Abstract
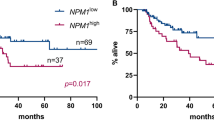
For acute myeloid leukemia (AML) with nucleophosmin 1 mutation (NPM1m), multiparameter flow cytometry (FCM) and real-time quantitative polymerase chain reaction (RQ-PCR) are used to monitor minimal residual disease (MRD). However, the results of the two methods are sometimes inconsistent. This study was designed to analyze how to address the discordant results of FCM and RQ-PCR in AML patients undergoing chemotherapy, especially when positive FCM (FCM+) and negative NPM1m (NPM1m−) results are detected in the same sample. Our study included 93 AML patients with NPM1m positive (NPM1m+) who received chemotherapy but did not undergo hematopoietic stem cell transplantation. We monitored NPM1m and leukemia-associated immunophenotypes (LAIPs) by RQ-PCR and FCM, respectively, to assess MRD after each chemotherapy course. After each course of chemotherapy, all patients were classified into four groups based on the results of FCM and RQ-PCR: both negative (group 1, FCM−NPM1m−), single positive (group 2, FCM−NPM1m+; group 3, FCM+NPM1m−), or both positive (group 4, FCM+NPM1m+). The results showed that there was not a significant difference in the 2-year cumulative incidence of relapse (CIR) after each course of chemotherapy between group 2 and group 3. Furthermore, patients in groups 2 and 3 had a lower 2-year CIR than those in group 4 and a significantly higher 2-year CIR than those in group 1 after the first two courses. The patients in group 4 had a significantly higher 2-year CIR than those in group 1 after the first two courses. These results suggested that in the MRD monitoring process of AML patients, when the results of FCM and RQ-PCR are inconsistent (especially when FCM is positive and NPM1m is negative), these single-positive results still have predictive significance for relapse.




Similar content being viewed by others
References
Freeman SD, Virgo P, Couzens S, Grimwade D, Russell N, Hills RK, Burnett AK (2013) Prognostic relevance of treatment response measured by flow cytometric residual disease detection in older patients with acute myeloid leukemia. J Clin Oncol 31(32):4123–4131
Walter RB, Othus M, Borthakur G, Ravandi F, Cortes JE, Pierce SA, Appelbaum FR, Kantarjian HA, Estey EH (2011) Prediction of early death after induction therapy for newly diagnosed acute myeloid leukemia with pretreatment risk scores: a novel paradigm for treatment assignment. J Clin Oncol 29(33):4417–4423
Haferlach C, Mecucci C, Schnittger S, Kohlmann A, Mancini M, Cuneo A, Testoni N, Rege-Cambrin G, Santucci A, Vignetti M, Fazi P, Martelli MP, Haferlach T, Falini B (2009) AML with mutated NPM1 carrying a normal or aberrant karyotype show overlapping biologic, pathologic, immunophenotypic, and prognostic features. Blood 114(14):3024–3032
O'Donnell MR, Tallman MS, Abboud CN, Altman JK, Appelbaum FR, Arber DA, Attar E, Borate U, Coutre SE, Damon LE, Lancet J, Maness LJ, Marcucci G, Martin MG, Millenson MM, Moore JO, Ravandi F, Shami PJ, Smith BD, Stone RM, Strickland SA, Wang ES, Gregory KM, Naganuma M (2013) Acute myeloid leukemia, version 2.2013. J Natl Compr Cancer Netw 11(9):1047–1055
Patel SS, Kuo FC, Gibson CJ, Steensma DP, Soiffer RJ, Alyea EP 3rd, Chen YA, Fathi AT, Graubert TA, Brunner AM, Wadleigh M, Stone RM, DeAngelo DJ, Nardi V, Hasserjian RP, Weinberg OK (2018) High NPM1-mutant allele burden at diagnosis predicts unfavorable outcomes in de novo AML. Blood 131(25):2816–2825
Buccisano F, Maurillo L, Del Principe MI, Del Poeta G, Sconocchia G, Lo-Coco F, Arcese W, Amadori S, Venditti A (2012) Prognostic and therapeutic implications of minimal residual disease detection in acute myeloid leukemia. Blood 119(2):332–341
Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, Wheatley K, Harrison CJ, Burnett AK (2010) Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood 116(3):354–365
