Abstract
Red blood cell transfusion independence (RBC-TI) is an important goal in treating lower-risk myelodysplastic syndromes with ring sideroblasts. In the phase 3 MEDALIST study, RBC-TI of ≥ 8 weeks was achieved by significantly more luspatercept- versus placebo-treated patients in the first 24 weeks of treatment. In this post hoc analysis, we evaluated RBC transfusion units and visits based on patients’ baseline transfusion burden level and the clinical benefit of luspatercept treatment beyond week 25 in initial luspatercept nonresponders (patients who did not achieve RBC-TI ≥ 8 weeks by week 25) but continued luspatercept up to 144 weeks. RBC transfusion burden, erythroid response, serum ferritin levels, and hemoglobin levels relative to baseline were evaluated. Through week 25, fewer RBC transfusion units and visits were observed in luspatercept-treated patients versus placebo, regardless of baseline transfusion burden. This continued through 144 weeks of luspatercept treatment, particularly in patients with low baseline transfusion burden. Sixty-eight patients were initial nonresponders at week 25 but continued treatment; most (81%) received the maximum dose of luspatercept (1.75 mg/kg). Sixteen percent achieved RBC-TI for ≥ 8 weeks during weeks 25–48, 26% had reduced RBC transfusion burden, 10% achieved an erythroid response, 44% had reduced serum ferritin, and hemoglobin levels increased an average of 1.3 g/dL from baseline. These data have implications for clinical practice, as transfusion units and visits are less in luspatercept-treated patients through week 25 regardless of baseline transfusion burden, and continuing luspatercept beyond week 25 can potentially provide additional clinical benefits for initial nonresponders. Trial registration: NCT02631070.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Myelodysplastic syndromes (MDS) are a heterogenous group of clonal bone marrow disorders characterized by inadequate hematopoiesis; peripheral cytopenias, particularly anemia; and an increased risk of progression to acute myeloid leukemia [1, 2]. The majority of patients with MDS fall into the lower-risk (LR)-MDS category, defined as having a risk of Very low, Low, or Intermediate disease according to the Revised International Prognostic Scoring System (IPSS-R) [3] with a score of ≤ 3.5 points [3,4,5]. Chronic anemia is the predominant cause of morbidity and quality of life impairment in patients with LR-MDS; a major goal of treatment is to manage anemia and its associated complications. Most patients will develop anemia at some point and will become dependent on red blood cell (RBC) transfusions, which carries the risk of iron overload and associated sequelae [6, 7]. Iron overload resulting from RBC transfusions is an independent, adverse prognostic factor for overall survival (OS) and leukemia-free survival in patients with MDS [8], significantly worsening the survival of patients with MDS with refractory anemia [6].
Real-world, observational studies have shown an inverse relationship between the burden of ongoing RBC transfusion and survival in patients with anemia due to LR-MDS, even among those with low initial RBC transfusion burden [9, 10]. Other reports have linked dependency on RBC transfusions to significantly shorter OS and leukemia-free survival [6, 11,12,13,14], more severe disease [15], and reduced health-related quality of life [14, 16] than observed in patients with RBC transfusion independence (RBC-TI). Transfusion dose density has also been shown to be inversely associated with progression-free survival, indicating that even at relatively low dose densities, reliance on transfusions is an indicator of inferior progression-free survival [9].
Eliminating or reducing the need for RBC transfusions could improve the overall health-related quality of life of patients with LR-MDS while also reducing health care resource use and associated costs. However, very few therapeutic options are available to these patients after failure of erythropoiesis-stimulating agents (ESAs). Alternative treatments are a priority, one of which is luspatercept, a first-in-class recombinant fusion protein that binds select transforming growth factor β superfamily ligands thus inhibiting downstream Smad2/3 signaling, which enables late-stage erythroid blast differentiation and erythroid maturation [17,18,19]. In the phase 3 MEDALIST study (NCT02631070), luspatercept treatment resulted in RBC-TI for 8 weeks or longer in 38% of patients treated with luspatercept versus 13% of patients treated with placebo by week 25 (p < 0.001) [20]. Erythroid responses with associated increases in hemoglobin levels were also achieved in a greater proportion of patients treated with luspatercept than placebo (53% vs 12%), demonstrating a reduction in the severity of anemia in these patients [20]. Based on these findings, luspatercept is now approved in the USA, Canada, and Europe to treat anemia resulting from LR-MDS and ring sideroblasts [21,22,23].
