Abstract
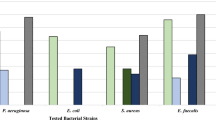
The aim of this study was to evaluate the antibacterial effects of glochidioboside and determine its mechanism of action. Glochidioboside has been reported to be isolated from some plants but the underlying biological properties have remained largely obscure until now. To identify the antibacterial activity of all biological properties, pathogenic bacteria susceptibility test was performed, and the result shows that the compound displays remarkable antibacterial activity against antibiotic-resistant bacteria not to mention general pathogen. To demonstrate membrane disruption and depolarization, SYTOX green and bis-(1,3-dibutylbarbituric acid) trimethine oxonol were used with Escherichia coli O157, and indicated that glochidioboside affected cytoplasmic membranes by permeabilization and depolarization, respectively. Calcein efflux was evident in a membrane model that encapsulated fluorescent dye, and supported the hypothesis of a membrane-active mechanism. To confirm the release of intracellular matrix owing to membrane damage, the movements of potassium ion were observed; the results indicated that the cells treated with glochidioboside leaked potassium ion, thus the damage induced by the compounds lead to leaking intracellular components. We propose that glochidioboside kills pathogenic bacteria via perturbation of integrity of the membrane.





Similar content being viewed by others
Abbreviations
- CH2Cl2 :
-
Dichloromethane
- EtOAc:
-
Ethyl acetate
- n-BuOH:
-
n-Butanol
- ATCC:
-
American Type Culture Collection
- MIC:
-
Minimum inhibitory concentrations
- PBS:
-
Phosphate-buffered saline
- DiBAC4(3):
-
Bis-(1,3-dibutylbarbituric acid) trimethine oxonol
- GUV:
-
Giant unilamellar vesicle
- PE:
-
Phosphatidylethanolamine
- PG:
-
Phosphatidylglycerol
- LUV:
-
Large unilamellar vesicle
References
Abreu AC, McBain AJ, Simões M (2012) Plants as sources of new antimicrobials and resistance-modifying agents. Nat Prod Rep 29:1007–1021
Angelova MI, Dimitrov DS (1986) Liposome electroformation. Faraday Discuss Chem Soc 81:303–311
Angelova MI, Sloeau S, Meleard P, Faucon JF, Bothorel P (1992) Preparation of giant vesicles by external AC electric fields. Kinetics and application. Prog Colloid Polym Sci 89:127–131
CLSI (2005) Clinical and Laboratory Standards Institute Performance Standards for Antimicrobial Susceptibility Testing, 15th edn. CLSI, Wayne
Coyne S, Courvalin P, Périchon B (2011) Efflux-mediated antibiotic resistance in Acinetobacter spp. Antimicrob Agents Chemother 55:947–953
Cushnie TP, Cushnie B, Lamb AJ (2014) Alkaloids: an overview of their antibacterial, antibiotic-enhancing and antivirulence activities. Int J Antimicrob Agents 44:377–386
Cushnie TP, Lamb AJ (2005) Antimicrobial activity of flavonoids. Int J Antimicrob Agents 26:343–356
de Freitas CD, Lopes JL, Beltramini LM, de Oliveira RS, Oliveira JT, Ramos MV (2011) Osmotin from Calotrpis procera latex: new insights into structure and antifungal properties. Biochim Biophys Acta 1808:2501–2507
Deering AJ, Mauer LJ, Pruitt RE (2012) Internalization of E. coli O157:H7 and Salmonella spp. in plants: A review. Food Res Int 45:567–575
Dong LP, Ni W, Dong JY, Li JZ, Chen CX, Liu HY (2006) A new neolignan glycoside from the leaves of Acer truncatum. Molecules 11:1009–1014
Fazio A, Plastina P, Meijerink J, Witkamp RF, Gavriele B (2013) Comparative analyses of seeds of wild fruits of Rubus and Sambucus species from Southern Italy: fatty acid composition of the oil, total phenolic content, antioxidant and anti-inflammatory properties of the methanolic extracts. Food Chem 150:817–824
Gibbons S (2008) Phytochemicals for bacterial resistance – strengths, weaknesses and opportunities. Planta Med 74:594–602
Hamada T, Hagihara H, Morita M, Vestergaard MDC, Tsujino Y, Takagi M (2012) Physicochemical profiling of surfactant-induced membrane dynamics in a cell-sized liposome. J Phys Chem Lett 3:430–435
Herbst R, Marciano-Cabral F, Leippe M (2004) Antimicrobial and pore-forming peptides of free-living and potentially highly pathogenic Naegleria fowleri are released from the same precursor molecule. J Biol Chem 275:25955–25958
