Abstract
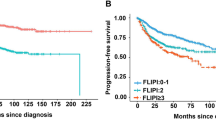
Follicular lymphoma (FL) is a low-grade lymphoma that is usually characterized by generalized lymphadenopathy. Extranodal invasion by FL generally involves the bone marrow, skin, and duodenum; splenic infiltration often occurs in the advanced stages. However, primary splenic FL is very rare. Hence, few studies have been performed on splenic FL, and its clinicopathological features have not been established. This study aimed to investigate the clinicopathological features of primary splenic FL, as compared to nodal FL. We analyzed 17 patients diagnosed with primary splenic FL and 153 control patients with systemic FL. Hepatitis C virus (HCV)-positive status was significantly more common in patients with splenic FL than in the control patients (p = 0.02). Ann Arbor stage III or IV (p = 0.0003) and high-risk FLIPI (Follicular Lymphoma International Prognostic Index) (p = 0.03) were significantly less common in patients with splenic FL than in the control patients; however, the overall and progression-free survival curves were not significantly different between the groups. Among the 17 patients with splenic FL, the progression-free survival was significantly worse in patients who underwent splenectomy without receiving postoperative chemotherapy than in those who did (p = 0.03). These results suggest that primary splenic FL should be considered different from systemic FL; accordingly, its management should also be conducted differently.



Similar content being viewed by others
References
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW (2009) World Health Organization classification of tumours: pathology and genetics of tumours of haematopoietic and lymphoid tissues, 4th edn. IARC Press, Lyon
Nakamura S, Ichimura K, Sato Y, Nakamura S, Nakamine H, Inagaki H, Sadahira Y, Ohshima K, Sakugawa S, Kondo E, Yanai H, Ohara N, Yoshino T (2006) Follicular lymphoma frequently originates in the salivary gland. Pathol Int 56:576–583
Bacon CM, Ye H, Diss TC, McNamara C, Kueck B, Hasserjian RP, Rohatiner AZ, Ferry J, Du MQ, Dogan A (2007) Primary follicular lymphoma of testis and epididymis in adults. Am J Surg Pathol 31:1050–1058
Grange F, Bekkenk MW, Wechsler J, Meijer CJ, Cerroni L, Bernengo M, Bosq J, Hedelin G, Fink Puches R, van Vloten WA, Joly P, Bagot M, Willemze R (2001) Prognostic factors in primary cutaneous large B-cell lymphomas: a European multicenter study. J Clin Oncol 19:3602–3610
Yoshino T, Miyake K, Ichimura K, Mannami T, Ohara N, Hamazaki S, Akagi T (2000) Increased incidence of follicular lymphoma in the duodenum. Am J Surg Pathol 24:688–693
Bende RJ, Smit LA, van Noesel CJ (2007) Molecular pathways in follicular lymphoma. Leukemia 21:18–29
Kimura Y, Sato K, Arakawa F, Karube K, Nomura Y, Shimizu K, Aoki R, Hashikawa K, Yoshida S, Kiyasu J, Takeuchi M, Nino D, Sugita Y, Morito T, Yoshino T, Nakamura S, Kikuchi M, Ohshima K (2010) Mantle cell lymphoma shows three morphological evolutions of classical, intermediate, and aggressive forms, which occur in parallel with increased labeling index of cyclin D1 and Ki-67. Cancer Sci 101:806–814
Iannito E, Tripodo C (2011) How I diagnose and treat splenic lymphomas. Blood 117:2585–2595
Shimizu-Kohno K, Kimura Y, Kiyasu J, Miyoshi H, Yoshida M, Ichikawa R, Niino D, Ohshima K (2012) Malignant lymphoma of the spleen in Japan: a clinicopathological analysis of 115 cases. Pathol Int 62:577–582
Howard MT, Dufresne S, Swerdlow SH, Cook JR (2009) Follicular lymphoma of the spleen: multiparameter analysis of 16 cases. Am J Clin Pathol 131:656–662
