Abstract
Purpose
We conducted a phase II study evaluating safety and efficacy of combination gemcitabine and capecitabine therapy for metastatic breast cancer patients following anthracycline and taxane treatment in Korea.
Methods
This was a single-arm, non-randomized phase II study. Patients received 1,000 mg/m2 gemcitabine intravenously over 30 min on days 1 and 8, and 1,250 mg/m2 capecitabine orally twice daily on days 1–14 until disease progression or intolerable toxicity occurred. This regimen was repeated every 3 weeks. The primary outcome assessed was overall response rate [ORR, complete response (CR) + partial response (PR) as the best response], and secondary outcomes were progression-free survival (PFS), overall survival (OS), disease control rate (DCR) [maintenance of CR + PR + stable disease (SD) for at least 3 months], drug toxicity, and predictive factors for response to this regimen.
Results
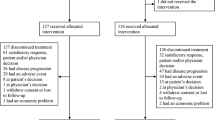
Of 41 patients, the ORR was 39.0 % (CR 0 %; PR 39.0 %), and DCR was 78.0 % using this chemotherapy. DCR for 6 and 12 months was 68.3 and 26.8 %, respectively. Median PFS was 10.0 months [95 % confidence interval (CI) 7.8–12.1], and median OS was 25.1 months (95 % CI 18.2–32.1). Prominent toxicities were neutropenia and hand–foot syndrome. Most adverse events were well known, relatively moderate, and reversible. Taxane sensitivity [odds ratio (OR) 0.169; 95 % CI 0.034–0.826; P = 0.028] and hepatic metastasis (OR 0.097; 95 % CI 0.017–0.559; P = 0.009) were significantly predictive of response to gemcitabine and capecitabine combination.
Conclusions
This study showed reproducible anticancer activity and tolerable toxicity of gemcitabine and capecitabine combination therapy in recurrent or metastatic Korean breast cancer patients previously treated with anthracycline and taxane.

Similar content being viewed by others
References
Chow LWC, Im Y-H (2008) Current treatment of locally advanced and metastatic breast cancer in the Asia-Pacific region: challenges and limitations. Asia-Pac J Clin Oncol 4:S14–S23. doi:10.1111/j.1743-7563.2008.00195.x
Bull JM, Tormey DC, Li SH, Carbone PP, Falkson G, Blom J, Perlin E, Simon R (1978) A randomized comparative trial of adriamycin versus methotrexate in combination drug therapy. Cancer 41:1649–1657
Roche H, Vahdat LT (2011) Treatment of metastatic breast cancer: second line and beyond. Ann Oncol 22:1000–1010. doi:10.1093/annonc/mdq429
Sledge GW, Neuberg D, Bernardo P, Ingle JN, Martino S, Rowinsky EK, Wood WC (2003) Phase III trial of doxorubicin, paclitaxel, and the combination of doxorubicin and paclitaxel as front-line chemotherapy for metastatic breast cancer: an intergroup trial (E1193). J Clin Oncol 21:588–592
Hortobagyi GN (2000) Developments in chemotherapy of breast cancer. Cancer 88:3073–3079
Rha SY, Moon YH, Jeung HC, Kim YT, Sohn JH, Yang WI, Suh CO, Kim GE, Roh JK, Chung HC (2005) Gemcitabine monotherapy as salvage chemotherapy in heavily pretreated metastatic breast cancer. Breast Cancer Res Treat 90:215–221. doi:10.1007/s10549-004-2468-4
Smorenburg CH, Bontenbal M, Seynaeve C, van Zuylen C, de Heus G, Verweij J, de Wit R (2001) Phase II study of weekly gemcitabine in patients with metastatic breast cancer relapsing or failing both an anthracycline and a taxane. Breast Cancer Res Treat 66:83–87
Modi S, Currie VE, Seidman AD, Bach AM, Panageas KS, Theodoulou M, Moasser MM, D’Andrea GM, Lake DE, Choi J, Norton L, Hudis CA (2005) A phase II trial of gemcitabine in patients with metastatic breast cancer previously treated with an anthracycline and taxane. Clin Breast Cancer 6:55–60. doi:10.3816/CBC.2005.n.009
Ershler WB (2006) Capecitabine monotherapy: safe and effective treatment for metastatic breast cancer. Oncologist 11:325–335. doi:10.1634/theoncologist.11-4-325
Fumoleau P, Largillier R, Clippe C, Dieras V, Orfeuvre H, Lesimple T, Culine S, Audhuy B, Serin D, Cure H, Vuillemin E, Morere JF, Montestruc F, Mouri Z, Namer M (2004) Multicentre, phase II study evaluating capecitabine monotherapy in patients with anthracycline- and taxane-pretreated metastatic breast cancer. Eur J Cancer 40:536–542. doi:10.1016/j.ejca.2003.11.007