Translating the findings from the MEDALIST study to clinical and health policy decision-making requires broader considerations of real-world patients and clinical scenarios. It is important to understand how luspatercept may reduce RBC transfusion burden relative to the patient’s level of transfusion burden before initiation of treatment. In addition, the potential clinical benefits of continuing luspatercept treatment beyond week 25, with the aim of achieving RBC-TI or substantially reducing anemia burden (for those who remained transfusion dependent at week 25), are not well understood and would be relevant to clinicians to help guide treatment decision-making.
In this post hoc analysis of MEDALIST, we evaluated the impact of luspatercept on cumulative RBC transfusion units and RBC transfusion visits over time for treated patients based on their pretreatment transfusion burden levels. Furthermore, we evaluated the clinical benefits of continuing luspatercept treatment beyond week 25 in patients who had not achieved RBC-TI for ≥ 8 weeks during the first 24 weeks of MEDALIST.
Methods
Study design
The design of the MEDALIST study has been described previously [20]. Briefly, eligible patients were ≥ 18 years of age; had LR-MDS according to IPSS-R criteria with ring sideroblasts; were refractory, intolerant, or unlikely to respond to ESAs; and had required regular RBC transfusions (≥ 2 units/8 weeks) in the 16 weeks prior to randomization.
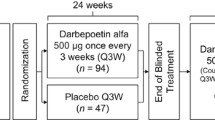
Patients were randomized 2:1 to receive either luspatercept 1.0 mg/kg subcutaneously or placebo every 3 weeks (Fig. 1). Patients could receive luspatercept dose adjustments during the trial (up to a maximum of 1.75 mg/kg) [20]. The primary efficacy endpoint of MEDALIST was the achievement of RBC-TI for ≥ 8 weeks during the primary treatment phase (weeks 1–24). This was evaluated at week 25, defined as 24 calendar weeks after the first dose, regardless of any dose delays.
MEDALIST study design. aInvestigator’s assessment of clinical benefit: achievement of RBC-TI ≥ 8 weeks and/or HI-E response during weeks 1–24. AML, acute myeloid leukemia; EPO, erythropoietin; ESA, erythropoiesis-stimulating agent; HI-E, hematologic improvement-erythroid; HMA, hypomethylating agent; IMiD, immunomodulatory imide drug; IPSS-R, Revised International Prognostic Scoring System; IWG, International Working Group; MDS, myeloid dysplastic syndromes; RBC, red blood cell; RBC-TI, red blood cell transfusion independence; RS, ring sideroblasts; SC, subcutaneous; SF3B1, splicing factor 3B subunit 1; WHO, World Health Organization
Regardless of meeting the primary endpoint at week 25, patients who had shown clinical benefit (as assessed by the investigators) without disease progression (according to International Working Group (IWG)-MDS criteria) [24] could continue to receive luspatercept or placebo in a double-blind treatment extension phase until no clinical benefit, disease progression, unacceptable side effects, or withdrawal from the study. The extension phase continued with 3-week treatment cycles.
Assessments
This post hoc analysis of MEDALIST evaluated the cumulative mean number of RBC transfusion units and RBC transfusion visits through 144 weeks of treatment according to baseline RBC transfusion burden level (low vs high). Low and high baseline RBC transfusion burden levels were defined as having < 6 RBC transfusion units or ≥ 6 RBC transfusion units over 8 weeks, respectively. The cumulative mean number of RBC transfusion units and RBC transfusion visits required during the treatment period according to baseline RBC transfusion burden level was also evaluated by luspatercept response at week 25. Patients in the luspatercept arm who achieved RBC-TI for ≥ 8 weeks by week 25 were defined as luspatercept responders; those who did not were defined as initial luspatercept nonresponders. The cumulative mean number of RBC transfusion units or RBC transfusion visits required was estimated using Nelson–Aalen nonparametric estimators with robust variance estimate for each treatment group with a 95% confidence interval.