Byrd JC, Mrozek K, Dodge RK, Carroll AJ, Edwards CG, Arthur DC, Pettenati MJ, Patil SR, Rao KW, Watson MS, Koduru PR, Moore JO, Stone RM, Mayer RJ, Feldman EJ, Davey FR, Schiffer CA, Larson RA, Bloomfield CD (2002) Pretreatment cytogenetic abnormalities are predictive of induction success, cumulative incidence of relapse, and overall survival in adult patients with de novo acute myeloid leukemia: results from Cancer and Leukemia Group B (CALGB 8461). Blood 100(13):4325–4336
Chou WC, Tang JL, Wu SJ, Tsay W, Yao M, Huang SY, Huang KC, Chen CY, Huang CF, Tien HF (2007) Clinical implications of minimal residual disease monitoring by quantitative polymerase chain reaction in acute myeloid leukemia patients bearing nucleophosmin (NPM1) mutations. Leukemia 21(5):998–1004
Grimwade D, Vyas P, Freeman S (2010) Assessment of minimal residual disease in acute myeloid leukemia. Curr Opin Oncol 22(6):656–663
Hokland P, Ommen HB (2011) Towards individualized follow-up in adult acute myeloid leukemia in remission. Blood 117(9):2577–2584
Kern W, Haferlach C, Haferlach T, Schnittger S (2008) Monitoring of minimal residual disease in acute myeloid leukemia. Cancer 112(1):4–16
Falini B, Mecucci C, Tiacci E, Alcalay M, Rosati R, Pasqualucci L, La Starza R, Diverio D, Colombo E, Santucci A, Bigerna B, Pacini R, Pucciarini A, Liso A, Vignetti M, Fazi P, Meani N, Pettirossi V, Saglio G, Mandelli F, Lo-Coco F, Pelicci PG, Martelli MF (2005) Cytoplasmic nucleophosmin in acute myelogenous leukemia with a normal karyotype. N Engl J Med 352(3):254–266
Boissel N, Renneville A, Biggio V, Philippe N, Thomas X, Cayuela JM, Terre C, Tigaud I, Castaigne S, Raffoux E, De Botton S, Fenaux P, Dombret H, Preudhomme C (2005) Prevalence, clinical profile, and prognosis of NPM mutations in AML with normal karyotype. Blood 106(10):3618–3620
Schnittger S, Schoch C, Kern W, Mecucci C, Tschulik C, Martelli MF, Haferlach T, Hiddemann W, Falini B (2005) Nucleophosmin gene mutations are predictors of favorable prognosis in acute myelogenous leukemia with a normal karyotype. Blood 106(12):3733–3739
Patkar N, Kodgule R, Kakirde C, Raval G, Bhanshe P, Joshi S, Chaudhary S, Badrinath Y, Ghoghale S, Kadechkar S, Khizer SH, Kannan S, Shetty D, Gokarn A, Punatkar S, Jain H, Bagal B, Menon H, Sengar M, Khattry N, Tembhare P, Subramanian P, Gujral S (2018) Clinical impact of measurable residual disease monitoring by ultradeep next generation sequencing in NPM1 mutated acute myeloid leukemia. Oncotarget 9(93):36613–36624
Mariotti B, Meconi F, Palmieri R, De Bellis E, Lavorgna S, Ottone T, Martini V, Lo-Coco F, Cicconi L (2019) Acute myeloid leukemia with concomitant BCR-ABL and NPM1 mutations. Case Rep Hematol 2019:6707506
Ruan GR, Li JL, Qin YZ, Li LD, Xie M, Chang Y, Zhang Y, Liu YR, Jiang B, Chen SS, Huang XJ (2009) Nucleophosmin mutations in Chinese adults with acute myelogenous leukemia. Ann Hematol 88(2):159–166
Falini B, Martelli MP, Bolli N, Sportoletti P, Liso A, Tiacci E, Haferlach T (2011) Acute myeloid leukemia with mutated nucleophosmin (NPM1): is it a distinct entity? Blood 117(4):1109–1120
Gorello P, Cazzaniga G, Alberti F, Dell'Oro MG, Gottardi E, Specchia G, Roti G, Rosati R, Martelli MF, Diverio D, Lo Coco F, Biondi A, Saglio G, Mecucci C, Falini B (2006) Quantitative assessment of minimal residual disease in acute myeloid leukemia carrying nucleophosmin (NPM1) gene mutations. Leukemia 20(6):1103–1108
Papadaki C, Dufour A, Seibl M, Schneider S, Bohlander SK, Zellmeier E, Mellert G, Hiddemann W, Spiekermann K (2009) Monitoring minimal residual disease in acute myeloid leukaemia with NPM1 mutations by quantitative PCR: clonal evolution is a limiting factor. Br J Haematol 144(4):517–523
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, Bloomfield CD, Cazzola M, Vardiman JW (2016) The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 127(20):2391–2405