Pierga JY, Fumoleau P, Brewer Y, Zelek L, Martin D, Turpin FL, Goudier MJ, Gil-Delgado M, Baticle JL, Namer M, Chollet P, Sutherland W, Barats JC, Cooperative Group of the French capecitabine compassionate use program (2004) Efficacy and safety of single agent capecitabine in pretreated metastatic breast cancer patients from the French compassionate use program. Breast Cancer Res Treat 88:117–129. doi:10.1007/s10549-004-1282-3
Pajk B, Cufer T, Canney P, Ellis P, Cameron D, Blot E, Vermorken J, Coleman R, Marreaud S, Bogaerts J, Basaran G, Piccart M (2008) Anti-tumor activity of capecitabine and vinorelbine in patients with anthracycline- and taxane-pretreated metastatic breast cancer: findings from the EORTC 10001 randomized phase II trial. Breast 17:180–185. doi:10.1016/j.breast.2007.09.002
Ren Q, Kao V, Grem JL (1998) Cytotoxicity and DNA fragmentation associated with sequential gemcitabine and 5-fluoro-2’-deoxyuridine in HT-29 colon cancer cells. Clin Cancer Res 4:2811–2818
Schilsky RL, Bertucci D, Vogelzang NJ, Kindler HL, Ratain MJ (2002) Dose-escalating study of capecitabine plus gemcitabine combination therapy in patients with advanced cancer. J Clin Oncol 20:582–587
Sawada N, Fujimoto-Ouchi K, Ishikawa T, Tanaka Y, Ishitsuka H (2002) Antitumor activity of combination therapy with capecitabine plus vinorelbine, and capecitabine plus gemcitabine in human tumor xenograft models (abstract 5388). Proc Am Assoc Cancer Res 43:1088a
Hess V, Salzberg M, Borner M, Morant R, Roth AD, Ludwig C, Herrmann R (2003) Combining capecitabine and gemcitabine in patients with advanced pancreatic carcinoma: a phase I/II trial. J Clin Oncol 21:66–68
Andres R, Mayordomo JI, Lara R, Lastra R, Ortega E, Polo E, Lambea J, Isla D, Saenz-Cusi A, Escudero P, Tres A (2005) Gemcitabine/capecitabine in patients with metastatic breast cancer pretreated with anthracyclines and taxanes. Clin Breast Cancer 6:158–162
Benekli M, Yildiz R, Uner A, Er O, Yamac D, Alkis N, Coskun U, Camci C, Buyukberber S (2007) Gemcitabine plus capecitabine combination in metastatic breast cancer patients previously treated with anthracyclines and taxanes. Oncology 72:308–313. doi:10.1159/000113053
Giannessi P, Martella F, Di Marsico R, Coltelli L, Safina V, Andreuccetti M, Falcone A (2005) Gemcitabine in combination with capecitabine in metastatic breast carcinoma: a phase I-II study. J Clin Oncol 23:870 (Abstract)
Sikov W, Theall K, Seidler C, Strenger R, Fenton M (2005) Gemcitabine and capecitabine in metastatic breast cancer (MBC): a Brown University Oncology Group (BrUOG) phase II study. ASCO Annu Meet Proc 23:785 (Abstract)
Ahn J, Kim J, Lee J, Kang Y, Kim W (2005) Phase II study of gemcitabine and capecitabine in patients with metastatic breast cancer pretreated with anthracyclines and taxanes. J Clin Oncol 23:895 (Abstract)
Teletaeva G, Erndiev S, Moiseyenko V (2003) Phase II trial of Gemcitabine and Capecitabine combination in patients with metastatic breast cancer resistant to standard therapy. Ann Oncol 14:55 (Abstract)
Pérez-Manga G, Lépez-Criado P, Mendez M (2004) Gemcitabine (G) plus capecitabine (C) in previously treated metastatic breast cancer (MBC) patients. Final results from a phase II GOTI trial. Breast Cancer Res Treat 88:5063 (Abstract)
Malmström A, Hansen J, Malmberg L, Carlsson L, Svensson JH, Ahlgren J, Ahlin C, Jansson T, Westberg R (2010) Gemcitabine and capecitabine in combination for advanced anthracycline and taxane pre-treated breast cancer patients: a phase II study. Acta Oncol 49:35–41. doi:10.3109/02841860903268031
Pallis AG, Boukovinas I, Ardavanis A, Varthalitis I, Malamos N, Georgoulias V, Mavroudis D (2012) A multicenter randomized phase III trial of vinorelbine/gemcitabine doublet versus capecitabine monotherapy in anthracycline- and taxane-pretreated women with metastatic breast cancer. Ann Oncol 23:1164–1169. doi:10.1093/annonc/mdr405