This post hoc analysis also evaluated the effect of continuing luspatercept treatment beyond the first 24 weeks. Assessment of clinical benefit was conducted up to the data cutoff date of July 1, 2019 (144 weeks of treatment). Clinical benefit indicators included RBC transfusion burden, hemoglobin and serum ferritin levels, and hematologic improvement-erythroid (HI-E) response. HI-E response was assessed per modified IWG 2006 criteria [24] and was defined as a reduction in RBC transfusions of ≥ 4 units/8 weeks for patients with baseline RBC transfusion burden of ≥ 4 units/8 weeks, and an increase in hemoglobin level of ≥ 1.5 g/dL over 8 weeks in the absence of RBC transfusions for patients with baseline RBC transfusion burden of < 4 units/8 weeks.
Results
Patient population
A total of 229 patients were enrolled in MEDALIST; of these, 153 were randomized to receive luspatercept and 76 to receive placebo. The baseline characteristics of the patients were well balanced across treatment arms and have been described previously [20]. The number of patients receiving pre- and post-week 25 luspatercept dose levels, following the initial 1.0 mg/kg starting dose, is provided in Table 1. A greater proportion of patients received the maximum dose of luspatercept (1.75 mg/kg; 55%) in the post-week 25 period than in the first 24 weeks (41%). During weeks 25–48, 81% of initial luspatercept nonresponders were treated with the maximum dose of luspatercept (1.75 mg/kg).
Of the patients treated with luspatercept, 87 (57%) were classified as having low baseline RBC transfusion burden and 66 (43%) as having high baseline RBC transfusion burden and were evaluated for cumulative RBC units and visits over time. In the placebo arm, the proportions of patients with low or high RBC transfusion burden were identical, with 57% (43/76) and 43% (33/76) having low and high baseline RBC transfusion burden, respectively. In the initial 24 weeks of treatment, 58 patients who received luspatercept achieved RBC-TI for ≥ 8 weeks. Of the 95 patients who did not achieve an initial response and were classified as initial luspatercept nonresponders, 68 (72%) continued luspatercept treatment for up to 144 weeks.
Effect of treatment on number of RBC transfusion units and visits
Overall, during the first 24 weeks of treatment, patients receiving luspatercept had lower mean cumulative RBC transfusion units and RBC transfusion visits than patients treated with placebo, regardless of baseline RBC transfusion burden status (Table 2). This was largely driven by luspatercept responders who had the lowest mean cumulative RBC transfusion units and RBC transfusion visits regardless of baseline RBC transfusion burden category. Expected cumulative number of RBC transfusion units and RBC transfusion visits by baseline transfusion burden is shown in Fig. 2. Beyond week 25, patients with a low baseline RBC transfusion burden who were treated with luspatercept continued to have a lower cumulative number of RBC transfusion units (Fig. 2A) and RBC transfusion visits (Fig. 2C) compared with patients treated with placebo.
Expected cumulative number of RBC transfusion units (A, B) and RBC transfusion visits (C, D) by baseline transfusion burden (low vs high). Low baseline RBC transfusion burden was defined as having < 6 RBC transfusion units over 8 weeks and high baseline RBC transfusion burden was defined as having ≥ 6 RBC transfusion units over 8 weeks. Cumulative mean function estimated using nonparametric method with robust variance estimate. The efficacy cutoff date is defined as the minimum date among death date, study discontinuation date, last dose date + 20, and database cut date. RBC, red blood cell
Effect of treatment on RBC transfusion units and visits by luspatercept response at week 25
When categorized by luspatercept response at week 25, patients treated with luspatercept who had responded to treatment by week 25 (achieved RBC-TI for ≥ 8 weeks) continued to show fewer mean cumulative RBC transfusion units required relative to initial luspatercept nonresponders regardless of baseline RBC transfusion burden level (Fig. 3A, B, Table 3). The median follow-up of luspatercept responders was 26 months. Similarly, luspatercept responders at week 25 continued to show a reduction in mean cumulative RBC transfusion visits relative to initial luspatercept nonresponders regardless of baseline RBC transfusion burden level (Fig. 3C, D, Table 3).
