Abstract
Purpose
Tumors involving the caudate lobe present a unique therapeutic challenge due to their complex anatomy and the safety and efficacy of locoregional therapy can be variable. The purpose of this study is to analyze the outcomes of radiation segmentectomy for primary caudate lobe tumors.
Materials and Methods
Eight patients [5 women and 3 men; median age = 69 y (range 50–79)] that underwent transarterial radioembolization (TARE) using yttrium-90 (Y90) glass microspheres for primary caudate lobe tumors (hepatocellular carcinoma = 6, intrahepatic cholangiocarcinoma = 2) from August 2017 to March 2021 were retrospectively analyzed. Descriptive statistics, treatment parameters, tumor response (using modified response evaluation criteria in solid tumors), adverse events [using common terminology criteria for adverse events (CTCAE)], and survival outcomes were evaluated.
Results
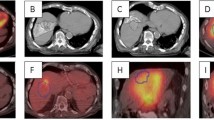
Eight primary caudate lobe tumors with a median size of 2.2 cm [interquartile range (IQR), 1.7–3.3] and Child–Pugh class A liver function underwent TARE of the caudate lobe. The median radiation dose was 596 Gy (IQR 356–1585), median total activity was 0.84 GBq (IQR 0.56–1.31), median specific activity was 473 Bq/sphere (IQR 226–671), and the median number of Y90 microspheres used was 1.4 million (IQR 1.2–3.4). All complications were CTCAE grade 1, and no clinically significant hilar plate complications were observed. In targeted tumors, complete response was seen in all patients. At a median follow-up period of 16.6 months (IQR 6.6–21.6) 75% (6/8) of patients were alive with no in-field progression.
Conclusion
Radiation segmentectomy of primary caudate lobe tumors appears effective and is well tolerated in this limited case series within the described treatment parameters.
Level of Evidence
Level 4, Case Series.



Similar content being viewed by others
References
Takayama T, Makuuchi M. Segmental liver resections, present and future-caudate lobe resection for liver tumors. Hepatogastroenterology. 1998;45:20–3.
Takayasu K, Muramatsu Y, Shima Y, et al. Clinical and radiologic features of hepatocellular carcinoma originating in the caudate lobe. Cancer. 1986;58:1557–62.
Hawkins WG, DeMatteo RP, Cohen MS, et al. Caudate hepatectomy for cancer: a single institution experience with 150 patients. J Am Coll Surg. 2005;200:345–52.
Philips P, Farmer RW, Scoggins CR, et al. Caudate lobe resections: a single-center experience and evaluation of factors predictive of outcomes. World J Surg Oncol. 2013;11:1–6.
Chaib E, Ribeiro MAF Jr, eSilva FdSC, et al. Surgical approach for hepatic caudate lobectomy: review of 401 cases. J Am Coll Surg. 2007;204:118–27.
Liu P, Yang J-M, Niu W-Y, et al. Prognostic factors in the surgical treatment of caudate lobe hepatocellular carcinoma. World J Gastroenterol WJG. 2010;16:1123.
Tanaka S, Shimada M, Shirabe K, et al. Surgical outcome of patients with hepatocellular carcinoma originating in the caudate lobe. Am J Surg. 2005;190:451–5.
Biederman DM, Titano JJ, Bishay VL, et al. Radiation segmentectomy versus TACE combined with microwave ablation for unresectable solitary hepatocellular carcinoma up to 3 cm: a propensity score matching study. Radiology. 2017;283:895–905.
Ibrahim SM, Kulik L, Baker T, et al. Treating and downstaging hepatocellular carcinoma in the caudate lobe with yttrium-90 radioembolization. Cardiovasc Intervent Radiol. 2012;35:1094–101.
Lewandowski RJ, Gabr A, Abouchaleh N, et al. Radiation segmentectomy: potential curative therapy for early hepatocellular carcinoma. Radiology. 2018;287:1050–8.
Braat MN, van den Hoven AF, van Doormaal PJ, et al. The caudate lobe: the blind spot in radioembolization or an overlooked opportunity? Cardiovasc Intervent Radiol. 2016;39:847–54.
Kim H-C, Joo I, Lee M, et al. Benign biliary stricture after yttrium-90 radioembolization for hepatocellular carcinoma. J Vasc Interv Radiol. 2020;31:2014–21.
Kim H-C, Kim YJ, Lee J-H, et al. Feasibility of boosted radioembolization for hepatocellular carcinoma larger than 5 cm. J Vasc Interv Radiol. 2019;30:1–8.
Kim HK, Chung Y-H, Song B-C, et al. Ischemic bile duct injury as a serious complication after transarterial chemoembolization in patients with hepatocellular carcinoma. J Clin Gastroenterol. 2001;32:423–7.
