Abstract
Purpose
The absorption, distribution, metabolism, and excretion of the hedgehog pathway inhibitor sonidegib (LDE225) were determined in healthy male subjects.
Methods
Six subjects received a single oral dose of 800 mg 14C-sonidegib (74 kBq, 2.0 µCi) under fasting conditions. Blood, plasma, urine, and fecal samples were collected predose, postdose in-house (days 1–22), and during 24-h visits (weekly, days 29–43; biweekly, days 57–99). Radioactivity was determined in all samples using accelerator mass spectrometry (AMS). Liquid chromatography–tandem mass spectrometry (LC–MS/MS) was used to determine concentrations of sonidegib and its main circulating metabolite in plasma. Metabolite profiles and structures were determined in pooled plasma, urine, and fecal samples using high-performance LC–AMS and LC–MS/MS, respectively.
Results
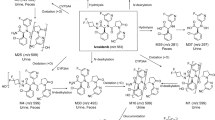
A single dose of 14C-sonidegib was well tolerated in healthy subjects. Unchanged sonidegib and total radioactivity reached peak concentration in plasma by 2 and 3 h, respectively, and demonstrated similarly long half-lives of 319 and 331 h, respectively. Absorbed sonidegib (estimated 6–7 %) was extensively distributed, and the approximate terminal volume of distribution was 2,500 L. Unchanged sonidegib and a metabolite resulting from amide hydrolysis were the major circulating components (36.4 and 15.4 % of radioactivity area under the curve, respectively). Absorbed sonidegib was eliminated predominantly through oxidative metabolism of the morpholine part and amide hydrolysis. Unabsorbed sonidegib was excreted through the feces. Metabolites in excreta accounted for 4.49 % of the dose (1.20 % in urine, 3.29 % in feces). The recovery of radioactivity in urine and feces was essentially complete (95.3 ± 1.93 % of the dose in five subjects; 56.9 % of the dose in one subject with incomplete feces collection suspected).
Conclusions
Sonidegib exhibited low absorption, was extensively distributed, and was slowly metabolized. Elimination of absorbed sonidegib occurred largely by oxidative and hydrolytic metabolism.




Similar content being viewed by others
References
Teglund S, Toftgard R (2010) Hedgehog beyond medulloblastoma and basal cell carcinoma. Biochim Biophys Acta 1805:181–208
Pasca di Magliano M, Hebrok M (2003) Hedgehog signalling in cancer formation and maintenance. Nat Rev Cancer 3:903–911
McMahon AP, Ingham PW, Tabin CJ (2003) Developmental roles and clinical significance of hedgehog signaling. Curr Top Dev Biol 53:1–114
Scales SJ, de Sauvage FJ (2009) Mechanisms of hedgehog pathway activation in cancer and implications for therapy. Trends Pharmacol Sci 30:303–312
Pan S, Wu X, Jiang J, Gao W, Wan Y, Cheng D, Han D, Liu J, Englund NP, Wang Y, Peukert S, Miller-Moslin K, Yuan J, Guo R, Matsumoto M, Vattay A, Jiang Y, Tsao J, Sun F, Pferdekamper AMC, Dodd S, Tuntland T, Maniara W, Kelleher JF III, Yao Y-M, Warmuth M, Williams J, Dorsch M (2010) Discovery of NVP-LDE225, a potent and selective smoothened antagonist. ACS Med Chem Lett 1:130–134
Buonamici S, Williams J, Morrissey M, Wang A, Guo R, Vattay A, Hsiao K, Yuan J, Green J, Ospina B, Yu Q, Ostrom L, Fordjour P, Anderson DL, Monahan JE, Kelleher JF, Peukert S, Pan S, Wu X, Maira SM, Garcia-Echeverria C, Briggs KJ, Watkins DN, Yao YM, Lengauer C, Warmuth M, Sellers WR, Dorsch M (2010) Interfering with resistance to smoothened antagonists by inhibition of the PI3K pathway in medulloblastoma. Sci Transl Med 2:51ra70
Chen JK, Taipale J, Cooper MK, Beachy PA (2002) Inhibition of hedgehog signaling by direct binding of cyclopamine to smoothened. Genes Dev 16:2743–2748
