Abstract
Purpose
Veliparib is an oral inhibitor of poly (ADP-ribose) polymerase (PARP)-1 and -2. PARP-1 expression may be increased in cancer, and this increase confers resistance to cytotoxic agents. We aimed to determine the recommended phase 2 dose (RP2D), maximum tolerated dose (MTD), dose-limiting toxicity (DLT), and pharmacokinetics (PK) of veliparib combined with paclitaxel and carboplatin.
Methods
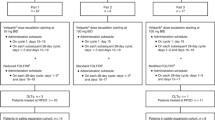
Eligibility criteria included patients with advanced solid tumors treated with ≤ 3 prior regimens. Paclitaxel and carboplatin were administered on day 3 of a 21-day cycle. Veliparib was given PO BID days 1–7, except for cycle 1 in the first 46 patients to serve as control for toxicity and PK. A standard “3 + 3” design started veliparib at 10 mg BID, paclitaxel at 150 mg/m2, and carboplatin AUC 6. The pharmacokinetic (PK) disposition of veliparib, paclitaxel, and carboplatin was determined by LC–MS/MS and AAS during cycles 1 and 2.
Results
Seventy-three patients were enrolled. Toxicities were as expected with carboplatin/paclitaxel chemotherapy, including neutropenia, thrombocytopenia, and peripheral neuropathy. DLTs were seen in two of seven evaluable patients at the maximum administered dose (MAD): veliparib 120 mg BID, paclitaxel 200 mg/m2, and carboplatin AUC 6 (febrile neutropenia, hyponatremia). The MTD and RP2D were determined to be veliparib 100 mg BID, paclitaxel 200 mg/m2, and carboplatin AUC 6. Median number of cycles of the three-agent combination was 4 (1–16). We observed 22 partial and 5 complete responses. Veliparib did not affect paclitaxel or carboplatin PK disposition.
Conclusion
Veliparib, paclitaxel, and carboplatin were well tolerated and demonstrated promising antitumor activity.


Similar content being viewed by others
References
Ray Chaudhuri A, Nussenzweig A (2017) The multifaceted roles of PARP1 in DNA repair and chromatin remodelling. Nat Rev Mol Cell Biol 18:610. https://doi.org/10.1038/nrm.2017.53
Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, Lopez E, Kyle S, Meuth M, Curtin NJ, Helleday T (2005) Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 434(7035):913–917. https://doi.org/10.1038/nature03443
Farmer H, McCabe N, Lord CJ, Tutt AN, Johnson DA, Richardson TB, Santarosa M, Dillon KJ, Hickson I, Knights C, Martin NM, Jackson SP, Smith GC, Ashworth A (2005) Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434(7035):917–921
Moynahan ME, Chiu JW, Koller BH, Jasin M (1999) Brca1 controls homology-directed DNA repair. Mol Cell 4(4):511–518
Moynahan ME, Pierce AJ, Jasin M (2001) BRCA2 is required for homology-directed repair of chromosomal breaks. Mol Cell 7(2):263–272
Bièche I, De Murcia G, Lidereau R (1996) Poly (ADP-ribose) polymerase gene expression status and genomic instability in human breast cancer. Clin Cancer Res 2(7):1163–1167
Molloy-Simard V, St-Laurent J-F, Vigneault F, Gaudreault M, Dargis N, Guérin M-C, Leclerc S, Morcos M, Black D, Molgat Y, Bergeron D, de Launoit Y, Boudreau F, Desnoyers S, Guérin S (2012) Altered expression of the poly(ADP-ribosyl)ation enzymes in uveal melanoma and regulation of PARG gene expression by the transcription factor ERM. Invest Ophthalmol Vis Sci 53(10):6219–6231. https://doi.org/10.1167/iovs.11-8853
Ossovskaya V, Koo IC, Kaldjian EP, Alvares C, Sherman BM (2010) Upregulation of poly (ADP-ribose) polymerase-1 (PARP1) in triple-negative breast cancer and other primary human tumor types. Genes Cancer 1(8):812–821. https://doi.org/10.1177/1947601910383418