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247. doi:10.1016/j.ejca.2008.10.026
A’hern R (2001) Sample size tables for exact single-stage phase II designs. Stat Med 20:859–866
Paridaens R, Biganzoli L, Bruning P, Klijn JG, Gamucci T, Houston S, Coleman R, Schachter J, Van Vreckem A, Sylvester R, Awada A, Wildiers J, Piccart M (2000) Paclitaxel versus doxorubicin as first-line single-agent chemotherapy for metastatic breast cancer: a European Organization for Research and Treatment of Cancer Randomized Study with cross-over. J Clin Oncol 18:724–733
Jones SE, Erban J, Overmoyer B, Budd GT, Hutchins L, Lower E, Laufman L, Sundaram S, Urba WJ, Pritchard KI, Mennel R, Richards D, Olsen S, Meyers ML, Ravdin PM (2005) Randomized phase III study of docetaxel compared with paclitaxel in metastatic breast cancer. J Clin Oncol 23:5542–5551. doi:10.1200/JCO.2005.02.027
Biganzoli L, Cufer T, Bruning P, Coleman R, Duchateau L, Calvert AH, Gamucci T, Twelves C, Fargeot P, Epelbaum R, Lohrisch C, Piccart MJ (2002) Doxorubicin and paclitaxel versus doxorubicin and cyclophosphamide as first-line chemotherapy in metastatic breast cancer: the European Organization for Research and Treatment of Cancer 10961 Multicenter Phase III Trial. J Clin Oncol 20:3114–3121
Cardoso F, Senkus-Konefka E, Fallowfield L, Costa A, Castiglione M, Group EGW (2010) Locally recurrent or metastatic breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 21(Suppl 5):15–19. doi:10.1093/annonc/mdq160
Ellis LM, Bernstein DS, Voest EE, Berlin JD, Sargent D, Cortazar P, Garrett-Mayer E, Herbst RS, Lilenbaum RC, Sima C, Venook AP, Gonen M, Schilsky RL, Meropol NJ, Schnipper LE (2014) American society of clinical oncology perspective: raising the bar for clinical trials by defining clinically meaningful outcomes. J Clin Oncol 32:1277–1280. doi:10.1200/JCO.2013.53.8009
Ciruelos EM, Cortes J, Cortes-Funes H, Mayordomo JI, Bermejo B, Ojeda B, Garcia E, Rodriguez CA, Munoz M, Gomez P, Manso L, Andres R, Lluch A, Saura C, Mendiola C, Baselga J (2010) Gemcitabine and capecitabine in previously anthracycline-treated metastatic breast cancer: a multicenter phase II study (SOLTI 0301 trial). Ann Oncol 21:1442–1447. doi:10.1093/annonc/mdp536
Sugiyama E, Kaniwa N, Kim SR, Kikura-Hanajiri R, Hasegawa R, Maekawa K, Saito Y, Ozawa S, Sawada J, Kamatani N, Furuse J, Ishii H, Yoshida T, Ueno H, Okusaka T, Saijo N (2007) Pharmacokinetics of gemcitabine in Japanese cancer patients: the impact of a cytidine deaminase polymorphism. J Clin Oncol 25:32–42. doi:10.1200/JCO.2006.06.7405
Phan VH, Moore MM, McLachlan AJ, Piquette-Miller M, Xu H, Clarke SJ (2009) Ethnic differences in drug metabolism and toxicity from chemotherapy. Expert Opin Drug Metab Toxicol 5:243–257. doi:10.1517/17425250902800153
Ma Y, Tang L, Wang HX, Xu YC, Ma Y, Zhang FC (2012) Capecitabine for the treatment for advanced gastric cancer: efficacy, safety and ethnicity. J Clin Pharm Ther 37:266–275. doi:10.1111/j.1365-2710.2011.01289.x
Leonard R, O’Shaughnessy J, Vukelja S, Gorbounova V, Chan-Navarro CA, Maraninchi D, Barak-Wigler N, McKendrick JJ, Harker WG, Bexon AS, Twelves C (2006) Detailed analysis of a randomized phase III trial: can the tolerability of capecitabine plus docetaxel be improved without compromising its survival advantage? Ann Oncol 17:1379–1385. doi:10.1093/annonc/mdl134
Leonard R, Hennessy BT, Blum JL, O’Shaughnessy J (2011) Dose-adjusting capecitabine minimizes adverse effects while maintaining efficacy: a retrospective review of capecitabine for metastatic breast cancer. Clin Breast Cancer 11:349–356. doi:10.1016/j.clbc.2011.06.005
Conflict of interest
No conflict of interest exists in the submission of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, J.S., Jeung, HC., Rha, S.Y. et al. Phase II gemcitabine and capecitabine combination therapy in recurrent or metastatic breast cancer patients pretreated with anthracycline and taxane. Cancer Chemother Pharmacol 74, 799–808 (2014). https://doi.org/10.1007/s00280-014-2551-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-014-2551-4