Abstract
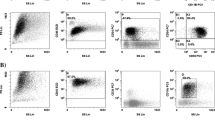
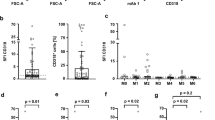
Potential clinical significance of CD34 expression in acute promyelocitic leukemia (APL) has not been deeply investigated. We hereby analyzed the clinico-biological features and treatment outcome of APL patients in relation to CD34 expression, even when expressed in a small subpopulation: 114 APL patients homogeneously treated with the AIDA schedule were included in the study and prognostic correlation with respect to CD34 expression, both when expressed in association with CD2 and as isolated expression (cutoff ≥2 to <10 % or ≥10 %), were investigated. CD34 was associated to CD2 in 30 patients and was isolated in 19 patients. When compared to the CD34-negative population, CD34/CD2 expression identified a subgroup with characteristic features: M3 variant subtype (26 vs 7 % in the negative group, p = 0.02), bcr3 transcript subtype (73 vs 32 %, p = 0.001), high risk according to the risk of relapse (66 vs 17 %, p = 0.002), high incidence of differentiation syndrome (26 vs 12 %, p = 0.01), lower overall survival (88 vs 95 %), and a significantly higher rate of relapse (22 vs 13.8 %, p = 0.05). We then evaluated the prognostic value of isolated CD34 expression: it was detected in nine patients with a cutoff of expression ≥10 % and in 10 patients with a cutoff ≥2 but <10 %. Isolated CD34 positivity identified a subgroup with a classic morphology (79 %), bcr1 prevalence (53 %), higher rate of relapse (37 vs 13.8 % in the negative group, p = 0.002), higher incidence of differentiation syndrome (55 vs 12 %, p = 0.03), and lower overall survival (60 vs 95 %, p = 0.001). The results of our study confirm that CD34/CD2 expression characterizes a subset of APL with a high WBC count and a variant morphological subtype, associated with an unfavorable clinical course. We also show that the isolated expression of CD34, even at a low cutoff, identifies a group of classic APL with a negative prognosis. Further studies aimed at identifying other molecular signatures in CD34-positive patients are needed in order to optimize the therapeutic strategy for this subset of patients.
Similar content being viewed by others
References
Sanz MA, Jarque I, Martin G (1988) Acute promyelocytic leukaemia therapy: results and prognostic factors. Cancer 1:7–13
Tallman MS, Kwaan HC (1992) Reassessing the hemostatic disorder associated with acute promyelocytic leukaemia. Blood 79:543–53
Dombret H, Scrobohaci ML, Ghorra P, Zini JM, Daniel MT, Castaigne S, Degos L (1993) Coagulation disorders associated with acute promyelocytic leukemia: corrective effect of all-trans retinoic acid treatment. Leukemia 7:2–9
Tallman MS, Lefebvre P, Baine RM, Shoji M, Cohen I, Green D et al (2004) Effects of all-trans retinoic acid or chemotherapy on the molecular regulation of systemic blood coagulation and fibrinolysis in patients with acute promyelocytic leukaemia. J Thromb Haemostasis 2:1341–1350
Sanz MA, Lo Coco F (2011) Modern approaches to treating acute promyelocytic leukemia. J Clin Oncol 29:495–503
Paietta E (2003) Expression of cell-surface antigens in acute promyelocytic leukaemia. Best Pract Res Clin Haematol 16:369–385
Guglielmi C, Martelli MP, Diverio D, Fenu S, Vegna ML, Cantù-Rajnoldi A et al (1998) Immunophenotype of adult and childhood acute promyelocytic leukaemia: correlation with morphology, type of PML gene breakpoint and clinical outcome. A cooperative Italian study on 196 cases. Br J Haematol 102:1035–1041
Foley R, Soamboonsrup P, Carter RF, Benger A, Meyer R, Walker I et al (2001) CD34-positive acute promyelocytic leukaemia is associated to leukocytosis, microgranular/hypogranular morphology, expression of CD2 and bcr3 isoform. Am J Hematol 67:34–41
Albano F, Mestice A, Pannunzio A, Lanza F, Martino B, Pastore D et al (2006) The biological characteristics of CD34 + CD2+ adult acute promyelocytic leukemia and the CD34-CD2- hypergranular and microgranular phenotypes. Haematologica 91:311–316
Avvisati G, Lo Coco F, Paoloni FP, Petti MC, Diverio D, Vignetti M et al (2011) AIDA 0493 protocol for newly diagnosed acute promyelocytic leukemia: very long-term results and role of maintenance. Blood 117:4716–4725