Mollejo M, Rodríguez-Pinilla MS, Montes-Moreno S, Algara P, Dogan A, Cigudosa JC, Juarez R, Flores T, Forteza J, Arribas A, Piris MA (2009) Splenic follicular lymphoma: clinicopathologic characteristics of a series of 32 cases. Am J Surg Pathol 33:730–738
Skarin AT, Davey FR, Moloney WC (1971) Lymphosarcoma of the spleen. Results of diagnostic splenectomy in 11 patients. Arch Intern Med 127:259–265
Kehoe J, Straus DJ (1988) Primary lymphoma of the spleen. Clinical features and outcome after splenectomy. Cancer 62:1433–1438
Miyoshi H, Sato K, Yoshida M, Kimura Y, Kiyasu J, Ichikawa A, Ishibashi Y, Arakawa F, Nakamura Y, Nakashima S, Niino D, Sugita Y, Ohshima K (2014) CD5-positive follicular lymphoma characterized by CD25, MUM1, low frequency of t(14;18) and poor prognosis. Pathol Int 64:95–103
Yoshida M, Ichikawa A, Miyoshi H, Takeuchi M, Kimura Y, Nino D, Ohshima K (2012) High frequency of t(14;18) in Hodgkin’s lymphoma associated with follicular lymphoma. Pathol Int 62:518–524
Karube K, Martínez D, Royo C, Navarro A, Pinyol M, Cazorla M, Castillo P, Valera A, Carrió A, Costa D, Colomer D, Rosenwald A, Ott G, Esteban D, Giné E, López-Guillermo A, Campo E (2014) Recurrent mutations of NOTCH genes in follicular lymphoma identify a distinctive subset of tumors. J Pathol 234:423–430
Yoshida N, Miyoshi H, Kato T, Sakata-Yanagimoto M, Niino D, Taniguchi H, Moriuchi Y, Miyahara M, Kurita D, Sasaki Y, Shimono J, Kawamoto K, Utsunomiya A, Imaizumi Y, Seto M, Ohshima K (2016) CCR4 frameshift mutation identifies a distinct group of adult T cell leukemia/lymphoma with poor prognosis. J Pathol 238:621–626
Kalpadakis C, Pangalis GA, Vassilakopoulous TP, Sachanas S, Angelopoulou MK (2014) Treatment of splenic marginal zone lymphoma: should splenectomy be abandoned? Leuk Lymphoma 55:1463–1470
Xing KH, Kahlon A, Skinnider BF, Connors JM, Gascoyne RD, Sehn LH, Savage KJ, Slack GW, Shenkier TN, Klasa R, Gerrie AS, Villa D (2015) Outcomes in splenic marginal zone lymphoma: analysis of 107 patients treated in British Columbia. Br J Haematol 169:520–527
Troussard X, Valensi F, Duchayne E, Garand R, Felman P, Tulliez M, Henry-Amar M, Bryon PA, Flandrin G (1996) Splenic lymphoma with villous lymphocytes: clinical presentation, biology and prognostic factors in a series of 100 patients. Groups Francais d’Hématologie Cellulaire (GFHC). Br J Haematol 93:731–736
Lenglet J, Traullé C, Mounier N, Benet C, Munoz-Bongrand N, Amorin S, Noguera ME, Traverse-Glehen A, Ffrench M, Baseggio L, Felman P, Callet-Bauchu E, Brice P, Berger F, Salles G, Brière J, Coiffier B, Thieblemont C (2014) Long-term follow-up analysis of 100 patients with splenic marginal zone lymphoma treated with splenectomy as first-line treatment. Leuk Lymphoma 55:1854–1860
Rossi D, Trifonov V, Fangazio M, Bruscaggin A, Rasi S, Spina V, Monti S, Vaisitti T, Arruga F, Famà R, Ciardullo C, Greco M, Cresta S, Piranda D, Holmes A, Fabbri G, Messina M, Rinaldi A, Wang J, Agostinelli C, Piccaluga PP, Lucioni M, Tabbò F, Serra R, Franceschetti S, Deambrogi C, Daniele G, Gattei V, Marasca R, Facchetti F, Arcaini L, Inghirami G, Bertoni F, Pileri SA, Deaglio S, Foà R, Dalla-Favera R, Pasqualucci L, Rabadan R, Gaidano G (2012) The coding genome of splenic marginal zone lymphoma: activation of NOTCH2 and other pathways regulating marginal zone development. J Exp Med 209:1537–1551
De Renzo A, Perna F, Persico M, Notaro R, Mainolfi C, de Sio I, Ciancia G, Picardi M, Del Vecchio L, Pane F, Rotoli B (2008) Excellent prognosis and prevalence of HCV infection of primary hepatic and splenic non-Hodgkin’s lymphoma. Eur J Haematol 81:51–57
