm 4:367. https://doi.org/10.1039/B510956F
Anilkumar P, Jayakannan M (2008) Hydroxyl-functionalized polyaniline nanospheres: tracing molecular interactions at the nanosurface via vitamin C sensing. Langmuir 24:9754. https://doi.org/10.1021/la801128j
Alves WF, Venancio EC, Leite FL, Kanda DH, Malmonge LF, Malmonge JA, Mattoso LH (2010) Thermo-analyses of polyaniline and its derivatives. Thermochim Acta 502:43. https://doi.org/10.1016/j.tca.2010.02.003
Zeng FW, Liu XX, Diamond D, Lau KT (2010) Humidity sensors based on polyaniline nanofibers. Sens Actuators B Chem 143:530. https://doi.org/10.1016/j.snb.2009.09.050
Kumar L, Rawal I, Kaur A, Annapoorni S (2017) Flexible room temperature ammonia sensor based on polyaniline. Sens Actuators B Chem 240:408. https://doi.org/10.1016/j.snb.2016.08.173
Andriianova AN, Salikhov RB, Latypova LR, Mullagaliev IN, Salikhov TR, Mustafin AG (2022) The structural factors affecting the sensory properties of polyaniline derivatives. Sustain Energy Fuels 6:3435. https://doi.org/10.1039/D2SE00405D
Tameev AR, Rakhmeev RG, Nikitenko VR, Salikhov RB, Bunakov AA, Lachinov AN, Vannikov AV (2011) Effect of excessive pressure on the drift mobility of charge carriers in poly(diphenylene phthalide) films. Phys Solid State 53:195. https://doi.org/10.1134/S106378341101032X
Kulkarni MV, Apte SK, Naik SD, Ambekar JD, Kale BB (2013) Ink-jet printed conducting polyaniline based flexible humidity sensor. Sens Actuators B Chem 178:140. https://doi.org/10.1016/j.snb.2012.12.046
Funding
This work was supported by the Russian Science Foundation, project number 22-23-00623, and the Ministry of Science and Higher Education of the Russian Federation as part of the state task no. 122031400278–2.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Andriianova, A., Mustafin, A. Synthesis and physicochemical properties of (co)polymers based on aniline and its ortho-cycloalkenyl-substituted derivatives. Polym. Bull. 80, 10215–10230 (2023). https://doi.org/10.1007/s00289-022-04554-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-022-04554-y