Abstract
Purpose
Thiopurine drugs like 6-Mercaptopurine (6MP) are the cornerstone of maintenance therapy in acute lymphoblastic leukemia (ALL). A recently described variant in alpha-ketoglutarate dependent dioxygenase (FTO) gene has been reported to play an important role in thiopurine induced myelosuppression.
Methods
In this study, we genotyped a coding variant (p.Ala134Thr, rs79206939) and an intronic variant (rs16952570) of FTO in 174 Indian children (age ≤ 12 years) with ALL on maintenance phase of chemotherapy and examined correlation with the risk of thiopurine induced myelosuppression and hepatic toxicity.
Results
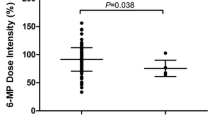
The prevalence of FTO-rs16952570 polymorphism was 18.4% (32/174) with 142 (82%) cases having TT genotype, 26 (15%) cases with TC genotype and 6 (3.4%) cases having CC genotype. FTO-rs79206939 was absent and non-polymorphic in our study group. The mean dose of 6-MP during 36 weeks of maintenance of TT, TC and CC carriers of FTO-rs16952570 was 53.7, 53.6 and 54.1 mg/m2/day. Number of patients tolerating starting dose of 60 mg/m2/day was significantly higher in CC (50%) than TT/TC (14%) genotype carrying cases (p = 0.014). However, no statistical significance was observed for total leukocyte count (TLC), absolute neutrophil count (ANC) as well as for platelets counts in patients harboring FTO-rs16952570 TT/TC/CC genotype at 4, 8, 12, 24 and 36 weeks after start of thiopurine therapy. Further, no significant correlation was noted between number of weeks of chemotherapy interruptions or episodes of febrile neutropenia and no evidence of hepatotoxicity was found with the genotype studied.
Conclusion
Polymorphism in FTO-rs16952570 did not show any correlation with thiopurine related toxicity in ALL patients.

Similar content being viewed by others
Data availability
Data will be made available on reasonable request.
References
Inaba H, Greaves M, Mullighan CG (2013) Acute lymphoblastic leukaemia. Lancet (Lond, Engl) 381:1943–1955
Hunger SP, Lu X, Devidas M, Camitta BM, Gaynon PS, Winick NJ, Reaman GH, Carroll WL (2012) Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: a report from the children’s oncology group. J Clin Oncol 30:1663–1669
Zaza G, Cheok M, Krynetskaia N et al (2010) Thiopurine pathway. Pharmacogenet Genom 20:573–574
Relling MV, Hancock ML, Rivera GK, Sandlund JT, Ribeiro RC, Krynetski EY, Pui CH, Evans WE (1999) Mercaptopurine therapy intolerance and heterozygosity at the thiopurine S-methyltransferase gene locus. J Natl Cancer Inst 91:2001–2008
Lennard L (2014) Implementation of TPMT testing. Br J Clin Pharmacol 77:704–714
Yang JJ, Landier W, Yang W et al (2015) Inherited NUDT15 variant is a genetic determinant of mercaptopurine intolerance in children with acute lymphoblastic leukemia. J Clin Oncol 33:1235–1242
Moriyama T, Yang W, Smith C, Pui CH, Evans WE, Relling MV, Bhatia S, Yang JJ (2022) Comprehensive characterization of pharmacogenetic variants in TPMT and NUDT15 in children with acute lymphoblastic leukemia. Pharmacogenet Genom 32:60–66
Wang Q, Mailloux J, Schwarz UI, Kim RB, Wilson A (2022) A novel NUDT15 variant identified in Caucasian TPMT wild type patients with inflammatory bowel disease and azathioprine-related myelotoxicity. Pharmacogenet Genom 32:39–41
Puangpetch A, Tiyasirichokchai R, Pakakasama S, Wiwattanakul S, Anurathapan U, Hongeng S, Sukasem C (2020) NUDT15 genetic variants are related to thiopurine-induced neutropenia in Thai children with acute lymphoblastic leukemia. Pharmacogenomics 21:403–410
Khera S, Trehan A, Bhatia P, Singh M, Bansal D, Varma N (2019) Prevalence of TPMT, ITPA and NUDT 15 genetic polymorphisms and their relation to 6MP toxicity in north Indian children with acute lymphoblastic leukemia. Cancer Chemother Pharmacol 83:341–348
