Abstract
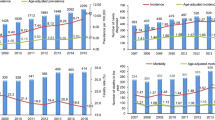
Current understanding of the epidemiology and outcomes for patients with multiple myeloma in Finland is scarce due to lack of comprehensive real-world evidence in clinical practice. The aim of this study was to gain understanding of epidemiological characteristics and treatment and survival outcomes by utilizing multiple real-world data sources with information of adults treated for active multiple myeloma (MM) during years 2005–2016 in Finland. A total of 3851 adult MM patients with C90.0 diagnosis fulfilling all inclusion criteria were included in the analysis. The average myeloma incidence was six cases per 100,000, which slightly increased (p = 0.011) during the follow-up. The age-standardized incidence was three cases per 100,000 in the years 2005–2016. On average, 25% of patients received autologous stem cell transplantation (ASCT), and this proportion increased during the years 2005–2015 from 17 to 30%. The majority of patients under 65 years of age received ASCT treatment (60.5%), whereas only 8.7% of patients 65 years of age or older were treated with ASCT. The net median overall survival improved by approximately 5 months from 2005–2010 (3.44 years) to 2011–2016 (3.89 years); after adjusting for covariates, this presented an annual 4% reduction in the risk of death. Longer median survival and decreased risk of death indicate improved treatment outcomes from 2005 to 2016 among adult MM patients in Finland.





Similar content being viewed by others
References
Cancer statistics [Internet]. Syöpärekisteri. [cited 2020 Jun 5]. Available from: https://cancerregistry.fi/statistics/cancer-statistics/
https://cancerregistry.fi/statistics/cancer-statistics/. - Google-haku [Internet]. [cited 2020 Jun 5].
JRC Data Catalogue - European Commission [Internet]. [cited 2020 Jun 5]. Available from: https://data.jrc.ec.europa.eu/collection/id-0095.
Monimuotoinen myelooma [Internet]. [cited 2020 Jun 5]. Available from: https://www.duodecimlehti.fi/duo14977.
Barwick BG, Gupta VA, Vertino PM, Boise LH (2019) Cell of origin and genetic alterations in the pathogenesis of multiple myeloma. Front Immunol 10:1121
Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, Kumar S, Hillengass J, Kastritis E, Richardson P, Landgren O, Paiva B, Dispenzieri A, Weiss B, LeLeu X, Zweegman S, Lonial S, Rosinol L, Zamagni E, Jagannath S, Sezer O, Kristinsson SY, Caers J, Usmani SZ, Lahuerta JJ, Johnsen HE, Beksac M, Cavo M, Goldschmidt H, Terpos E, Kyle RA, Anderson KC, Durie BGM, Miguel JFS (2014) International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 15:e538–e548
Röllig C, Knop S, Bornhäuser M (2015) Multiple myeloma. Lancet 385:2197–2208
Moreau P, San Miguel J, Ludwig H et al (2013) Multiple myeloma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 24:vi133–vi137
Moreau P, San Miguel J, Sonneveld P et al (2017) Multiple myeloma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 28:iv52–iv61
Kansalliset hoitosuositukset (FMG) | SHY [Internet]. [cited 2020 Jun 5]. Available from: https://www.hematology.fi/fi/hoito-ohjeet/veritaudit/plasmasolutaudit/myelooma/hoito/kansalliset-hoitosuositukset-fmg.
Petrakis I, Kontogiorgis C, Nena E, Athanasakis K, Gougoula V, Kotsianidis I, Constantinidis TC (2019) Unraveling innovation potential in the real-world setting: eighteen novel agents with twenty-six approved European indications, in the management of leukemias, lymphomas, and multiple myeloma. Expert Rev Hematol 12:1063–1075
Jagannath S, Roy A, Kish J, Lunacsek O, Globe D, Eaddy M, Kuriakose ET, Willey J, Butler-Bird S, Siegel D (2016) Real-world treatment patterns and associated progression-free survival in relapsed/refractory multiple myeloma among US community oncology practices. Expert Rev Hematol 9:707–717
Song X, Cong Z, Wilson K (2016) Real-world treatment patterns, comorbidities, and disease-related complications in patients with multiple myeloma in the United States. Curr Med Res Opin 32:95–103
Remes K, Anttila P, Silvennoinen R, et al. Real-world treatment outcomes in multiple myeloma: multicenter registry results from Finland 2009-2013. Bertolini F, editor. PLoS One. 2018;13:e0208507.
Thygesen SK, Christiansen CF, Christensen S, Lash TL, Sørensen HT (2011) The predictive value of ICD-10 diagnostic coding used to assess Charlson comorbidity index conditions in the population-based Danish National Registry of Patients. BMC Med Res Methodol 11:83
Blimark CH, Turesson I, Genell A, Ahlberg L, Björkstrand B, Carlson K, Forsberg K, Juliusson G, Linder O, Mellqvist UH, Nahi H, Kristinsson SY, for the Swedish Myeloma Registry (2018) Outcome and survival of myeloma patients diagnosed 2008–2015. Real-world data on 4904 patients from the Swedish Myeloma Registry. Haematologica. 103:506–513
Acknowledgements
We would like to acknowledge writing assistance from Kai Kysenius and Sari Vanhatupa (current employee and former employee at Medaffcon).
Funding
This study was funded by Takeda Oy.
Author information
Authors and Affiliations
Contributions
IT, TM, MIL, MB, PA contributed to the study design and objectives, interpretation of results, and revising the manuscript. IT was further responsible for data analysis and MIL was responsible for manuscript development. JL critically reviewed the results and contributed to interpretation as well as revision of the manuscript. All authors have reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by each register holder.
Conflict of interest
IT, MIL, and TM are employees of Medaffcon Oy; TM is and MB was an employee of Takeda Finland; JL has received consultation fees from Amgen, Celgene, Bristol-Myers Squibb, Sanofi, Takeda, Janssen, and travel grants from Janssen, Celgene, Amgen, Novartis, TEVA, Takeda, Roche, Abbvie, Pfizer; PA has been the Hematologist in charge for treatment of plasma cell malignancies at the Helsinki University Hospital and has received consultation fees from Amgen, BMS, Celgene, GSK, Janssen, Novartis, Sanofi and Takeda and travel grants from AbbVie, Amgen, BMS, Celgene, Sanofi Genzyme, Janssen, Mundipharma, Novartis, Roche and Teva.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Toppila, I., Miettinen, T., Lassenius, M.I. et al. Characteristics and survival trends in Finnish multiple myeloma patients—a nationwide real-world evidence study. Ann Hematol 100, 1779–1787 (2021). https://doi.org/10.1007/s00277-021-04481-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-021-04481-4