Salemi M, Galia A, Fraggetta F, La Corte C, Pepe P, La Vignera S, Improta G, Bosco P, Calogero AE (2013) Poly (ADP-ribose) polymerase 1 protein expression in normal and neoplastic prostatic tissue. Eur J Histochem 57(2):e13. https://doi.org/10.4081/ejh.2013.e13
Sulzyc-Bielicka V, Domagala P, Hybiak J, Majewicz-Broda A, Safranow K, Domagala W (2012) Colorectal cancers differ in respect of PARP-1 protein expression. Polish J Pathol 63(2):87–92
Klauschen F, von Winterfeld M, Stenzinger A, Sinn BV, Budczies J, Kamphues C, Bahra M, Wittschieber D, Weichert W, Striefler J, Riess H, Dietel M, Denkert C (2012) High nuclear poly-(ADP-ribose)-polymerase expression is prognostic of improved survival in pancreatic cancer. Histopathology 61(3):409–416. https://doi.org/10.1111/j.1365-2559.2012.04225.x
Galia A, Calogero AE, Condorelli R, Fraggetta F, La Corte A, Ridolfo F, Bosco P, Castiglione R, Salemi M (2012) PARP-1 protein expression in glioblastoma multiforme. Eur J Histochem 56(1):e9. https://doi.org/10.4081/ejh.2012.e9
Calabrese CR, Almassy R, Barton S, Batey MA, Calvert AH, Canan-Koch S, Durkacz BW, Hostomsky Z, Kumpf RA, Kyle S, Li J, Maegley K, Newell DR, Notarianni E, Stratford IJ, Skalitzky D, Thomas HD, Wang LZ, Webber SE, Williams KJ, Curtin NJ (2004) Anticancer chemosensitization and radiosensitization by the novel poly(ADP-ribose) polymerase-1 inhibitor AG14361. J Natl Cancer Inst 96(1):56–67
Horton JK, Wilson SH (2013) Strategic combination of DNA-damaging agent and PARP inhibitor results in enhanced cytotoxicity. Front Oncol 3:257. https://doi.org/10.3389/fonc.2013.00257
Chen A (2011) PARP inhibitors: its role in treatment of cancer. Chin J Cancer 30(7):463–471. https://doi.org/10.5732/cjc.011.10111
Huggins-Puhalla S, JH Beumer, LJ Appleman, HA Tawbi, RG Stoller, Y Lin, B Kiesel, AR. Tan, D Gibbon, Y Jiang, A Garcia, HK. Chew, R Morgan, SP Shepherd, VL Giranda, AP Chen, CP Belani, E Chu (2012) A phase I study of chronically dosed, single-agent veliparib (ABT-888) in patients (pts) with either BRCA 1/2-mutated cancer (BRCA +), platinum-refractory ovarian cancer, or basal-like breast cancer (BRCA-wt). J Clinical Oncol 30 (Supplement): Abstr 3054
Kaufman B, Shapira-Frommer R, Schmutzler RK, Audeh MW, Friedlander M, Balmana J, Mitchell G, Fried G, Stemmer SM, Hubert A, Rosengarten O, Steiner M, Loman N, Bowen K, Fielding A, Domchek SM (2015) Olaparib monotherapy in patients with advanced cancer and a germline BRCA1/2 mutation. J Clin Oncol 33(3):244–250. https://doi.org/10.1200/jco.2014.56.2728
Tutt A, Robson M, Garber JE, Domchek SM, Audeh MW, Weitzel JN, Friedlander M, Arun B, Loman N, Schmutzler RK, Wardley A, Mitchell G, Earl H, Wickens M, Carmichael J (2010) Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and advanced breast cancer: a proof-of-concept trial. Lancet 376(9737):235–244. https://doi.org/10.1016/s0140-6736(10)60892-6
Mateo J, Carreira S, Sandhu S, Miranda S, Mossop H, Perez-Lopez R, Nava Rodrigues D, Robinson D, Omlin A, Tunariu N, Boysen G, Porta N, Flohr P, Gillman A, Figueiredo I, Paulding C, Seed G, Jain S, Ralph C, Protheroe A, Hussain S, Jones R, Elliott T, McGovern U, Bianchini D, Goodall J, Zafeiriou Z, Williamson CT, Ferraldeschi R, Riisnaes R, Ebbs B, Fowler G, Roda D, Yuan W, Wu YM, Cao X, Brough R, Pemberton H, A’Hern R, Swain A, Kunju LP, Eeles R, Attard G, Lord CJ, Ashworth A, Rubin MA, Knudsen KE, Feng FY, Chinnaiyan AM, Hall E, de Bono JS (2015) DNA-repair defects and olaparib in metastatic prostate cancer. N Engl J Med 373(18):1697–1708. https://doi.org/10.1056/NEJMoa1506859