Wang DS, Yu CH, Lin CY et al (2021) Childhood acute lymphoblastic leukemia mercaptopurine intolerance is associated with NUDT15 variants. Pediatr Res 89:217–222
Kim HS, Cheon JH, Jung ES et al (2017) A coding variant in FTO confers susceptibility to thiopurine-induced leukopenia in East Asian patients with IBD. Gut 66:1926–1935
Chen S, Tan WZ, Sutiman N et al (2020) An intronic FTO variant rs16952570 confers protection against thiopurine-induced myelotoxicities in multiethnic Asian IBD patients. Pharmacogenom J 20:505–515
Das N, Banavali S, Bakhshi S et al (2022) Protocol for ICiCLe-ALL-14 (InPOG-ALL-15-01): a prospective, risk stratified, randomised, multicentre, open label, controlled therapeutic trial for newly diagnosed childhood acute lymphoblastic leukaemia in India. Trials. https://doi.org/10.1186/S13063-022-06033-1
Schmiegelow K, Nielsen SN, Frandsen TL, Nersting J (2014) Mercaptopurine/methotrexate maintenance therapy of childhood acute lymphoblastic leukemia: clinical facts and fiction. J Pediatr Hematol Oncol 36:503–517
Bhatia S, Landier W, Shangguan M et al (2012) Nonadherence to oral mercaptopurine and risk of relapse in Hispanic and non-Hispanic white children with acute lymphoblastic leukemia: a report from the children’s oncology group. J Clin Oncol 30:2094–2101
Prognostic importance of 6-mercaptopurine dose intensity in acute lymphoblastic leukemia—PubMed. https://pubmed.ncbi.nlm.nih.gov/10216075/. Accessed 6 Sep 2022
Zimm S, Collins JM, Riccardi R, O’Neill D, Narang PK, Chabner B, Poplack DG (1983) Variable bioavailability of oral mercaptopurine. Is maintenance chemotherapy in acute lymphoblastic leukemia being optimally delivered? N Engl J Med 308:1005–1009
Lafolie P, Hayder S, Bjork O, Ahström L, Liliemark J, Peterson C (1986) Large interindividual variations in the pharmacokinetics of oral 6-mercaptopurine in maintenance therapy of children with acute leukaemia and non-Hodgkin lymphoma. Acta Paediatr Scand 75:797–803
Sato T, Takagawa T, Kakuta Y et al (2017) NUDT15, FTO, and RUNX1 genetic variants and thiopurine intolerance among Japanese patients with inflammatory bowel diseases. Intest Res 15:328–337
Yao L, Yin H, Hong M, Wang Y, Yu T, Teng Y, Li T, Wu Q (2021) RNA methylation in hematological malignancies and its interactions with other epigenetic modifications. Leukemia 35:1243–1257
Huang H, Wang Y, Kandpal M et al (2020) FTO-dependent N 6-methyladenosine modifications inhibit ovarian cancer stem cell self-renewal by blocking cAMP signaling. Cancer Res 80:3200–3214
Liu J, Ren D, Du Z, Wang H, Zhang H, Jin Y (2018) m 6 A demethylase FTO facilitates tumor progression in lung squamous cell carcinoma by regulating MZF1 expression. Biochem Biophys Res Commun 502:456–464
Li J, Han Y, Zhang H, Qian Z, Jia W, Gao Y, Zheng H, Li B (2019) The m6A demethylase FTO promotes the growth of lung cancer cells by regulating the m6A level of USP7 mRNA. Biochem Biophys Res Commun 512:479–485
Zejuan Li A, Weng H, Su R, Jin J, He C, Chen J (2017) FTO plays an oncogenic role in acute myeloid leukemia as a N 6-methyladenosine RNA demethylase accession numbers GSE34184 GSE30285 GSE76414 GSE84944 GSE85008. Cancer Cell 31:127–141
Acknowledgements
The study was funded by intramural special research grant for MD/DM from Postgraduate Institute of Medical Education & Research, Chandigarh.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, M., Bhaskar, D., Bhatia, P. et al. Evaluation of FTO polymorphism in 6-mercaptopurine related intolerance in children with acute lymphoblastic leukemia. Cancer Chemother Pharmacol 92, 51–56 (2023). https://doi.org/10.1007/s00280-023-04546-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-023-04546-3