Abstract
Purpose
GTI-2040 is a novel antisense oligonucleotide inhibitor of the R2 subunit of ribonucleotide reductase. This phase I study assessed safety and maximum tolerated dose (MTD) of GTI-2040 in combination with weekly gemcitabine in patients with advanced solid tumors.
Methods
GTI-2040 was given as a 14-day continuous intravenous infusion, while gemcitabine was administered on days 1, 8, and 15. This combination was repeated every 4 weeks and study followed a modified 3 + 3 Fibonacci schema. Incidence, severity of adverse events, pharmacokinetics (PK), and pharmacodynamics (PD) was assessed. Responses were assessed using RECIST criteria version 1.0 with CT scans performed after every other cycle.
Results
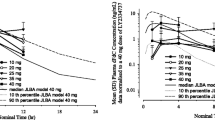
A total of 16 patients received at least one dose of GTI-2040/gemcitabine and were included in the safety analysis. The MTD of this regimen is 100 mg/m2/day of GTI-2040 over 14 days combined with 400 mg/m2/day of gemcitabine administered weekly on days 1, 8, and 15. The dose-limiting toxicities (DLTs) included grade 3 fatigue and thrombocytopenia with hematemesis (both at 100/600 mg/m2/day). The most common adverse events were grade 1/2 fatigue, nausea, vomiting, diarrhea, and anorexia. There was no evidence of alteration in gemcitabine PKs. PD modulation of R2mRNA expression in peripheral blood mononuclear cells was observed. No objective tumor response was observed although stable disease was seen in 25% patients.
Conclusions
The combination of GTI-2040 and gemcitabine has an acceptable safety profile in a heavily pre-treated patient population with advanced solid tumors. No clear signal of anti-tumor activity was observed; however, several patients had prolonged stable disease.


Similar content being viewed by others
References
Herrick J, Sclavi B (2007) Ribonucleotide reductase and the regulation of DNA replication: an old story and an ancient heritage. Mol Microbiol 63(1):22–34. https://doi.org/10.1111/j.1365-2958.2006.05493.x
Cerqueira NM, Pereira S, Fernandes PA, Ramos MJ (2005) Overview of ribonucleotide reductase inhibitors: an appealing target in anti-tumour therapy. Curr Med Chem 12(11):1283–1294
Jordan A, Reichard P (1998) Ribonucleotide reductases. Annu Rev Biochem 67:71–98. https://doi.org/10.1146/annurev.biochem.67.1.71
Anderson H, Hopwood P, Stephens RJ, Thatcher N, Cottier B, Nicholson M, Milroy R, Maughan TS, Falk SJ, Bond MG, Burt PA, Connolly CK, McIllmurray MB, Carmichael J (2000) Gemcitabine plus best supportive care (BSC) vs BSC in inoperable non-small cell lung cancer—a randomized trial with quality of life as the primary outcome. UK NSCLC gemcitabine group. Non-small cell lung cancer. Br J Cancer 83(4):447–453. https://doi.org/10.1054/bjoc.2000.1307
Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, Zhu J, Johnson DH (2002) Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 346(2):92–98. https://doi.org/10.1056/NEJMoa011954
von Minckwitz G, Bauknecht T, Visseren-Grul CM, Neijt JP (1999) Phase II study of gemcitabine in ovarian cancer. Ann Oncol 10(7):853–855
du Bois A, Luck HJ, Pfisterer J, Schroeder W, Blohmer JU, Kimmig R, Moebus V, Quaas J (2001) Second-line carboplatin and gemcitabine in platinum sensitive ovarian cancer—a dose-finding study by the Arbeitsgemeinschaft Gynakologische Onkologie (AGO) Ovarian Cancer Study Group. Ann Oncol 12(8):1115–1120