Kennedy DO, Wightman EL (2011) Herbal extracts and phytochemicals: plant secondary metabolites and the enhancement of human brain function. Adv Nutr 2:32–50
Kumar K, Awasthi D, Lee SY, Cummings JE, Knudson SE, Slayden RA, Ojima I (2013) Benzimidazole-based antibacterial agents against Francisella tularensis. Bioorg Med Chem 21:3318–3326
Lam KS (2007) New aspects of natural products in drug discovery. Trends Microbiol 15:279–289
Lee J, Choi H, Cho J, Lee DG (2011) Effects of positively charged arginine residues on membrane pore forming activity of Rev-NIS peptide in bacterial cells. Biochim Biophys Acta 1808:2421–2427
Lennen RM, Kruziki MA, Kumar K, Zinkel RA, Burnum KE, Lipton MS, Hoover SW, Ranatunga DR, Wittkopp TM, Marner WD, Pfleger BF (2011) Membrane stresses induced by overproduction of free fatty acids in Escherichia coli. Appl Environ Microbiol 77:8114–8128
Lüders S, David F, Steinwand M, Jordan E, Hust M, Dübel S, Franco-Lara E (2011) Influence of the hydromechanical stress and temperature on growth and antibody fragment production with Bacillus megaterium. Appl Microbiol Biotechnol 91:81–90
Makovitzki A, Avrahami D, Shai Y (2006) Ultrashort antibacterial and antifungal lipopeptides. Proc Natl Acad Sci USA 103:15997–16002
Meister A, Finger S, Hause G, Blume A (2014) Morphological changes of bacterial model membrane vesicles. Eur J Lipid Sci Technol 116:1228–1233
Moerman L, Bosteels S, Noppe W, Willems J, Clynen E, Schoofs L, Thevissen K, Tytgat J, Van Eldere J, Van Der Walt J, Verdonck F (2002) Antibacterial and antifungal properties of alpha-helical, cationic peptides in the venom of scorpions from southern Africa. Eur J Biochem 269:4799–4810
Orlov DS, Nguyen T, Lehrer RI (2002) Potassium release, a useful tool for studying antimicrobial peptides. J Microbiol Methods 49:325–328
Pilsczek FH, Salina D, Poon KK, Fahey C, Yipp BG, Sibley CD, Robbins SM, Green FH, Surette MG, Sugai M, Bowden MG, Hussain M, Zhang K, Kubes P (2010) A novel mechanism of rapid nuclear neutrophil extracellular trap formation in response to Staphylococcus aureus. J Immunol 185:7413–7425
Scalbert A (1991) Antimicrobial properties of tannins. Phytochemistry 30:3875–3883
Silverman JA, Perlmutter NG, Shapiro HM (2003) Correlation of daptomycin bactericidal activity and membrane depolarization in Staphylococcus aureus. Antimicob Agents Chemother 47:2538–2544
Sueyoshi E, Liu H, Matusunami K, Otsuka H, Shinzato T, Aramoto M, Takeda Y (2006) Bridelionosides A-F: megastigmane glucosides from Bridelia glauca F. balansae. Phytochemistry 67:2483–2493
Takeda Y, Mima C, Masuda T, Hirata E, Takushi A, Otsuka H (1998) Glochidioboside, a glucoside of (7S, 8R)-dihydrodehydrodiconiferyl alcohol from leaves of glochidion obovatum. Phytochemistry 49:2137–2139
Tamba Y, Ariyama H, Levadny V, Yamazaki M (2010) Kinetic pathway of antimicrobial peptide magainin 2-induced pore formation in lipid membranes. J Phys Chem B 114:12018–12026
Wang C, Chen T, Zhang N, Yang M, Li B, Lü X, Cao X, Ling C (2009) Melittin, a major component of bee venom, sensitizes human hepatocellular carcinoma cells to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis by activating CaMKII-TAK1-JNK/p38 and inhibiting IkappaBalpha kinase-NFkappaB. J Biol Chem 6:3804–3813
Yang L, Harroun TA, Weiss TM, Ding L, Huang HW (2001) Barrel-stave model or toroidal model? A case study on melittin pores. Biophys J 81:1475–1485
Yoneyama F, Imura Y, Ohno K, Zendo T, Nakayama J, Matsuzaki K, Sonomoto K (2009) Peptide-lipid huge toroidal pore, a new antimicrobial mechanism mediated by a lactococcal bacteriocin. lactin Q. Antimicrob Agents Chemother 53:3211–3217
Acknowledgments
This work was supported by a grant from the Next-Generation BioGreen 21 Program (Project No. PJ01104303), Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, H., Woo, ER. & Lee, D.G. Glochidioboside Kills Pathogenic Bacteria by Membrane Perturbation. Curr Microbiol 71, 1–7 (2015). https://doi.org/10.1007/s00284-015-0807-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-015-0807-9