Rodon J, Tawbi HA, Thomas AL, Stoller RG, Turtschi CP, Baselga J, Sarantopoulos J, Mahalingham D, Shou Y, Moles MA, Yang L, Granvil C, Hurh E, Rose KL, Amakye DD, Dummer R, Mita AC (2014) A phase I, multicenter, open-label, first-in-human, dose-escalation study of the oral smoothened inhibitor sonidegib (LDE225) in patients with advanced solid tumors. Clin Cancer Res 20:1900–1909
International Commission on Radiological Protection (ICRP) (2007) The 2007 recommendations of the International Commission on Radiological Protection. ICRP Publication 103. Ann ICRP 37(2–4):116–117
Zoppi U, Crye J, Song Q, Arjomand A (2007) Performance evaluation of the new AMS system at Accium Biosciences. Radiocarbon 49:171–180
Garner RC (2000) Accelerator mass spectrometry in pharmaceutical research and development—a new ultrasensitive analytical method for isotope measurement. Curr Drug Metab 1:205–213
Ognibene TJ, Bench G, Vogel JS, Peaslee GF, Murov S (2003) A high-throughput method for the conversion of CO2 obtained from biochemical samples to graphite in septa-sealed vials for quantification of 14C via accelerator mass spectrometry. Anal Chem 75:2192–2196
Zoppi U, Arjomand A (2010) Simultaneous AMS determination of 14C content and total carbon mass in biological samples. Nucl Instrum Methods Phys Res Sect B 268:1307–1308
Faed EM (1984) Properties of acyl glucuronides: implications for studies of the pharmacokinetics and metabolism of acidic drugs. Drug Metab Rev 15:1213–1249
Shipkova M, Armstrong VW, Oellerich M, Wieland E (2003) Acyl glucuronide drug metabolites: toxicological and analytical implications. Ther Drug Monit 25:1–16
Benet LZ, Spahn-Langguth H, Iwakawa S, Volland C, Mizuma T, Mayer S, Mutschler E, Lin ET (1993) Predictability of the covalent binding of acidic drugs in man. Life Sci 53:PL141–PL146
Bolze S, Bromet N, Gay-Feutry C, Massiere F, Boulieu R, Hulot T (2002) Development of an in vitro screening model for the biosynthesis of acyl glucuronide metabolites and the assessment of their reactivity toward human serum albumin. Drug Metab Dispos 30:404–413
Acknowledgments
We thank Camille Perret, PhD (Drug Metabolism and Pharmacokinetics, Novartis, Basel, Switzerland, currently Hoffmann-La Roche), for her contributions to the study design; Albrecht Glaenzel, PhD (Isotope Laboratory, Novartis, Basel, Switzerland), Rolf Gerber, PhD, and Celine Decaudin (Technical Research and Development, Novartis, Basel, Switzerland) for preparing the radiolabeled drug substance and drug product; Julie Zalikowski and Ali Arjomand, PhD, from Accium BioSciences, Inc. (Seattle, WA, USA), for HPLC fractionations and AMS measurements; Ruud Lutgerink from Pharmaceutical Research Associates, Inc. (PRA, The Netherlands), for managing the clinical part of the study; Arnold Demailly and Xavier Homo (Drug Metabolism and Pharmacokinetics, Novartis, Basel, Switzerland) for supporting the metabolic and bioanalytical part of the study; and Stefan Peukert, PhD (Global Discovery Chemistry, Novartis, Cambridge, MA, USA), Matthias Kittelmann, PhD, Fabian Eggimann, Andreas Fredenhagen, PhD, and Juergen Kuehnoel (Global Discovery Chemistry, Novartis, Basel, Switzerland) for the synthesis of reference compounds. Medical editorial assistance was provided by Jillian Brechbiel, PhD, Karen Kaluza, PhD, and Karen Miller-Moslin, PhD; financial support for editorial assistance was provided by Novartis Pharmaceuticals Corporation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zollinger, M., Lozac’h, F., Hurh, E. et al. Absorption, distribution, metabolism, and excretion (ADME) of 14C-sonidegib (LDE225) in healthy volunteers. Cancer Chemother Pharmacol 74, 63–75 (2014). https://doi.org/10.1007/s00280-014-2468-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-014-2468-y