von der Maase H, Hansen SW, Roberts JT, Dogliotti L, Oliver T, Moore MJ, Bodrogi I, Albers P, Knuth A, Lippert CM, Kerbrat P, Sanchez Rovira P, Wersall P, Cleall SP, Roychowdhury DF, Tomlin I, Visseren-Grul CM, Conte PF (2000) Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol 18(17):3068–3077. https://doi.org/10.1200/jco.2000.18.17.3068
Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M, Seay T, Tjulandin SA, Ma WW, Saleh MN, Harris M, Reni M, Dowden S, Laheru D, Bahary N, Ramanathan RK, Tabernero J, Hidalgo M, Goldstein D, Van Cutsem E, Wei X, Iglesias J, Renschler MF (2013) Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 369(18):1691–1703. https://doi.org/10.1056/NEJMoa1304369
Desai AA, Schilsky RL, Young A, Janisch L, Stadler WM, Vogelzang NJ, Cadden S, Wright JA, Ratain MJ (2005) A phase I study of antisense oligonucleotide GTI-2040 given by continuous intravenous infusion in patients with advanced solid tumors. Ann Oncol 16(6):958–965. https://doi.org/10.1093/annonc/mdi178
Stadler WM, Desai AA, Quinn DI, Bukowski R, Poiesz B, Kardinal CG, Lewis N, Makalinao A, Murray P, Torti FM (2008) A Phase I/II study of GTI-2040 and capecitabine in patients with renal cell carcinoma. Cancer Chemother Pharmacol 61(4):689–694. https://doi.org/10.1007/s00280-007-0524-6
Shibata SI, Doroshow JH, Frankel P, Synold TW, Yen Y, Gandara DR, Lenz HJ, Chow WA, Leong LA, Lim D, Margolin KA, Morgan RJ, Somlo G, Newman EM (2009) Phase I trial of GTI-2040, oxaliplatin, and capecitabine in the treatment of advanced metastatic solid tumors: a California Cancer Consortium Study. Cancer Chemother Pharmacol 64(6):1149–1155. https://doi.org/10.1007/s00280-009-0977-x
Leighl NB, Laurie SA, Chen XE, Ellis P, Shepherd FA, Knox JJ, Goss G, Burkes RL, Pond GR, Dick C, Yen Y, Zwiebel JA, Moore MJ (2009) A phase I/II study of GTI-2040 plus docetaxel as second-line treatment in advanced non-small cell lung cancer: a study of the PMH phase II consortium. J Thorac Oncol 4(9):1163–1169. https://doi.org/10.1097/JTO.0b013e3181a949b2
Sridhar SS, Canil CM, Chi KN, Hotte SJ, Ernst S, Wang L, Chen EX, Juhasz A, Yen Y, Murray P, Zwiebel JA, Moore MJ (2011) A phase II study of the antisense oligonucleotide GTI-2040 plus docetaxel and prednisone as first-line treatment in castration-resistant prostate cancer. Cancer Chemother Pharmacol 67(4):927–933. https://doi.org/10.1007/s00280-010-1389-7
Klisovic RB, Blum W, Wei X, Liu S, Liu Z, Xie Z, Vukosavljevic T, Kefauver C, Huynh L, Pang J, Zwiebel JA, Devine S, Byrd JC, Grever MR, Chan K, Marcucci G (2008) Phase I study of GTI-2040, an antisense to ribonucleotide reductase, in combination with high-dose cytarabine in patients with acute myeloid leukemia. Clin Cancer Res 14(12):3889–3895. https://doi.org/10.1158/1078-0432.ccr-08-0109
Funding
National Cancer Institute (U01CA069853) funded this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None to declare. All authors had full control of all primary data, which are available for review upon request.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Welfare of animals
This article does not contain any studies with animals performed by the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Malik, L., Zwiebel, A. & Cooper, J. A phase I pharmacokinetic and pharmacodynamic study of GTI-2040 in combination with gemcitabine in patients with solid tumors. Cancer Chemother Pharmacol 82, 533–539 (2018). https://doi.org/10.1007/s00280-018-3647-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3647-z