Lo Coco F, Avvisati G, Vignetti M, Breccia M, Gallo E, Rambaldi A et al (2010) Front-line treatment of acute promyelocytic leukemia with AIDA induction followed by risk-adapted consolidation for adults younger than 61 years: results of the AIDA 2000 trial of the GIMEMA group. Blood 116:3171–3179
Diverio D, Riccioni R, Pistilli A, Buffolino S, Avvisati G, Mandelli F, Lo Coco F (1996) Improved rapid detection of the PML/RARalpha fusion gene in acute promyelocytic leukemia. Leukemia 10:1214–1216
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Annal Biochem 162:156–159
van Dongen JJ, Macintyre EA, Gabert JA, Delabesse E, Rossi V, Saglio G et al (1999) Standardized RT-PCR analysis of fusion gene transcripts from chromosome aberrations in acute leukemia for detection of minimal residual disease. Report of the BIOMED-1 Concerted Action: investigation of minimal residual disease in acute leukemia. Leukemia 13:1901–1928
Noguera NI, Breccia M, Divona M, Diverio D, Costa V, De Santis S et al (2002) Alterations of the FLT3 gene in acute promyelocytic leukemia: association with diagnostic characteristics and analysis of clinical outcome in patients treated with the Italian AIDA protocol. Leukemia 16:2185–2189
Sanz MA, Lo Coco F, Martin G, Avvisati G, Rayon C, Barbui T et al (2000) Definition of relapse risk and role of nonanthracycline drugs for consolidation in patients with acute promyelocytic leukemia: a joint study of the PETHEMA and GIMEMA cooperative groups. Blood 96:1247–53
Frankel SR (1992) (1992). The “retinoic acid syndrome” in acute promyelocytic leukemia. Ann Intern Med 117:292–296
Paietta E, Goloubeva O, Neuberg D, Bennett JM, Gallagher R, Racevskis J et al (2004) A surrogate marker profile for PML/RAR alpha expressing acute promyelocytic leukemia and the association of immunophenotypic markers with morphologic and molecular subtypes. Cytometry B Clin Cytom 59:1–9
Grimwade D, Outram SV, Flora R, Ings SJ, Pizzey AR, Morilla R et al (2002) The T-lineage affiliated CD2 gene lies within an open chromatin environment in acute promyelocytic leukaemia cells. Cancer Res 62:4730–4735
Claxton DF, Reading CL, Nagarajan L, Tsujimoto Y, Andersson BS, Estey E et al (1992) Correlation of CD2 expression with PML gene breakpoints in patients with acute promyelocytic leukaemia. Blood 80:582–586
Lin P, Hao S, Medeiros LJ, Estey EH, Pierce SA, Wang X et al (2004) Expression of CD2 in acute promyelocytic leukaemia correlates with short form of PML-RARa transcripts and poorer prognosis. Am J Clin Pathol 121:402–407
Gilliland DG, Griffin JD (2002) The roles of FLT3 in hematopoiesis and leukaemia. Blood 100:132–1542
Chillon MC, Santamaria C, Garcia-Sanz R, Balanzategui A, Sarasquete ME, Alcoceba M et al (2010) Long FLT3 internal tandem duplications and reduced PML-RARα expression at diagnosis characterize a high-risk subgroup of acute promyelocytic leukemia patients. Haematologica 95:745–751
Schnittger S, Bacher U, Haferlach C, Kern W, Alpermann T, Haferlach T (2011) Clinical impact of FLT3 mutation load in acute promyelocytic leukemia with t(15;17)/PML-RARA. Haematologica 96:1799–1807
Gale RE, Hills R, Pizzey AR, Kottaridis PD, Swirsky D, Gilkes AF, Adult Leukemia Working Party NCRI et al (2005) Relationship between FLT3 mutation status, biologic characteristics, and response to targeted therapy in acute promyelocytic leukemia. Blood 106:3768–3776
Marasca R, Maffei R, Zucchini P, Castelli I, Saviola A, Martinelli S et al (2006) Gene expression profiling of acute promyelocytic leukaemia identifies two subtypes mainly associated with FLT3 mutational status. Leukemia 20:103–114
Conflict of interest
The authors declare that they have no conflict of interest. Informed consent was obtained from all patients for being included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Breccia, M., De Propris, M.S., Stefanizzi, C. et al. Negative prognostic value of CD34 antigen also if expressed on a small population of acute promyelocitic leukemia cells. Ann Hematol 93, 1819–1823 (2014). https://doi.org/10.1007/s00277-014-2130-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-014-2130-0