Miyayama S, Yamashiro M, Okuda M, et al. Main bile duct stricture occurring after transcatheter arterial chemoembolization for hepatocellular carcinoma. Cardiovasc Intervent Radiol. 2010;33:1168–79.
Salem R, Johnson GE, Kim E, et al. Yttrium-90 radioembolization for the treatment of solitary. Unresectable HCC LEGACY Study Hepatol. 2021;74:2342–52.
Toskich B, Vidal LL, Olson MT, et al. Pathologic response of hepatocellular carcinoma treated with yttrium-90 glass microsphere radiation segmentectomy prior to liver transplantation: a validation study. J Vasc Interv Radiol. 2021;32(518–526): e511.
Dhondt E, Lambert B, Hermie L, et al. 90Y radioembolization versus drug-eluting bead chemoembolization for unresectable hepatocellular carcinoma: results from the TRACE phase II randomized controlled trial. Radiology. 2022;2022:211806.
Toskich BB, Liu DM. Y90 radioembolization dosimetry: concepts for the interventional radiologist. Tech Vasc Interv Radiol. 2019;22:100–11.
Llovet JM, Lencioni R. mRECIST for HCC: performance and novel refinements. J Hepatol. 2020;72:288–306.
Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 2017:https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf. Accessed on: 20 Oct, 2021
Padia SA, Johnson GE, Horton KJ, et al. Segmental Yttrium-90 radioembolization versus segmental chemoembolization for localized hepatocellular carcinoma: results of a single-center, retrospective, propensity score–matched study. J Vasc Interv Radiol. 2017;28(777–785): e771.
Vouche M, Habib A, Ward TJ, et al. Unresectable solitary hepatocellular carcinoma not amenable to radiofrequency ablation: multicenter radiology-pathology correlation and survival of radiation segmentectomy. Hepatology. 2014;60:192–201.
Lee U, Murakami G, Han S. Arterial supply and biliary drainage of the dorsal liver: a dissection study using controlled specimens. Anat Sci Int. 2004;79:158–66.
Yoon CJ, Chung JW, Cho BH, et al. Hepatocellular carcinoma in the caudate lobe of the liver: angiographic analysis of tumor-feeding arteries according to subsegmental location. J Vasc Interv Radiol. 2008;19:1543–50.
Benson AB, D’Angelica MI, Abbott DE, et al. Hepatobiliary cancers, version 2.2021, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2021;19:541–65.
Reig M, Forner A, Rimola J, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022;76:681–93.
Schullian P, Laimer G, Putzer D, et al. Stereotactic radiofrequency ablation of primary liver tumors in the caudate lobe. HPB (Oxford). 2020;22:470–8.
Yan Y, Lin ZY, Chen J. Analysis of imaging-guided thermal ablation puncture routes for tumors of the hepatic caudate lobe. J Cancer Res Ther. 2020;16:258–62.
Kim H-C, Chung JW, Jae HJ, et al. Caudate lobe hepatocellular carcinoma treated with selective chemoembolization. Radiology. 2010;257:278–87.
Terayama N, Miyayama S, Tatsu H, et al. Subsegmental transcatheter arterial embolization for hepatocellular carcinoma in the caudate lobe. J Vasc Interv Radiol. 1998;9:501–8.
Montazeri SA, De la Garza-Ramos C, Lewis AR, et al. Hepatocellular carcinoma radiation segmentectomy treatment intensification prior to liver transplantation increases rates of complete pathologic necrosis: an explant analysis of 75 tumors. Eur J Nucl Med Mol Imaging. 2022. Online ahead of print.
Funding
This study was not supported by any funding.
Author information
Authors and Affiliations
Contributions
The study conception and design were performed by ZD and BT. Material preparation, data collection, and analysis were performed by MA, ZD, BT, CP, SAM, AL, CR, GF, RP-F, and JMM. The first draft of the manuscript was written by MA and ZD, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Author BT is an advisor to Boston Scientific Corporation, Sirtex, Johnson and Johnson, AstraZeneca, Genentech, Eisai, Histosonics, and Vivos. Authors MA, AL, CP, AM, CR, GF, RPF, MM, and ZD declare that they have no conflict of interest.
Ethical Approval
For this type of study, formal consent is not required.
Informed Consent
This study has obtained IRB approval from Mayo Clinic College of Medicine and Science and the need for informed consent was waived.
Consent for Publication
For this type of study, consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arora, M., Toskich, B.B., Lewis, A.R. et al. Radiation Segmentectomy for the Treatment of Primary Hepatic Malignancies of the Caudate Lobe: A Case Series. Cardiovasc Intervent Radiol 45, 1485–1493 (2022). https://doi.org/10.1007/s00270-022-03250-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-022-03250-8