Abstract
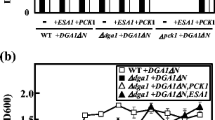
Mechanisms that may regulate the storage of energy as triacylglycerol in Saccharomyces cerevisiae were examined. First, the kinetics of Dga1p, which mediates the majority of diacylglycerol esterification, the lone committed step in triacylglycerol synthesis, was measured in vitro. With an apparent K m of 17.0 μM, Dga1p has higher affinity for oleoyl-CoA than the only S. cerevisiae acyltransferase previously kinetically characterized, Lpt1p. Lpt1p is a 1-acylglycerol-3-phosphate O-acyltransferase that produces phosphatidate, a precursor to diacylglycerol. Therefore, limiting triacylglycerol synthesis to situations of elevated acyl-CoA concentration is unlikely. However, Dga1p’s apparent V max of 5.8 nmol/min/mg was 20 times lower than Lpt1p’s. This supports Dga1p being rate limiting for TAG synthesis. Dga1p activity was not activated or inhibited when seven different molecules (e.g., ATP) which reflect cellular energy status were provided at physiological concentrations. Thus, allosteric regulation was not found. Coordination between triacylglycerol and glycogen synthesis was also tested. Yeast genetically deficient in triacylglycerol synthesis did not store more energy in glycogen and vice versa. Lastly, we tested whether genetically limiting energy storage in triacylglycerol, glycogen, steryl esters, or combinations of these will increase ethanol production efficiency. In nutrient-rich media containing 5 % glucose, solely limiting glycogen synthesis had the greatest affect, increasing ethanol production efficiency by 12 %. Since limiting glycogen synthesis only had a modest effect on growth in media containing 10 % ethanol, such genetic manipulation may improve commercial ethanol production.






Similar content being viewed by others
References
Aitchison J (1982) The statistical analysis of compositional data. J R Stat Soc B 44:139–177
Alper H, Moxley J, Nevoigt E, Fink GR, Stephanopoulos G (2016) Engineering yeast transcription machinery for improved ethanol tolerance and production. Science 314:1565–1568
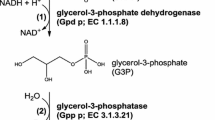
Athenstaedt K, Weys S, Paltauf F, Daum G (1999) Redundant systems of phosphatidic acid biosynthesis via acylation of glycerol-3-phosphate or dihydroxyacetone phosphate in the yeast Saccharomyces cerevisiae. J Bacteriol 179:7611–7616
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1998) Current protocols in molecular biology. Wiley, New York
Bajpai P, Sharma A, Raghuram N, Bajpai PK (1988) Rapid production of ethanol in high concentration by immobilized cells of Saccharomyces cerevisiae through soya flour supplementation. Biotechnol Lett 10:217–220
Cao H (2011) Structure-function analysis of diacylglycerol acyltransferase sequences from 70 organisms. BMC Res Notes 4:249
De Smet CH, Vittone E, Scherer M, Houweling M, Liebisch G, Brouwers JF, de Kroon AI (2012) The yeast acyltransferase Sct1p regulates fatty acid desaturation by competing with the desaturase Ole1p. Mol Biol Cell 23:1146–1156
Eleutherio E, Panek A, De Mesquita JF, Trevisol E, Magalhães R (2015) Revisiting yeast trehalose metabolism. Curr Genet 61:263–274
Enjalbert B, Parrou JL, Teste MA, François J (2004) Combinatorial control by the protein kinases PKA, PHO85 and SNF1 of transcriptional induction of the Saccharomyces cerevisiae GSY2 gene at the diauxic shift. Mol Genet Genom 271:697–708
Farkas I, Hardy TA, Goebl MG, Roach PJ (1991) Two glycogen synthase isoforms in Saccharomyces cerevisiae are coded by distinct genes that are differentially controlled. J Biol Chem 266:15602–15607
Gsell M, Fankl A, Klug L, Mascher G, Schmidt C, Hrastnik C, Zellnig G, Daum G (2015) A yeast mutant deleted of GPH1 bears defects in lipid metabolism. PLoS One 10:e0136957
Henderson CM, Lozada-Contreras M, Jiranek V, Longo ML, Block DE (2013) Ethanol production and maximum cell growth are highly correlated with membrane lipid composition during fermentation as determined by lipidomic analysis of 22 Saccharomyces cerevisiae strains. Appl Environ Microbiol 79:91–104
Illingworth RF, Rose AH, Beckett A (1973) Changes in the lipid composition and fine structure of Saccharomyces cerevisiae during ascus formation. J Bacteriol 113:373–386
Jain S, Stanford N, Bhagwat N, Seiler B, Costanzo M, Boone C, Oelkers P (2007) Identification of a novel lysophospholipid acyltransferase in Saccharomyces cerevisiae. J Biol Chem 282:30562–30569
Kane SM, Roth R (1974) Carbohydrate metabolism during ascospore development in yeast. J Bacteriol 118:8–14
Lam FH, Ghaderi A, Fink GR, Stephanopoulos G (2014) Biofuels engineering alcohol tolerance in yeast. Science 346:71–75
Lardizabal KD, Mai JT, Wagner NW, Wyrick A, Voelker T, Hawkins DJ (2001) DGAT2 is a new diacylglycerol acyltransferase gene family: purification, cloning, and expression in insect cells of two polypeptides from Mortierella ramanniana with diacylglycerol acyltransferase activity. J Biol Chem 276:38862–38869