Expected cumulative number of RBC transfusion units (A, B) and RBC transfusion visits (C, D) by baseline RBC transfusion burden (low vs high) by luspatercept response at week 25. Low baseline RBC transfusion burden was defined as having < 6 RBC transfusion units over 8 weeks and high baseline RBC transfusion burden was defined as having ≥ 6 RBC transfusion units over 8 weeks. Cumulative mean function estimated using nonparametric method with robust variance estimate. The efficacy cutoff date is defined as the minimum date among death date, study discontinuation date, last dose date + 20, and database cut date. RBC, red blood cell
Clinical outcomes in patients who did not achieve RBC-TI at ≥ 8 weeks but continued luspatercept treatment beyond week 25
Weeks 25–48 of the extension phase
Of the 68 patients who were initial luspatercept nonresponders (by HI-E) at week 25 and continued treatment, 11 patients (16%) achieved RBC-TI for ≥ 8 weeks during weeks 25–48 of the extension phase. Of these, 3 patients were RBC-TI for ≥ 16 weeks (Online Resource 1 Figure). The median time to achieving RBC-TI for ≥ 8 weeks was 5 months from the beginning of week 25 (i.e., approximately 11 months from the first dose in the primary treatment phase, weeks 1–24).
During weeks 25–48, 18 (26%) of the 68 initial luspatercept nonresponders had a reduced RBC transfusion burden relative to baseline. In this group, the mean change (standard deviation (SD)) from baseline in the RBC units transfused was − 3.1 units (6.51) during the first 24 weeks of treatment, rising to − 6.5 units (6.31) during weeks 25–48. In initial luspatercept nonresponders with a full 48 weeks of available treatment data (n = 36), the mean change (SD) from baseline in RBC units transfused was − 1.3 units (7.98) over a 24-week treatment period. Serum ferritin levels relative to baseline were reduced during weeks 25–48 in 30 (44%) of the 68 initial luspatercept nonresponders. By week 48, the serum ferritin level in patients who had a baseline serum ferritin level of ≥ 1000 µg/L had dropped to < 1000 µg/L in 7 (18%) of 39 patients.
As a descriptive analysis, the mean change from baseline in hemoglobin levels from weeks 25 to 48 for initial luspatercept nonresponders is shown in Fig. 4 (for patients who had been assessed for hemoglobin at the time point). The greatest mean increase from baseline was 1.3 g/dL, which occurred at extension phase cycle 3 (beyond week 25) (Fig. 4).
RBC transfusion events for the 68 initial luspatercept nonresponders during the 16 weeks prior to and 48 weeks after the start of luspatercept treatment are shown in Fig. 5. This includes the 11 (16%) patients who continued luspatercept beyond week 25 and ultimately achieved RBC-TI for ≥ 8 weeks despite being classified as initial luspatercept nonresponders at week 25.
Weeks 1–48 and during the entire treatment period
Clinical benefit as assessed with the response indicators RBC-TI for ≥ 8 weeks, ≥ 50% reduction in transfusion burden, and HI-E response is shown in Fig. 6 across analysis periods during treatment. During weeks 1–48, of the 68 patients who were initial luspatercept nonresponders at week 25, 41 (60%) had achieved a ≥ 50% reduction in RBC transfusion burden for ≥ 8 weeks from baseline and 32 (47%) had achieved an HI-E response. Beyond 48 weeks and through 144 weeks, 4 additional patients achieved RBC-TI for ≥ 8 weeks and 1 additional patient achieved an HI-E response.
Discussion
In the MEDALIST study, luspatercept demonstrated a consistent benefit in reducing RBC transfusion burden within the first 24 weeks of treatment among patients with LR-MDS [20]. In this analysis, patients who had responded by week 25 of MEDALIST continued to show a benefit of luspatercept treatment in terms of lower cumulative RBC transfusion units and RBC transfusion visits, regardless of baseline RBC transfusion burden levels, over a median follow-up of 26 months. A considerable proportion of the 68 patients who did not achieve RBC-TI for ≥ 8 weeks at week 25 and continued on luspatercept experienced clinical benefit during the entire treatment period. Clinical benefits, observed through 144 weeks in initial luspatercept nonresponders, included RBC-TI for ≥ 8 weeks in 22% of patients, ≥ 50% reduction in RBC transfusion burden in 60%, an HI-E response in 49%, improved hemoglobin levels of up to a mean gain of 1.3 g/dL from baseline, and reduced serum ferritin compared with baseline in 44%. For most (81%) initial luspatercept nonresponders, luspatercept was titrated to the maximum dose level (1.75 mg/kg) during weeks 25–48 for these clinical benefits to be achieved.
