Abstract
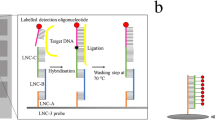
A commercially available microarray (IDENTIBAC AMR-ve) for the detection of antibiotic resistance determinants was investigated for its potential to genotype 30 clinical isolates and two control strains of Klebsiella pneumoniae. Resistance profiles and the production of extended-spectrum β-lactamases were determined by disc diffusion and the results were compared with the microarray profiles in order to assess its scope and limitations. Genes associated with resistance to a wide range of antibiotics, including current first line therapy options, were detected. In addition, the array also detected class 1 integrases. The array is easy to use and interpret, and is useful in providing a general description of the numbers and types of resistance determinants in K. pneumoniae. It also provides an indication of the potential for resistance gene acquisition. However, in most instances detected resistance to specific antibiotics could not unequivocally be assigned to hybridization with a specific array probe. We conclude that the microarray is a valuable and rapid means of investigating the presence of resistance gene classes of therapeutic importance. It can also provide a starting point for selecting analyses of greater resolving power, such as phylogenetic subtyping by PCR sequencing.

Similar content being viewed by others
References
Almaghrabi R, Clancy CJ, Doi Y, Hao B et al (2014) Carbapenem-resistant Klebsiella pneumoniae strains exhibit diversity in aminoglycoside-modifying enzymes, which exert differing effects on plazomicin and other agents. Antimicrob Agents Chemother 58:4443–4451
Altschul SF, Gish W, Miller W, Myers EW et al (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Ambler RP (1980) The structure of β-lactamases. Philos Trans R Soc Lond B Biol Sci 289:321–331
Babini GS, Livermore DM (2000) Are SHV β-lactamases universal in Klebsiella pneumoniae? Antimicrob Agents Chemother 44:2230
Batchelor M, Hopkins KL, Liebana E, Slickers P et al (2008) Development of a miniaturised microarray-based assay for the rapid identification of antimicrobial resistance genes in gram-negative bacteria. Int J Antimicrob Agents 31:440–451
Bhattacharjee A, Sen MR, Prakash P, Gaur A et al (2010) Observation on integron carriage among clinical isolates of Klebsiella pneumoniae producing extended-spectrum β-lactamases. Ind J Med Microbiol 28:207–210
Bokaeian M, Saeidi S, Shahi Z, Kadaei V (2014) TetA and tetB Genes in Klebsiella pneumoniae isolated from clinical samples. Gene Cell Tissue 1:e18152. doi:10.17795/gct-18152
Bonnet R (2004) Growing group of extended-spectrum β-lactamases: the CTX-M-type enzymes. Antimicrob Agents Chemother 48:1–14
Bush K, Jacoby GA (2010) Updated functional classification of β-lactamases. Antimicrob Agents Chemother 54:969–976
Card R, Zhang J, Das P, Cook C et al (2013) Evaluation of an expanded microarray for detecting antibiotic resistance genes in a broad range of gram-negative bacterial pathogens. Antimicrob Agents Chemother 57:458–465. doi:10.1128/AAC.01223-12
Casin I, Hanau-Berçot B, Podglajen I, Vahaboglu H et al (2003) Salmonella enterica serovar Typhimurium blaPER-1-carrying plasmid pSTI1 encodes an extended-spectrum aminoglycoside 6′-N-acetyltransferase of type Ib. Antimicrob Agents Chemother 47:697–703
Charnock C, Nordlie AL, Hjeltnes B (2014) Toxin production and antibiotic resistances in Escherichia coli Isolated from bathing areas along the coastline of the Oslo Fjord. Curr Microbiol 69:317–328
Chaves J, Ladona MG, Segura C, Coira A et al (2001) SHV-1 β-lactamase is mainly a chromosomally encoded species-specific enzyme in Klebsiella pneumoniae. Antimicrob Agents Chemother 45:2856–2861
Edelstein M, Pimkin M, Palagin I, Edelstein I et al (2003) Prevalence and molecular epidemiology of CTX-M extended-spectrum β-lactamase-producing Escherichia coli and Klebsiella pneumoniae in Russian hospitals. Antimicrob Agents Chemother 47:3724–3732
Endimiani A, DePasquale JM, Forero S, Perez F et al (2009) Emergence of blaKPC-containing Klebsiella pneumoniae in a long-term acute care hospital: a new challenge to our healthcare system. J Antimicrob Chemother 64:1102–1110