Mansure JJ, Panek AD, Crowe LM, Crowe JH (1994) Trehalose inhibits ethanol effects on intact yeast cells and liposomes. Biochim Biophys Acta 1191:309–316
Morlock KR, Lin YP, Carman GM (1988) Regulation of phosphatidate phosphatase activity by inositol in Saccharomyces cerevisiae. J Bacteriol 170:3561–3566
Nagiec MM, Wells GB, Lester RL, Dickson RC (1993) A suppressor gene that enables Saccharomyces cerevisiae to grow without making sphingolipids encodes a protein that resembles an Escherichia coli fatty acyltransferase. J Biol Chem 268:22156–22163
O’Hara L, Han GS, Peak-Chew S, Grimsey N, Carman GM, Siniossoglou S (2006) Control of phospholipid synthesis by phosphorylation of the yeast lipin Pah1p/Smp2p Mg2+-dependent phosphatidate phosphatase. J Biol Chem 281:34537–34548
Oelkers P, Cromley D, Padamsee M, Billheimer JT, Sturley SL (2002) The DGA1 gene determines a second triglyceride synthetic pathway in yeast. J Biol Chem 277:8877–8881
Oelkers P, Tinkelenberg A, Erdeniz N, Cromley D, Billheimer JT, Sturley SL (2000) A lecithin cholesterol acyltransferase-like gene mediates diacylglycerol esterification in yeast. J Biol Chem 275:15609–15612
Parrou JL, Francois J (1997) A simplified procedure for a rapid and reliable assay of both glycogen and trehalose in whole yeast cells. Anal Biochem 248:186–188
Pascual F, Hsieh LS, Soto-Cardalda A, Carman GM (2014) Yeast Pah1p phosphatidate phosphatase is regulated by proteasome-mediated degradation. J Biol Chem 289:9811–9822
Pascual F, Soto-Cardalda A, Carman GM (2013) PAH1-encoded phosphatidate phosphatase plays a role in the growth phase- and inositol-mediated regulation of lipid synthesis in Saccharomyces cerevisiae. J Biol Chem 288:35781–35792
Perez-Torrado R, Matallana E (2015) Enhanced fermentative capacity of yeasts engineered in storage carbohydrate metabolism. Biotechnol Prog 31:20–24
Riekhof WR, Wu J, Jones JL, Voelker DR (2007) Identification and characterization of the major lysophosphatidylethanolamine acyltransferase in Saccharomyces cerevisiae. J Biol Chem 282:28344–28352
Rothstein R (1991) Targeting, disruption, replacement, and allele rescue: integrative DNA transformation in yeast. Methods Enzymol 194:281–301
Rubin MM, Changeux JP (1966) On the nature of allosteric transitions: implications of non-exclusive ligand binding. J Mol Biol 21:265–274
Runguphan W, Keasling JD (2014) Metabolic engineering of Saccharomyces cerevisiae for production of fatty acid-derived biofuels and chemicals. Metab Eng 21:103–113
Sandager L, Gustavsson MH, Stahl U, Dahlqvist A, Wiberg E, Banas A, Lenman M, Ronne H, Stymne S (2002) Storage lipid synthesis is non-essential in yeast. J Biol Chem 277:6478–6482
Shpilka T, Welter E, Borovsky N, Amar N, Mari M, Reggiori F, Elazar Z (2015) Lipid droplets and their component triglycerides and steryl esters regulate autophagosome biogenesis. EMBO J 34:2117–2131
Thomas BJ, Rothstein R (1989) Elevated recombination rates in transcriptionally active DNA. Cell 56:619–630
Wang Z, Wilson WA, Fujino MA, Roach PJ (2001) Antagonistic controls of autophagy and glycogen accumulation by Snf1p, the yeast homolog of AMP-activated protein kinase, and the cyclin-dependent kinase Pho85p. Mol Cell Biol 21:5742–5752
Yang H, Bard M, Bruner DA, Gleeson A, Deckelbaum RJ, Aljinovic G, Pohl TM, Rothstein R, Sturley SL (1996) Sterol esterification in yeast: a two-gene process. Science 272:1353–1356
Yu KO, Jung J, Ramzi AB, Kim SW, Park C, Han SO (2012) Improvement of ethanol yield from glycerol via conversion of pyruvate to ethanol in metabolically engineered Saccharomyces cerevisiae. Appl Biochem Biotechnol 166:856–865
Zhou H, Cheng JS, Wang BL, Fink GR, Stephanopoulos G (2012) Xylose isomerase overexpression along with engineering of the pentose phosphate pathway and evolutionary engineering enable rapid xylose utilization and ethanol production by Saccharomyces cerevisiae. Metab Eng 14:611–622
Acknowledgments
Peter oelkers was supported by a Step 2 Grant sponsored by the University of Michigan-Dearborn Office of Research and Sponsored Programs. We thank Eric Pomaranski for performing pilot experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Silpha Jain contributed to this manuscript, experiments, and writing, while she was a graduate student at Drexel University. She is now employed by Trac Services, a pharmaceutical regulatory consulting company as a senior regulatory affairs executive. No pharmaceuticals were used in this study. There are no other potential conflicts of interest to report.
Research Involving Human and Animal Rights
No human or animal subjects were used in these studies.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jain, S., Dholakia, H., Kirtley, W. et al. Energy Storage in Yeast: Regulation and Competition with Ethanol Production. Curr Microbiol 73, 851–858 (2016). https://doi.org/10.1007/s00284-016-1127-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-016-1127-4