The findings of this analysis have value in the real-world clinical setting where luspatercept can lower transfusion burden and continue to offer clinical benefits beyond 25 weeks of treatment. This analysis has revealed a lower cumulative transfusion burden in terms of the number of RBC transfusion units and visits with continued luspatercept treatment and has shown that transfusion burden among initial luspatercept nonresponders can be improved by extending treatment. Continued luspatercept treatment beyond 25 weeks may provide RBC-TI and hemoglobin improvement for patients who require additional time to achieve a response indicative of clinical benefit.
Achieving transfusion independence in LR-MDS is an important treatment goal given the negative impact of RBC transfusions on OS and leukemia-free survival in patients with LR-MDS [6, 11,12,13,14], the humanistic burden of chronic anemia in patients [14, 16], and the risk of iron overload [8]. Our findings have shown that extending treatment with luspatercept beyond 25 weeks may be of benefit for patients with LR-MDS who were initially less responsive.
According to the package insert, treatment with luspatercept should be discontinued if a patient does not experience a decrease in transfusion burden after 9 weeks of treatment at the maximum dose level or in cases of unacceptable toxicity [21, 22]. In the MEDALIST study, decisions regarding continuation of treatment in patients not meeting stringent parameters indicating response were made by individual clinicians based on their assessment of clinical benefit in terms of reductions in RBC transfusion burden compared with baseline; increased hemoglobin and decreased serum ferritin levels compared with baseline; and HI-E response. In the real-world setting, clinicians’ assessment of improvement in clinical features and, critically, how their patients perceive the balance between burden of treatment and benefit, in terms of reduced anemia and transfusion burden, should guide decisions regarding continuation of treatment. In line with prescribing guidelines, most of those patients going on to achieve benefit after week 25 in the MEDALIST study did so on the highest dose of luspatercept, making maximal titration, when tolerated, a key consideration before discontinuation.
A strength of this analysis is that the patient-level data were obtained from a large multinational phase 3 randomized, placebo-controlled trial. Limitations include its nature as a post hoc analysis focused on a subgroup of patients not attaining a response, resulting in the loss of statistical inference attainable from a randomized population. Therefore, these results should be confirmed in future prospective analyses [25]. In addition, the substantial drop-out in the placebo group beyond week 25 may have minimized the robust comparison between groups during the longer follow-up period.
In conclusion, our findings suggest that continuing luspatercept treatment beyond 25 weeks may provide clinical benefit for a meaningful proportion of patients with LR-MDS with ring sideroblasts who otherwise have limited treatment options. These findings may inform clinical practice treatment decisions with regard to the timing of clinical benefit, the relevance of increasing the dose level of luspatercept in the absence of an initial response, and the consideration of luspatercept treatment for patients with high or low pretreatment RBC transfusion burden to facilitate a rational identification of the patients most likely to benefit.
Data availability
The BMS policy on data sharing may be found at https://www.bms.com/researchers-and-partners/independent-research/data-sharing-request-process.html.