Michels J, Vitale I, Senovilla L, Enot DP, Garcia P, Lissa D, Olaussen KA, Brenner C, Soria JC, Castedo M, Kroemer G (2013) Synergistic interaction between cisplatin and PARP inhibitors in non-small cell lung cancer. Cell cycle 12(6):877–883. https://doi.org/10.4161/cc.24034
Rottenberg S, Jaspers JE, Kersbergen A, van der Burg E, Nygren AO, Zander SA, Derksen PW, de Bruin M, Zevenhoven J, Lau A, Boulter R, Cranston A, O’Connor MJ, Martin NM, Borst P, Jonkers J (2008) High sensitivity of BRCA1-deficient mammary tumors to the PARP inhibitor AZD2281 alone and in combination with platinum drugs. Proc Natl Acad Sci USA 105(44):17079–17084. https://doi.org/10.1073/pnas.0806092105
Hastak K, Alli E, Ford JM (2010) Synergistic chemosensitivity of triple-negative breast cancer cell lines to poly(ADP-ribose) polymerase inhibition, gemcitabine, and cisplatin. Cancer Res 70(20):7970–7980. https://doi.org/10.1158/0008-5472.can-09-4521
Middleton MR, Friedlander P, Hamid O, Daud A, Plummer R, Falotico N, Chyla B, Jiang F, McKeegan E, Mostafa NM, Zhu M, Qian J, McKee M, Luo Y, Giranda VL, McArthur GA (2015) Randomized phase II study evaluating veliparib (ABT-888) with temozolomide in patients with metastatic melanoma. Ann Oncol 26(10):2173–2179. https://doi.org/10.1093/annonc/mdv308
Pietanza MC, Waqar SN, Krug LM, Dowlati A, Hann CL, Chiappori A, Owonikoko TK, Woo KM, Cardnell RJ, Fujimoto J, Long L, Diao L, Wang J, Bensman Y, Hurtado B, de Groot P, Sulman EP, Wistuba II, Chen A, Fleisher M, Heymach JV, Kris MG, Rudin CM, Byers LA (2018) Randomized, double-blind, phase II study of temozolomide in combination with either veliparib or placebo in patients with relapsed-sensitive or refractory small-cell lung cancer. J Clin Oncol 36(23):2386–2394. https://doi.org/10.1200/jco.2018.77.7672
Kummar S, Ji J, Morgan R, Lenz HJ, Puhalla SL, Belani CP, Gandara DR, Allen D, Kiesel B, Beumer JH, Newman EM, Rubinstein L, Chen A, Zhang Y, Wang L, Kinders RJ, Parchment RE, Tomaszewski JE, Doroshow JH (2012) A phase I study of veliparib in combination with metronomic cyclophosphamide in adults with refractory solid tumors and lymphomas. Clin Cancer Res 18(6):1726–1734. https://doi.org/10.1158/1078-0432.ccr-11-2821
Belani CP, Kearns CM, Zuhowski EG, Erkmen K, Hiponia D, Zacharski D, Engstrom C, Ramanathan RK, Capozzoli MJ, Aisner J, Egorin MJ (1999) Phase I trial, including pharmacokinetic and pharmacodynamic correlations, of combination paclitaxel and carboplatin in patients with metastatic non-small-cell lung cancer. J Clin Oncol 17(2):676–684
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst 92(3):205–216. https://doi.org/10.1093/jnci/92.3.205
Parise RA, Shawaqfeh M, Egorin MJ, Beumer JH (2008) Liquid chromatography-mass spectrometric assay for the quantitation in human plasma of ABT-888, an orally available, small molecule inhibitor of poly(ADP-ribose) polymerase. J Chromatogr B Analyt Technol Biomed Life Sci 872(1–2):141–147. https://doi.org/10.1016/j.jchromb.2008.07.032
Colville H, Dzadony R, Kemp R, Stewart S, Zeh HJ 3rd, Bartlett DL, Holleran J, Schombert K, Kosovec JE, Egorin MJ, Beumer JH (2010) In vitro circuit stability of 5-fluorouracil and oxaliplatin in support of hyperthermic isolated hepatic perfusion. J Extra-Corpor Technol 42(1):75–79
Parise RA, Ramanathan RK, Zamboni WC, Egorin MJ (2003) Sensitive liquid chromatography-mass spectrometry assay for quantitation of docetaxel and paclitaxel in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 783(1):231–236
