Abstract
Purpose
Sirolimus (rapamycin) is a mammalian target of rapamycin pathway blocker. The efficacy of sirolimus is currently studied for its antiproliferative properties in various malignancies and particularly in squamous cell carcinoma and other oral disorders. Topical application at the oral cavity can augment sirolimus availability at the site of action by increasing sirolimus levels in saliva and hence efficacy, along with improved safety (low levels in the blood to avoid side effects) and compliance. Our purpose was to evaluate the release profile and safety of a topical sirolimus sustained-release varnish drug delivery system.
Subjects and methods
Sirolimus sustained-release varnish drug delivery system containing a total of 0.5 mg of the drug was applied to nine healthy male volunteers. Saliva and blood levels were determined utilizing mass spectrometry and chemiluminescent microparticle immunoassay, respectively. The prolonged release profile and safety were evaluated for the oral topical delivery system.
Results
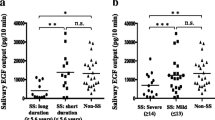
After the application of the drug delivery system, a sustained-release profile was observed in the oral cavity. We have measured moderate sirolimus levels for up to 12 h. The safety was confirmed, and systemic sirolimus blood levels were negligible.
Conclusions
After an application of sirolimus sustained-release varnish drug delivery system, prolonged drug levels can be achieved in the saliva. The oral topical sirolimus concentrations were potentially therapeutic along with minimal systemic exposure. These results broaden the potential clinical use of sustained-release oral topical rapalogs.




Similar content being viewed by others
References
Porta C, Paglino C, Mosca A (2014) Targeting PI3K/Akt/mTOR signaling in cancer. Front Oncol 4:64. doi:10.3389/fonc.2014.00064
Bose P, Brockton NT, Dort JC (2013) Head and neck cancer: from anatomy to biology. Int J Cancer 133(9):2013–2023. doi:10.1002/ijc.28112
Czerninski R, Amornphimoltham P, Patel V, Molinolo AA, Gutkind JS (2009) Targeting mammalian target of rapamycin by rapamycin prevents tumor progression in an oral-specific chemical carcinogenesis model. Cancer Prev Res 2(1):27–36. doi:10.1158/1940-6207.CAPR-08-0147
Fruman DA, Rommel C (2014) PI3K and cancer: lessons, challenges and opportunities. Nat Rev Drug Discov 13(2):140–156. doi:10.1038/nrd4204
Scully C, Bagan J (2009) Oral squamous cell carcinoma: overview of current understanding of aetiopathogenesis and clinical implications. Oral Dis 15(6):388–399. doi:10.1111/j.1601-0825.2009.01563.x
ClinicalTrails.gov. Rapamycin therapy in head and neck squamous cell carcinoma. https://clinicaltrials.gov/ct2/show/NCT01195922?term=%22sirolimus%22+And+%22head+and+neck%22&rank=2. Accessed Nov 2014
de Oliveira MA, Martins EMF, Wang Q, Sonis S, Demetri G, George S, Butrynski J, Treister NS (2011) Clinical presentation and management of mTOR inhibitor-associated stomatitis. Oral Oncol 47(10):998–1003. doi:10.1016/j.oraloncology.2011.08.009
Friedman M, Steinberg D (1990) Sustained-release delivery systems for treatment of dental diseases. Pharm Res 7(4):313–317
Steinberg D, Tal T, Friedman M (2006) Sustained-release delivery systems of triclosan for treatment of Streptococcus mutans biofilm. J Biomed Mater Res B Appl Biomater 77(2):282–286
Madke B (2013) Topical rapamycin (sirolimus) for facial angiofibromas. Indian Dermatol Online J 4(1):54–57. doi:10.4103/2229-5178.105488
Kremer M, Sliva K, Klemke CD, Schnierle BS (2010) Cutaneous T-cell lymphoma cells are sensitive to rapamycin. Exp Dermatol 19(9):800–805. doi:10.1111/j.1600-0625.2010.01102.x
Nguyen QD, Ibrahim MA, Watters A, Bittencourt M, Yohannan J, Sepah YJ, Dunn JP, Naor J, Shams N, Shaikh O, Leder HA, Do DV (2013) Ocular tolerability and efficacy of intravitreal and subconjunctival injections of sirolimus in patients with non-infectious uveitis: primary 6-month results of the SAVE Study. J Ophthalmic Inflamm Infect 3(1):32. doi:10.1186/1869-5760-3-32