Takeshita M, Sakai H, Okamura S, Oshiro Y, Higaki K, Nakashima O, Uike N, Yamamoto I, Kinjo M, Matsubara F (2005) Splenic large B-cell lymphoma in patients with hepatitis C virus infection. Hum Pathol 36:878–885
Michot JM, Canioni D, Driss H, Alric L, Cacoub P, Suarez F, Sibon D, Thieblemont C, Dupuis J, Terrier B, Feray C, Tilly H, Pol S, Leblond V, Settegrana C, Rabiega P, Barthe Y, Hendel-Chavez H, Nguyen-Khac F, Merle-Béral H, Berger F, Molina T, Charlotte F, Carrat F, Davi F, Hermine O, Besson C, ANRS HC-13Lympho-C Study Group (2015) Antiviral therapy is associated with a better survival in patients with hepatitis C virus and B-cell non Hodgkin lymphomas, ANRS HC-13 lympho-C study. Am J Hematol 90:197–203
Arcaini L, Besson C, Frigeni M, Fontaine H, Goldaniga M, Casato M, Visentini M, Torres HA, Loustaud-Ratti V, Peveling-Oberhag J, Fabris P, Rossotti R, Zaja F, Rigacci L, Rattotti S, Bruno R, Merli M, Dorival C, Alric L, Jaccard A, Pol S, Carrat F, Ferretti VV, Visco C, Hermine O (2016) Interferon-free antiviral treatment in B-cell lymphoproliferative disorders associated with hepatitis C virus infection. Blood 128:2527–2532
Zuckerman E, Zuckerman T, Sahar D, Streichman S, Attias D, Sabo E, Yeshurun D, Rowe JM (2001) The effect of antiviral therapy on t(14;18) translocation and immunoglobulin gene rearrangement in patients with chronic hepatitis C virus infection. Blood 97:1555–1559
Chan CH, Hadlock KG, Foung SK, Levy S (2001) V(H)1-69 gene is preferentially used by hepatitis C virus-associated B cell lymphomas and by normal B cells responding to the E2 viral antigen. Blood 97:1023–1026
Zuckerman E, Zuckerman T, Sahar D, Streichman S, Attias D, Sabo E, Yeshurun D, Rowe J (2001) bcl-2 and immunoglobulin gene rearrangement in patients with hepatitis C virus infection. Br J Haematol 112:364–369
Giannelli F, Moscarella S, Giannini C, Caini P, Monti M, Gragnani L, Romanelli RG, Solazzo V, Laffi G, La Villa G, Gentilini P, Zignego AL (2003) Effect of antiviral treatment in patients with chronic HCV infection and t(14;18) translocation. Blood 102:1196–1201
Poetsch M, Weber-Matthiesen K, Plendl HJ, Grote W, Schlegelberger B (1996) Detection of the t(14;18) chromosomal translocation by interphase cytogenetics with yeast-artificial-chromosome probes in follicular lymphoma and nonneoplastic lymphoproliferation. J Clin Oncol 14:963–969
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Supplementary Fig. 1
Differences in overall survival (a) and progression-free survival (b) between patients with splenic follicular lymphoma (FL) (n = 17) and nodal FL (n = 153). The overall survival and progression-free survival curves did not significantly differ between patients with splenic vs. nodal FL (p = 0.69 and 0.24, respectively). (GIF 69 kb)
Supplementary Fig. 2
Overall survival (a) and progression-free survival (b) in the 17 patients with splenic follicular lymphoma (FL) according to the diagnosis of Type A (n = 12) or Type B (n = 5) splenic FL. The overall survival and progression-free survival curves did not significantly differ between patients with Type A vs. B splenic FL (p = 0.34 and 0.43, respectively). (GIF 52 kb)
Supplementary Table 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Shimono, J., Miyoshi, H., Kamimura, T. et al. Clinicopathological features of primary splenic follicular lymphoma. Ann Hematol 96, 2063–2070 (2017). https://doi.org/10.1007/s00277-017-3139-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-017-3139-y