Sheiner LB, Beal SL (1981) Some suggestions for measuring predictive performance. J Pharmacokinet Biopharm 9(4):503–512
Joerger M, Kraff S, Jaehde U, Hilger RA, Courtney JB, Cline DJ, Jog S, Baburina I, Miller MC, Salamone SJ (2017) Validation of a Commercial assay and decision support tool for routine paclitaxel therapeutic drug monitoring (TDM). Ther Drug Monit 39(6):617–624. https://doi.org/10.1097/ftd.0000000000000446
Joerger M, von Pawel J, Kraff S, Fischer JR, Eberhardt W, Gauler TC, Mueller L, Reinmuth N, Reck M, Kimmich M, Mayer F, Kopp HG, Behringer DM, Ko YD, Hilger RA, Roessler M, Kloft C, Henrich A, Moritz B, Miller MC, Salamone SJ, Jaehde U (2016) Open-label, randomized study of individualized, pharmacokinetically (PK)-guided dosing of paclitaxel combined with carboplatin or cisplatin in patients with advanced non-small-cell lung cancer (NSCLC). Ann Oncol 27(10):1895–1902. https://doi.org/10.1093/annonc/mdw290
Salem AH, Giranda VL, Mostafa NM (2014) Population pharmacokinetic modeling of veliparib (ABT-888) in patients with non-hematologic malignancies. Clin Pharmacokinet 53(5):479–488. https://doi.org/10.1007/s40262-013-0130-1
Nakada T, Kudo T, Kume T, Kusuhara H, Ito K (2018) Quantitative analysis of elevation of serum creatinine via renal transporter inhibition by trimethoprim in healthy subjects using physiologically-based pharmacokinetic model. Drug Metab Pharmacokinet 33(1):103–110. https://doi.org/10.1016/j.dmpk.2017.11.314
US Dept of Health and Human Services FDA, Center for Drug Evaluation and Research (2016) Rucaparib: multidiscipline review summary. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/209115Orig1s000Approv.pdf. Accessed 20 Apr 2019
McCormick A, Swaisland H (2017) In vitro assessment of the roles of drug transporters in the disposition and drug-drug interaction potential of olaparib. Xenobiotica 47(10):903–915. https://doi.org/10.1080/00498254.2016.1241449
Beumer JH, Inker LA, Levey AS (2018) Improving carboplatin dosing based on estimated GFR. Am J Kidney Dis 71(2):163–165. https://doi.org/10.1053/j.ajkd.2017.10.005
Cockcroft DW, Gault MH (1976) Prediction of creatinine clearance from serum creatinine. Nephron 16(1):31–41. https://doi.org/10.1159/000180580
German P, Liu HC, Szwarcberg J, Hepner M, Andrews J, Kearney BP, Mathias A (2012) Effect of cobicistat on glomerular filtration rate in subjects with normal and impaired renal function. J Acquir Immune Defic Syndr 61(1):32–40. https://doi.org/10.1097/QAI.0b013e3182645648
Calvert AH, Newell DR, Gumbrell LA, O’Reilly S, Burnell M, Boxall FE, Siddik ZH, Judson IR, Gore ME, Wiltshaw E (1989) Carboplatin dosage: prospective evaluation of a simple formula based on renal function. J Clin Oncol 7(11):1748–1756. https://doi.org/10.1200/jco.1989.7.11.1748
Fehr M, Maranta AF, Reichegger H, Gillessen S, Cathomas R (2018) Carboplatin dose based on actual renal function: no excess of acute haematotoxicity in adjuvant treatment in seminoma stage I. ESMO open 3(3):e000320. https://doi.org/10.1136/esmoopen-2018-000320
Wesson L (1969) Physiology of the human kidney. Grune and Stratton, New York
Rajan A, Carter CA, Kelly RJ, Gutierrez M, Kummar S, Szabo E, Yancey MA, Ji J, Mannargudi B, Woo S, Spencer S, Figg WD, Giaccone G (2012) A phase I combination study of olaparib with cisplatin and gemcitabine in adults with solid tumors. Clin Cancer Res 18(8):2344–2351. https://doi.org/10.1158/1078-0432.CCR-11-2425