References
Adès L, Itzykson R, Fenaux P (2014) Myelodysplastic syndromes. Lancet 383:2239–2252. https://doi.org/10.1016/S0140-6736(13)61901-7
Platzbecker U, Kubasch AS, Homer-Bouthiette C, Prebet T (2021) Current challenges and unmet medical needs in myelodysplastic syndromes. Leukemia 35:2182–2198. https://doi.org/10.1038/s41375-021-01265-7
Greenberg PL, Tuechler H, Schanz J, Sanz G, Garcia-Manero G, Solé F, Bennett JM, Bowen D, Fenaux P, Dreyfus F, Kantarjian H, Kuendgen A, Levis A, Malcovati L, Cazzola M, Cermak J, Fonatsch C, Le Beau MM, Slovak ML, Krieger O, Luebbert M, Maciejewski J, Magalhaes SM, Miyazaki Y, Pfeilstöcker M, Sekeres M, Sperr WR, Stauder R, Tauro S, Valent P, Vallespi T, van de Loosdrecht AA, Germing U, Haase D (2012) Revised international prognostic scoring system for myelodysplastic syndromes. Blood 120:2454–2465. https://doi.org/10.1182/blood-2012-03-420489
Pfeilstöcker M, Tuechler H, Sanz G, Schanz J, Garcia-Manero G, Solé F, Bennett JM, Bowen D, Fenaux P, Dreyfus F, Kantarjian H, Kuendgen A, Malcovati L, Cazzola M, Cermak J, Fonatsch C, Le Beau MM, Slovak ML, Levis A, Luebbert M, Maciejewski J, Machherndl-Spandl S, Magalhaes SM, Miyazaki Y, Sekeres MA, Sperr WR, Stauder R, Tauro S, Valent P, Vallespi T, van de Loosdrecht AA, Germing U, Haase D, Greenberg PL (2016) Time-dependent changes in mortality and transformation risk in MDS. Blood 128:902–910. https://doi.org/10.1182/blood-2016-02-700054
Neukirchen J, Lauseker M, Blum S, Giagounidis A, Lübbert M, Martino S, Siragusa S, Schlenk RF, Platzbecker U, Hofmann WK, Götze K, Palumbo GA, Magrin S, Kündgen A, Aul C, Hildebrandt B, Hasford J, Kobbe G, Haas R, Germing U (2014) Validation of the revised international prognostic scoring system (IPSS-R) in patients with myelodysplastic syndrome: a multicenter study. Leuk Res 38:57–64. https://doi.org/10.1016/j.leukres.2013.10.013
Malcovati L, Della Porta MG, Pascutto C, Invernizzi R, Boni M, Travaglino E, Passamonti F, Arcaini L, Maffioli M, Bernasconi P, Lazzarino M, Cazzola M (2005) Prognostic factors and life expectancy in myelodysplastic syndromes classified according to WHO criteria: a basis for clinical decision making. J Clin Oncol 23:7594–7603. https://doi.org/10.1200/JCO.2005.01.7038
Wood JC (2015) Estimating tissue iron burden: current status and future prospects. Br J Haematol 170:15–28. https://doi.org/10.1111/bjh.13374
Jabbour E, Kantarjian HM, Koller C, Taher A (2008) Red blood cell transfusions and iron overload in the treatment of patients with myelodysplastic syndromes. Cancer 112:1089–1095. https://doi.org/10.1002/cncr.23280
de Swart L, Crouch S, Hoeks M, Smith A, Langemeijer S, Fenaux P, Symeonidis A, Cermâk J, Hellström-Lindberg E, Stauder R, Sanz G, Mittelman M, Holm MS, Malcovati L, Mądry K, Germing U, Tatic A, Savic A, Almeida AM, Gredelj-Simec N, Guerci-Bresler A, Beyne-Rauzy O, Culligan D, Kotsianidis I, Itzykson R, van Marrewijk C, Blijlevens N, Bowen D, de Witte T, Registry Participants EUMDS (2020) Impact of red blood cell transfusion dose density on progression-free survival in patients with lower-risk myelodysplastic syndromes. Haematologica 105:632–639. https://doi.org/10.3324/haematol.2018.212217
Sangerman MA, Pomares H, Alonso E, Grau J, Galiano M, Encuentra M, Sureda A (2019) Impact of red blood cell transfusion burden status in patients with lower-risk myelodysplastic syndromes. Blood 134(Supplement_1):3013. https://doi.org/10.1182/blood-2019-127486
Hiwase DK, Kutyna MM, Chhetri R, Howell S, Harrison PB, Nath SV, Nath LS, Wickham N, Szabo F, Hui C-H, Ross D, Gray J, Melo JV, Bardy P, To LB (2013) Transfusion dependency is associated with inferior survival even in very low and low risk IPSS-R patients. Blood 122:1518. https://doi.org/10.1182/blood.V122.21.1518.1518
Hiwase DK, Singhal D, Strupp C, Chhetri R, Kutyna MM, Wee LA, Harrison PB, Nath SV, Wickham N, Hui CH, Gray JX, Bardy P, Ross DM, Lewis ID, Reynolds J, To LB, Germing U (2017) Dynamic assessment of RBC-transfusion dependency improves the prognostic value of the revised-IPSS in MDS patients. Am J Hematol 92:508–514. https://doi.org/10.1002/ajh.24704