Frank T, Gautier V, Talarmin A, Bercion R et al (2007) Characterization of sulphonamide resistance genes and class 1 integron gene cassettes in Enterobacteriaceae, Central African Republic (CAR). J Antimicrob Chemother 59:742–745
Hammond DS, Schooneveldt JM, Nimmo GR, Huygens F et al (2005) blaSHV genes in Klebsiella pneumoniae: different allele distributions are associated with different promoters within individual isolates. Antimicrob Agents Chemother 49:256–263
Heritage J, M’Zali FH, Gascoyne-Binzi D, Hawkey PM (1999) Evolution and spread of SHV extended-spectrum β-lactamases in Gram-negative bacteria. J Antimicrob Chemother 44:309–318
Miller GH, Sabatelli FJ, Hare RS, Glupczynski Y et al (1997) The most frequent aminoglycoside resistance mechanisms—changes with time and geographic area: a reflection of aminoglycoside usage patterns? Clin Infect Dis 24:46–62
Munoz-Price LS, Poirel L, Bonomo RA, Schwaber MJ et al (2013) Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect Dis 13:785–796
Park SH, Byun JH, Choi SM, Lee DG et al (2012) Molecular epidemiology of extended-spectrum β-lactamase-producing Escherichia coli in the community and hospital in Korea: emergence of ST131 producing CTX-M-15. BMC Infect Dis 12:149
Petit A, Yaghlane-Bouslama HB, Sofer L, Labia R (1992) Does high level production of SHV-type penicillinase confer resistance to ceftazidime in Enterobacteriaceae? FEMS Microbiol Lett 92:89–94
Rice LB, Carias LL, Hujer AM, Bonafede M et al (2000) High-level expression of chromosomally encoded SHV-1 β-lactamase and an outer membrane protein change confer resistance to ceftazidime and piperacillin-tazobactam in a clinical isolate of Klebsiella pneumoniae. Antimicrob Agents Chemother 44:362–367
Robicsek A, Strahilevitz J, Jacoby GA, Macielag M et al (2006) Fluoroquinolone-modifying enzyme: a new adaptation of a common aminoglycoside acetyltransferase. Nat Med 12:83–88
Rossolini GM, D’andrea MM, Mugnaioli C (2008) The spread of CTX-M-type extended-spectrum β-lactamases. Clin Microbiol Infect 14:33–41
Schwaber MJ, Carmeli Y (2008) Carbapenem-resistant Enterobacteriaceae: a potential threat. Jama 300:2911–2913
Seiffert SN, Hilty M, Perreten V, Endimiani A (2013) Extended-spectrum cephalosporin-resistant Gram-negative organisms in livestock: an emerging problem for human health? Drug Res Updates 16:22–45
Shams F, Hasani A, Rezaee MA, Nahaie MR et al (2015) Carriage of class 1 and 2 integrons in quinolone, extended-spectrum-β-lactamase-producing and multi drug resistant E. coli and K. pneumoniae: high burden of antibiotic resistance. Adv Pharm Bull 5:335–342
Tzouvelekis LS, Markogiannakis A, Psichogiou M, Tassios PT (2012) Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clin Microbiol Rev 25:682–707
Vakulenko SB, Mobashery S (2003) Versatility of aminoglycosides and prospects for their future. Clin Microbiol Rev 16:430–450
Walsh F, Cooke NM, Smith SG, Moran GP et al (2010) Comparison of two DNA microarrays for detection of plasmid-mediated antimicrobial resistance and virulence factor genes in clinical isolates of Enterobacteriaceae and non-Enterobacteriaceae. Int J Antimicrob Agents 35:593–598
Wang G, Huang T, Surendraiah PKM, Wang K et al (2013) CTX-M β-lactamase-producing Klebsiella pneumoniae in suburban New York City, New York, USA. Emerg Infect Dis 9:1803–1810
Wang W, Guo Q, Xu X, Sheng ZK et al (2014) High-level tetracycline resistance mediated by efflux pumps tet (A) and tet (A)-1 with two start codons. J Med Microbiol 63:1454–1459
Acknowledgements
The authors wish to thank the ‘NORM surveillance programme for antimicrobial resistance in human pathogens’ at the University Hospital of North Norway for providing financial support to the project. Thanks to Anders Benteson Nygaard for assistance with Fig. 1.
Funding
The study received on open application a research grant from NORM surveillance programme for antimicrobial resistance in human pathogens to partially cover the purchase of consumables to the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Charnock, C., Samuelsen, Ø., Nordlie, AL. et al. Use of a Commercially Available Microarray to Characterize Antibiotic-Resistant Clinical Isolates of Klebsiella pneumoniae . Curr Microbiol 75, 163–172 (2018). https://doi.org/10.1007/s00284-017-1361-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-017-1361-4