van der Noll R, Ang J, Jager A, Marchetti S, Mergui-Roelvink M, De Bono J, Lolkema M, Brunetto A, Arkenau H, De Jonge M, van der Biessen D, Tchakov I, Bowen K, Schellens J (2013) Phase I study of olaparib in combination with carboplatin and/or paclitaxel in patients with advanced solid tumors. J Clin Oncol 31: (suppl; abstr 2579)
Dent RA, Lindeman GJ, Clemons M, Wildiers H, Chan A, McCarthy NJ, Singer CF, Lowe ES, Watkins CL, Carmichael J (2013) Phase I trial of the oral PARP inhibitor olaparib in combination with paclitaxel for first- or second-line treatment of patients with metastatic triple-negative breast cancer. Breast Cancer Res 15(5):R88. https://doi.org/10.1186/bcr3484
Kummar S, Chen A, Ji J, Zhang Y, Reid JM, Ames M, Jia L, Weil M, Speranza G, Murgo AJ, Kinders R, Wang L, Parchment RE, Carter J, Stotler H, Rubinstein L, Hollingshead M, Melillo G, Pommier Y, Bonner W, Tomaszewski JE, Doroshow JH (2011) Phase I study of PARP inhibitor ABT-888 in combination with topotecan in adults with refractory solid tumors and lymphomas. Cancer Res 71(17):5626–5634. https://doi.org/10.1158/0008-5472.can-11-1227
Ramalingam SS, Blais N, Mazieres J, Reck M, Jones CM, Juhasz E, Urban L, Orlov S, Barlesi F, Kio E, Keiholz U, Qin Q, Qian J, Nickner C, Dziubinski J, Xiong H, Ansell P, McKee M, Giranda V, Gorbunova V (2017) Randomized, placebo-controlled, phase II study of veliparib in combination with carboplatin and paclitaxel for advanced/metastatic non-small cell lung cancer. Clin Cancer Res 23(8):1937–1944. https://doi.org/10.1158/1078-0432.ccr-15-3069
Mizugaki H, Yamamoto N, Nokihara H, Fujiwara Y, Horinouchi H, Kanda S, Kitazono S, Yagishita S, Xiong H, Qian J, Hashiba H, Shepherd SP, Giranda V, Tamura T (2015) A phase 1 study evaluating the pharmacokinetics and preliminary efficacy of veliparib (ABT-888) in combination with carboplatin/paclitaxel in Japanese subjects with non-small cell lung cancer (NSCLC). Cancer Chemother Pharmacol 76(5):1063–1072. https://doi.org/10.1007/s00280-015-2876-7
Han HS, Dieras V, Robson M, Palacova M, Marcom PK, Jager A, Bondarenko I, Citrin D, Campone M, Telli ML, Domchek SM, Friedlander M, Kaufman B, Garber JE, Shparyk Y, Chmielowska E, Jakobsen EH, Kaklamani V, Gradishar W, Ratajczak CK, Nickner C, Qin Q, Qian J, Shepherd SP, Isakoff SJ, Puhalla S (2018) Veliparib with temozolomide or carboplatin/paclitaxel versus placebo with carboplatin/paclitaxel in patients with BRCA1/2 locally recurrent/metastatic breast cancer: randomized phase II study. Ann Oncol 29(1):154–161. https://doi.org/10.1093/annonc/mdx505
Acknowledgements
The authors would like to express their sincere thanks to the patients and their family members who were enrolled in this study. The authors would also like to thank the clinical research staff who were actively involved in the conduct of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Financial support
The study was supported in part by the following grants: NIH/NCI UM1-CA186690, NIH/NCI U01-CA099168. This project used the UPMC Hillman Cancer Pharmacokinetics and Pharmacodynamics Facility (CPPF), Biostatistics Facility (BF), and Clinical Research Services (CRS), and was supported in part by award NIH/NCI P30-CA47904.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Appleman, L.J., Beumer, J.H., Jiang, Y. et al. Phase 1 study of veliparib (ABT-888), a poly (ADP-ribose) polymerase inhibitor, with carboplatin and paclitaxel in advanced solid malignancies. Cancer Chemother Pharmacol 84, 1289–1301 (2019). https://doi.org/10.1007/s00280-019-03960-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-019-03960-w