Harnan S, Ren S, Gomersall T, Everson-Hock ES, Sutton A, Dhanasiri S, Kulasekararaj A (2016) Association between transfusion status and overall survival in patients with myelodysplastic syndromes: a systematic literature review and meta-analysis. Acta Haematol 136:23–42. https://doi.org/10.1159/000445163
Lemos MB, Rodrigues SR, Schroeder T, Kulasekararaj AG, Matos JE, Tang D (2021) Association between red blood cell transfusion dependence and burden in patients with myelodysplastic syndromes: a systematic literature review and meta-analysis. Eur J Haematol 107:3–23. https://doi.org/10.1111/ejh.13619
Malcovati L, Della Porta MG, Cazzola M (2006) Predicting survival and leukemic evolution in patients with myelodysplastic syndrome. Haematologica 91:1588–1590. https://doi.org/10.3324/%25x
Crawford J, Cella D, Cleeland CS, Cremieux PY, Demetri GD, Sarokhan BJ, Slavin MB, Glaspy JA (2002) Relationship between changes in hemoglobin level and quality of life during chemotherapy in anemic cancer patients receiving epoetin alfa therapy. Cancer 95:888–895. https://doi.org/10.1002/cncr.10763
Suragani RN, Cadena SM, Cawley SM, Sako D, Mitchell D, Li R, Davies MV, Alexander MJ, Devine M, Loveday KS, Underwood KW, Grinberg AV, Quisel JD, Chopra R, Pearsall RS, Seehra J, Kumar R (2014) Transforming growth factor-β superfamily ligand trap ACE-536 corrects anemia by promoting late-stage erythropoiesis. Nat Med 20:408–414. https://doi.org/10.1038/nm.3512
Fenaux P, Kiladjian JJ, Platzbecker U (2019) Luspatercept for the treatment of anemia in myelodysplastic syndromes and primary myelofibrosis. Blood 133:790–794. https://doi.org/10.1182/blood-2018-11-876888
Zermati Y, Fichelson S, Valensi F, Freyssinier JM, Rouyer-Fessard P, Cramer E, Guichard J, Varet B, Hermine O (2000) Transforming growth factor inhibits erythropoiesis by blocking proliferation and accelerating differentiation of erythroid progenitors. Exp Hematol 28:885–894. https://doi.org/10.1016/s0301-472x(00)00488-4
Fenaux P, Platzbecker U, Mufti GJ, Garcia-Manero G, Buckstein R, Santini V, Díez-Campelo M, Finelli C, Cazzola M, Ilhan O, Sekeres MA, Falantes JF, Arrizabalaga B, Salvi F, Giai V, Vyas P, Bowen D, Selleslag D, DeZern AE, Jurcic JG, Germing U, Götze KS, Quesnel B, Beyne-Rauzy O, Cluzeau T, Voso MT, Mazure D, Vellenga E, Greenberg PL, Hellström-Lindberg E, Zeidan AM, Adès L, Verma A, Savona MR, Laadem A, Benzohra A, Zhang J, Rampersad A, Dunshee DR, Linde PG, Sherman ML, Komrokji RS, List AF (2020) Luspatercept in patients with lower-risk myelodysplastic syndromes. N Engl J Med 382:140–151. https://doi.org/10.1056/NEJMoa1908892
US Food and Drug Administration (FDA). REBLOZYL® prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761136orig2lbl.pdf. Accessed 7 Mar 2022
European Medicines Agency (EMA). REBLOZYL® prescribing information. https://www.ema.europa.eu/en/documents/product-information/reblozyl-epar-product-information_en.pdf. Accessed 7 Mar 2022
Health Canada. REBLOZYL® Canada Product Monograph. https://pdf.hres.ca/dpd_pm/00059994.PDF. Accessed 7 Mar 2022
Cheson BD, Greenberg PL, Bennett JM, Lowenberg B, Wijermans PW, Nimer SD, Pinto A, Beran M, de Witte TM, Stone RM, Mittelman M, Sanz GF, Gore SD, Schiffer CA, Kantarjian H (2006) Clinical application and proposal for modification of the International Working Group (IWG) response criteria in myelodysplasia. Blood 108:419–425. https://doi.org/10.1182/blood-2005-10-4149
Curran-Everett D, Milgrom H (2013) Post-hoc data analysis: benefits and limitations. Curr Opin Allergy Clin Immunol 13:223–224. https://doi.org/10.1097/ACI.0b013e3283609831
Acknowledgements
The authors received writing and editorial assistance in the preparation of this manuscript from Saba Choudhary, PhD, of Excerpta Medica, funded by Bristol Myers Squibb. The authors are fully responsible for all content and editorial decisions for this manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was supported by Celgene, a Bristol Myers Squibb Company, in collaboration with Acceleron Pharma Inc., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA.