Zhao XS, Jin S, Zhu HH, Xu LP, Liu DH, Chen H, Liu KY, Huang XJ (2012) Wilms' tumor gene 1 expression: an independent acute leukemia prognostic indicator following allogeneic hematopoietic SCT. Bone Marrow Transplant 47(4):499–507
Chang YJ, Wang Y, Liu YR, Xu LP, Zhang XH, Chen H, Chen YH, Wang FR, Han W, Sun YQ, Yan CH, Tang FF, Mo XD, Liu KY, Huang XJ (2017) Haploidentical allograft is superior to matched sibling donor allograft in eradicating pre-transplantation minimal residual disease of AML patients as determined by multiparameter flow cytometry: a retrospective and prospective analysis. J Hematol Oncol 10(1):134
Hong Y, Zhao X, Qin Y, Zhou S, Chang Y, Wang Y, Zhang X, Xu L, Huang X (2018) The prognostic role of E2A-PBX1 expression detected by real-time quantitative reverse transcriptase polymerase chain reaction (RQ-PCR) in B cell acute lymphoblastic leukemia after allogeneic hematopoietic stem cell transplantation. Ann Hematol 97(9):1547–1554
Zhao X, Hong Y, Qin Y, Xu Y, Chang Y, Wang Y, Zhang X, Xu L, Huang X (2017) The clinical significance of monitoring the expression of the SIL-TAL1 fusion gene in T-cell acute lymphoblastic leukemia after allogeneic hematopoietic stem cell transplantation. Int J Lab Hematol 39(6):613–619
Zhao XS, Liu YR, Zhu HH, Xu LP, Liu DH, Liu KY, Huang XJ (2012) Monitoring MRD with flow cytometry: an effective method to predict relapse for ALL patients after allogeneic hematopoietic stem cell transplantation. Ann Hematol 91(2):183–192
Ivey A, Hills RK, Simpson MA, Jovanovic JV, Gilkes A, Grech A, Patel Y, Bhudia N, Farah H, Mason J, Wall K, Akiki S, Griffiths M, Solomon E, McCaughan F, Linch DC, Gale RE, Vyas P, Freeman SD, Russell N, Burnett AK, Grimwade D (2016) Assessment of minimal residual disease in standard-risk AML. N Engl J Med 374(5):422–433
Kronke J, Schlenk RF, Jensen KO, Tschurtz F, Corbacioglu A, Gaidzik VI, Paschka P, Onken S, Eiwen K, Habdank M, Spath D, Lubbert M, Wattad M, Kindler T, Salih HR, Held G, Nachbaur D, von Lilienfeld-Toal M, Germing U, Haase D, Mergenthaler HG, Krauter J, Ganser A, Gohring G, Schlegelberger B, Dohner H, Dohner K (2011) Monitoring of minimal residual disease in NPM1-mutated acute myeloid leukemia: a study from the German-Austrian acute myeloid leukemia study group. J Clin Oncol 29(19):2709–2716
Bill M, Grimm J, Jentzsch M, Kloss L, Goldmann K, Schulz J, Beinicke S, Hantschel J, Cross M, Vucinic V, Ponisch W, Behre G, Franke GN, Lange T, Niederwieser D, Schwind S (2018) Digital droplet PCR-based absolute quantification of pre-transplant NPM1 mutation burden predicts relapse in acute myeloid leukemia patients. Ann Hematol 97(10):1757–1765
Forghieri F, Comoli P, Marasca R, Potenza L, Luppi M (2018) Minimal/measurable residual disease monitoring in NPM1-mutated acute myeloid leukemia: a clinical viewpoint and perspectives. Int J Mol Sci 19(11):E3492
Zhao XS, Yan CH, Liu DH, Xu LP, Liu YR, Liu KY, Qin YZ, Wang Y, Huang XJ (2013) Combined use of WT1 and flow cytometry monitoring can promote sensitivity of predicting relapse after allogeneic HSCT without affecting specificity. Ann Hematol 92(8):1111–1119
Acknowledgments
The authors thank American Journal Experts (www.journalexperts.com) for their assistance in editing this manuscript. The work was supported by the National Key Research and Development Program of China (2017YFA0104500), the National Natural Science Foundation of China (grant no. 81670175, grant no. 81870137), and the Innovative Research Groups of the National Natural Science Foundation of China (grant no. 81621001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict(s) of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Gao, MG., Ruan, GR., Chang, YJ. et al. The predictive value of minimal residual disease when facing the inconsistent results detected by real-time quantitative PCR and flow cytometry in NPM1-mutated acute myeloid leukemia. Ann Hematol 99, 73–82 (2020). https://doi.org/10.1007/s00277-019-03861-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-019-03861-1