Abstract
Purpose
To evaluate safety of balugrastim, a recombinant human serum albumin and granulocyte colony-stimulating factor (G-CSF), administered over a range of therapeutic doses in women with breast cancer receiving doxorubicin plus docetaxel chemotherapy.
Methods
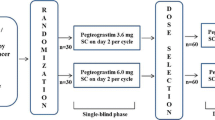
The phase I, sequential dose-escalation first segment compared subcutaneous balugrastim 50, 150, 300, and 450 µg/kg during chemotherapy cycles 0–2. The randomized (2:2:1), open-label, phase IIa second segment compared balugrastim 300 or 450 µg/kg with pegfilgrastim 6 mg during chemotherapy cycles 1 and 2.
Results
In the phase I segment, balugrastim was escalated to 450 µg/kg in 13 patients without dose-limiting toxicity. Three (9.7 %) of the 31 adverse events (AEs) reported in nine patients were grade 3 (agranulocytosis, vomiting, hypertension); none was grade 4. In the open-label phase IIa segment (N = 51), the majority of the 64 AEs reported in 31 (75.6 %) balugrastim-treated patients were grade 1 (59.4 %), with 39.1 % grade 2, 1.6 % grade 3 (one AE of vomiting), and none grade 4. Of the 16 AEs reported in seven (70.0 %) pegfilgrastim-treated patients, 87.5 % were grade 1, 6.3 % were grade 2, 6.3 % were grade 3 (one AE of thrombocytopenia), and none were grade 4. Overall, there were six bone pain AEs reported, one in the balugrastim 300 µg/kg group and five in the balugrastim 450 µg/kg group. No AEs in either study necessitated treatment interruption/discontinuation. The incidence and duration of grade 3–4 neutropenia were similar between balugrastim- and pegfilgrastim-treated patients.
Conclusions
Balugrastim was well tolerated in this small population of breast cancer patients.


Similar content being viewed by others
References
Pettengell R, Schwenkglenks M, Leonard R et al (2008) Neutropenia occurrence and predictors of reduced chemotherapy delivery: results from the INC-EU prospective observational European neutropenia study. Support Care Cancer 16:1299–1309
Joerger M, Thurlimann B (2013) Chemotherapy regimens in early breast cancer: major controversies and future outlook. Exp Rev Anticancer Ther 13:165–178
del Giglio A, Eniu A, Ganea-Motan D, Topuzov E, Lubenau H (2008) XM02 is superior to placebo and equivalent to Neupogen in reducing the duration of severe neutropenia and the incidence of febrile neutropenia in cycle 1 in breast cancer patients receiving docetaxel/doxorubicin chemotherapy. BMC Cancer 8:332–338
Alba E, Martin M, Ramos M et al (2004) Multicenter randomized trial comparing sequential with concomitant administration of doxorubicin and docetaxel as first-line treatment of metastatic breast cancer: a Spanish Breast Cancer Research Group (GEICAM-9903) phase III study. J Clin Oncol 22:2587–2593
Nabholtz JM, Falkson C, Campos D et al (2003) Docetaxel and doxorubicin compared with doxorubicin and cyclophosphamide as first-line chemotherapy for metastatic breast cancer: results of a randomized, multicenter, phase III trial. J Clin Oncol 21:968–975
Crawford J, Ozer H, Stoller R et al (1991) Reduction by granulocyte colony-stimulating factor of fever and neutropenia induced by chemotherapy in patients with small-cell lung cancer. N Engl J Med 325:164–170
Roberts AW (2005) G-CSF: a key regulator of neutrophil production, but that’s not all! Growth Factors 23:33–41
Neulasta [package insert] (2014) Amgen Inc., Thousand Oaks, CA
Yang BB, Kido A (2011) Pharmacokinetics and pharmacodynamics of pegfilgrastim. Clin Pharmacokinet 50:295–306
Elsadek B, Kratz F (2012) Impact of albumin on drug delivery–new applications on the horizon. J Control Release 157:4–28
Oken MM, Creech RH, Tormey DC et al (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5:649–655
NCCN Clinical Practice Guidelines in Oncology (2014) Myeloid Growth Factors v.2.2014. National Comprehensive Cancer Network. www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed 23 Dec 2014
Smith TJ, Khatcheressian J, Lyman GH et al (2006) 2006 update of recommendations for the use of white blood cell growth factors: an evidence-based clinical practice guideline. J Clin Oncol 24:3187–3205
Aapro MS, Bohlius J, Cameron DA et al (2011) 2010 update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumours. Eur J Cancer 47:8–32
Neupogen [package insert] (2013) Amgen Inc., Thousand Oaks, CA
Molineux G, Kinstler O, Briddell B et al (1999) A new form of Filgrastim with sustained duration in vivo and enhanced ability to mobilize PBPC in both mice and humans. Exp Hematol 27:1724–1734
Sleep D, Cameron J, Evans LR (2013) Albumin as a versatile platform for drug half-life extension. Biochim Biophys Acta 1830:5526–5534
Halpern W, Riccobene TA, Agostini H et al (2002) Albugranin, a recombinant human granulocyte colony stimulating factor (G-CSF) genetically fused to recombinant human albumin induces prolonged myelopoietic effects in mice and monkeys. Pharm Res 19:1720–1729
Johnston E, Crawford J, Blackwell S et al (2000) Randomized, dose-escalation study of SD/01 compared with daily filgrastim in patients receiving chemotherapy. J Clin Oncol 18:2522–2528
Acknowledgments
Medical writing and editorial assistance was provided by Ruth Sussman, Chameleon Communications, New York, NY, and Lisa Feder, PhD, Peloton Advantage, Parsippany, NJ, and was funded by Teva Branded Pharmaceutical Products R&D, Inc. The authors also acknowledge Scott Newcomer, MS, Teva Pharmaceuticals, Frazer, PA, and Pippa Loupe, PhD, Teva Pharmaceuticals, Kansas City, MO, for their medical writing and editorial assistance in the development of this manuscript. Teva provided a full review of the article. This study was sponsored by Teva Biopharmaceuticals USA (formerly CoGenesys, Inc).
Conflict of interest
Noa Avisar and Liat Adar are employees of Teva Pharmaceuticals, Netanya, Israel. Laurie Pukac, Jason Bock, and Steve Barash are employees of Teva Biopharmaceuticals, Rockville, MD, and Udo Müller is an employee of Teva GmbH, Ulm, Germany, both subsidiaries of Teva Pharmaceuticals. David Shen was an employee of Teva Pharmaceuticals at the time of this study and during manuscript preparation.
Ethical standard
This study was approved by the appropriate ethics committee at each study site and was conducted in accordance with the International Conference on Harmonisation guidelines for Good Clinical Practice and the current version of the Declaration of Helsinki (Tokyo, 2004). All study participants provided written informed consent prior to their participation.
Author information
Authors and Affiliations
Corresponding author
Additional information
EudraCT Number: 2006-005997-28
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Avisar, N., Adar, L., Bock, J. et al. First-in-human, phase I/IIa dose-escalation and safety study of balugrastim in breast cancer patients receiving myelosuppressive chemotherapy. Cancer Chemother Pharmacol 75, 929–939 (2015). https://doi.org/10.1007/s00280-015-2703-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-015-2703-1