Author information
Authors and Affiliations
Contributions
U.G., A.Y., D.T.: conception/design, data interpretation; P.F., U.P., R.B., V.S., M.D-C., D.M., C.H., R.S.K., G. G-M.: data interpretation; S.F.: data acquisition, data interpretation; G.Z., R.Z., X.H.: data analysis; A.M.Z.: data acquisition.
Corresponding author
Ethics declarations
Ethics approval
All participating sites received Institutional Review Board or ethics committees approval for the protocol. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5).
Consent to participate
All patients in the MEDALIST study provided written informed consent.
Conflict of interest
UG has received research support from BMS and Novartis; speaker honorarium from BMS, Jazz Pharmaceuticals, and Novartis; and consulting fees from BMS. PF has received research support and honoraria from BMS. UP has received research funding from Amgen, BMS, Janssen, Merck, and Novartis; consulting fees from BMS; honoraria from AbbVie, BMS, Geron, Janssen, Novartis, and Takeda. RB has received research funding and honoraria for advisory boards from BMS and Taiho Pharma. VS has served on advisory boards for BMS, Geron, Gilead, Menarini, Novartis, and Takeda; and has served on speaker’s bureau for BMS and Novartis. MD-C reports honoraria, consulting fees, and travel support, and has served on advisory boards for BMS, Novartis, and Takeda. AY, DT, GZ, DM, and CH are employees of BMS, and hold stock ownership. SF and XH are employees of BMS. RZ is an employee of GSK and former employee of BMS. RSK reports consulting fees from AbbVie, Acceleron Pharma, BMS, Geron, Jazz Pharmaceuticals, and Novartis; and speakers bureau or honoraria for AbbVie, BMS, CTI BioPharma, Innovent, Jazz Pharmaceuticals, Novartis, PharmaEssentia, Taiho Oncology, and Takeda. AMZ reports consultancy fees from AbbVie, Acceleron, Agios, Amgen, Aprea, Astellas, BeyondSpring, Boehringer-Ingelheim, Cardiff Oncology, Cardinal Health, Celgene/BMS, Daiichi Sankyo, Epizyme, Geron, Gilead, Incyte, Ionis, Janssen, Jazz, Kura, Novartis, Otsuka, Pfizer, Seattle Genetics, Syndax, Taiho, Takeda, Trovagene, and Tyme; research funding for AbbVie, ADC Therapeutics, Amgen, Aprea, Astex, Boehringer-Ingelheim, Cardiff Oncology, BMS, Incyte, Medimmune/Astrazeneca, Novartis, Otsuka, Pfizer, Takeda, and Trovagene; honoraria for AbbVie, Acceleron, Agios, Amgen, Aprea, Astellas, Beyondpspring, Boehringer-Ingelheim, Cardiff Oncology, Cardinal Health, Celgene/BMS, Daiichi Sankyo, Epizyme, Geron, Gilead, Incyte, Ionis, Janssen, Jazz, Kura, Novartis, Otsuka, Pfizer, Seattle Genetics, Syndax, Taiho, Takeda, Trovagene, and Tyme; clinical trial committees for Abbvie, BMS, Geron, Gilead, Kura, and Novartis; and travel support for Cardiff Oncology, Novartis, and Pfizer. GG-M reports grant or research support from AbbVie, Amphivena, Aprea, BMS, Curis, Forty Seven, H3 Biomedicine, Helsinn, Merck, Novartis, and Onconova; and consultancy fees from Astex, BMS, Genentech, and Helsinn.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Germing, U., Fenaux, P., Platzbecker, U. et al. Improved benefit of continuing luspatercept therapy: sub-analysis of patients with lower-risk MDS in the MEDALIST study. Ann Hematol 102, 311–321 (2023). https://doi.org/10.1007/s00277-022-05071-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-022-05071-8