Soria A, Agbo-Godeau S, Taieb A, Frances C (2009) Treatment of refractory oral erosive lichen planus with topical rapamycin: 7 cases. Dermatology 218(1):22–25. doi:10.1159/000172830
Alayev A, Holz MK (2013) mTOR signaling for biological control and cancer. J Cell Physiol 228(8):1658–1664. doi:10.1002/jcp.24351
Nguyen SA, Walker D, Gillespie MB, Gutkind JS, Day TA (2012) mTOR inhibitors and its role in the treatment of head and neck squamous cell carcinoma. Curr Treat Options Oncol 13(1):71–81. doi:10.1007/s11864-011-0180-2
Pallet N, Beaune P, Legendre C, Anglicheau D (2006) Rapamycine and mTOR inhibitors: from bench to bedside. Ann Biol Clin 64(2):107–115
Lodi G, Sardella A, Bez C, Demarosi F, Carrassi A (2006) Interventions for treating oral leukoplakia. The Cochrane database of systematic reviews (4):CD001829. doi:10.1002/14651858.CD001829.pub3
Khuri FR, Lee JJ, Lippman SM, Kim ES, Cooper JS, Benner SE, Winn R, Pajak TF, Williams B, Shenouda G, Hodson I, Fu K, Shin DM, Vokes EE, Feng L, Goepfert H, Hong WK (2006) Randomized phase III trial of low-dose isotretinoin for prevention of second primary tumors in stage I and II head and neck cancer patients. J Natl Cancer Inst 98(7):441–450. doi:10.1093/jnci/djj091
Lippman SM, Lee JJ, Martin JW, El-Naggar AK, Xu X, Shin DM, Thomas M, Mao L, Fritsche HA Jr, Zhou X, Papadimitrakopoulou V, Khuri FR, Tran H, Clayman GL, Hittelman WN, Hong WK, Lotan R (2006) Fenretinide activity in retinoid-resistant oral leukoplakia. Clin Cancer Res 12(10):3109–3114. doi:10.1158/1078-0432.CCR-05-2636
Nudelman Z, Friedman M, Barasch D, Nemirovski A, Findler M, Pikovsky A, Gutkind J, Czerninski R (2014) Levels of sirolimus in saliva and blood following mouthwash application. Oral Dis. doi:10.1111/odi.12229
Courtais C, Dupuy AM, Sophie Bargnoux A, Pageaux GP, Fegueux N, Mourad G, Cristol JP (2010) Evaluation of two sirolimus assays using the ARCHITECT-i1000((R)) CMIA or RxL((R)) ACMIA methods in comparison with the IMx((R)) MEIA method. Clin Chem Lab Med 48(10):1523–1525. doi:10.1515/CCLM.2010.288
ClinicalTrails.gov. Rapalogs in clinical trials for the treatment of oral squamous cell carcinoma. https://clinicaltrials.gov/ct2/results?term=%22Sirolimus%22+and+%22squamous+cell+carcinoma+%22&Search=Search. Accessed Nov 2014
Iglesias-Bartolome R, Patel V, Cotrim A, Leelahavanichkul K, Molinolo AA, Mitchell JB, Gutkind JS (2012) mTOR inhibition prevents epithelial stem cell senescence and protects from radiation-induced mucositis. Cell Stem Cell 11(3):401–414. doi:10.1016/j.stem.2012.06.007
Coppock JD, Wieking BG, Molinolo AA, Gutkind JS, Miskimins WK, Lee JH (2013) Improved clearance during treatment of HPV-positive head and neck cancer through mTOR inhibition. Neoplasia 15(6):620–630
Beyth N, Redlich M, Harari D, Friedman M, Steinberg D (2003) Effect of sustained-release chlorhexidine varnish on Streptococcus mutans and Actinomyces viscosus in orthodontic patients. Am J Orthod Dentofac Orthop 123(3):345–348. doi:10.1067/mod.2003.19
Steinberg D, Rozen R, Klausner EA, Zachs B, Friedman M (2002) Formulation, development and characterization of sustained release varnishes containing amine and stannous fluorides. Caries Res 36(6):411–416. doi:10.1159/000066539
Czerninski R, Sivan S, Steinberg D, Gati I, Kagan L, Friedman M (2010) A novel sustained-release clotrimazole varnish for local treatment of oral candidiasis. Clin Oral Investig 14(1):71–78. doi:10.1007/s00784-009-0275-3
Nudelman Z, Keshet N, Elhalal MD, Friedman M, Czerninski R (2013) Levels of sirolimus in saliva vs. blood—the rationale of topical oral use for oral malignancy. Anticancer Res 33(2):661–663
Acknowledgments
This study was supported by a Project Grant from the Israel Cancer Research Fund.
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nudelman, Z., Findler, M., Barasch, D. et al. Levels of sirolimus in saliva and blood following oral topical sustained-release varnish delivery system application. Cancer Chemother Pharmacol 75, 969–974 (2015). https://doi.org/10.1007/s00280-015-2721-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-015-2721-z