Abstract
Purpose
This retrospective study evaluated the efficacy, safety, and factors affecting the prognosis of transarterial chemoembolisation with irinotecan-eluting beads with CalliSpheres (DEB-TACE) for intrahepatic cholangiocarcinoma (ICC).
Materials and Methods
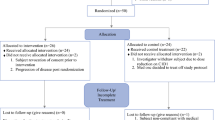
We retrospectively collected data on 39 patients with unresectable ICC who received DEB-TACE therapy. We assessed the indicators of tumour response, progression-free survival (PFS), overall survival (OS), and the incidence of adverse events. PFS and OS were analysed using Kaplan–Meier curves, while Cox analysis was used to identify factors affecting the prognosis.
Results
The 3-month objective response rate (ORR) and disease control rate (DCR) of the 39 patients with unresectable ICC were 35.9% and 56.4%, respectively, while the 6-month ORR and DCR were 23.0% and 40.9%, respectively. The median OS and PFS were 11.0 months and 8.0 months, respectively. Cox analysis demonstrated that combined therapy (adjuvant sorafenib after DEB-TACE) and a low cancer antigen (CA) 125 level (≤ 35 U/ml) were independent favourable prognostic factors. Transient elevation of the aminotransferase level, nausea, vomiting, abdominal pain, fever, and hyper-bilirubinaemia were common adverse events in patients with unresectable ICC treated with DEB-TACE with CalliSphere beads (CBs). Hepatic abscess was the most serious complication, observed in one patient.
Conclusions
DEB-TACE with CBs is a safe and well-tolerated therapy in patients with unresectable ICC with a low incidence of adverse events and relatively prolonged survival. Combined therapy and low CA125 are prognostic factors associated with longer survival.



Similar content being viewed by others
Abbreviations
- AEs:
-
Adverse events
- CB:
-
CalliSphere bead
- CI:
-
Confidence interval
- CR:
-
Complete response
- CT:
-
Computed tomography
- C-TACE:
-
Conventional TACE
- DCR:
-
Disease control rate
- DEB-TACE:
-
Drug-eluting bead TACE
- ECOG:
-
Eastern cooperative oncology group
- HR:
-
Hazard ratio
- ICC:
-
Intrahepatic cholangiocarcinoma
- MRI:
-
Magnetic resonance imaging
- mRECIST:
-
Modified response evaluation criteria in solid tumors
- ORR:
-
Objective response rate
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- PR:
-
Partial response
- SD:
-
Stable disease
References
Razumilava N, Gores GJ. Cholangiocarcinoma. Lancet. 2014;383(9935):2168–79.
Gupta A, Dixon E. Epidemiology and risk factors: intrahepatic cholangiocarcinoma. Hepatobiliary Surg Nutr. 2017;6(2):101–4.
Banales JM, Marin JJG, Lamarca A, et al. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol. 2020;17(9):557–88.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Choi S-B, Kim K-S, Choi J-Y, et al. The prognosis and survival outcome of intrahepatic cholangiocarcinoma following surgical resection: association of lymph node metastasis and lymph node dissection with survival. Ann Surg Oncol. 2009;16(11):3048–56.
Aljiffry M, Walsh MJ, Molinari M. Advances in diagnosis, treatment and palliation of cholangiocarcinoma: 1990–2009. World J Gastroenterol. 2009;15(34):4240–62.
Kim RD, McDonough S, El-Khoueiry AB, et al. Randomised phase II trial (SWOG S1310) of single agent MEK inhibitor trametinib Versus 5-fluorouracil or capecitabine in refractory advanced biliary cancer. Eur J Cancer. 2020;130:219–27.
Markussen A, Jensen LH, Diness LV, Larsen FO. Treatment of patients with advanced biliary tract cancer with either oxaliplatin, gemcitabine, and capecitabine or cisplatin and gemcitabine-a randomized phase II trial. Cancers. 2020; 12(7)
Mazzaferro V, Gorgen A, Roayaie S, Droz Dit Busset M, Sapisochin G. Liver resection and transplantation for intrahepatic cholangiocarcinoma. J Hepatol. 2020;72(2):364–77.
Hu L-S, Zhang X-F, Weiss M, et al. Recurrence patterns and timing courses following curative-intent resection for intrahepatic cholangiocarcinoma. Ann Surg Oncol. 2019;26(8):2549–57.
Wang M-L, Ke Z-Y, Yin S, Liu C-H, Huang Q. The effect of adjuvant chemotherapy in resectable cholangiocarcinoma: a meta-analysis and systematic review. Hepatobiliary Pancreat Dis Int. 2019;18(2):110–6.
Tan E, Cao B, Kim J, et al. Phase 2 study of copanlisib in combination with gemcitabine and cisplatin in advanced biliary tract cancers. Cancer. 2021;127(8):1293–300.
Chiang N, Chen M, Yang S, et al. Multicentre, phase II study of gemcitabine and S-1 in patients with advanced biliary tract cancer: TG1308 study. Liver Int. 2020;40(10):2535–43.
Chen Q, Sun Y, Dai H, Guo P, Hou S, Sha X. Assessment of irinotecan loading and releasing profiles of a novel drug-eluting microsphere (CalliSpheres). Cancer Biother Radiopharm. 2020;
Liu S, Yu G, Wang Q, et al. CalliSpheres microspheres drug-eluting bead transhepatic artery chemoembolization with or without sorafenib for the treatment of large liver cancer: a multi-center retrospective study. Am J Transl Res. 2021;13(12):13931–40.
Zhang X, Zhou J, Zhu DD, et al. CalliSpheres® drug-eluting beads (DEB) transarterial chemoembolization (TACE) is equally efficient and safe in liver cancer patients with different times of previous conventional TACE treatments: a result from CTILC study. Clin Transl Oncol. 2019;21(2):167–77.
Venturini M, Sallemi C, Agostini G, et al. Chemoembolization with drug eluting beads preloaded with irinotecan (DEBIRI) vs doxorubicin (DEBDOX) as a second line treatment for liver metastases from cholangiocarcinoma: a preliminary study. Br J Radiol. 2016;89(1067):20160247.
Martin RCG, Howard J, Tomalty D, et al. Toxicity of irinotecan-eluting beads in the treatment of hepatic malignancies: results of a multi-institutional registry. Cardiovasc Intervent Radiol. 2010;33(5):960–6.
Zhou T-Y, Zhou G-H, Zhang Y-L, et al. Drug-eluting beads transarterial chemoembolization with CalliSpheres microspheres for treatment of unresectable intrahepatic cholangiocarcinoma. J Cancer. 2020;11(15):4534–41.
Pereira PL, Iezzi R, Manfredi R, et al. The CIREL cohort: a prospective controlled registry studying the real-life use of irinotecan-loaded chemoembolisation in colorectal cancer liver metastases: interim analysis. Cardiovasc Intervent Radiol. 2021;44(1):50–62.
Lencioni R. New data supporting modified RECIST (mRECIST) for hepatocellular Carcinoma. Clin Cancer Res. 2013;19(6):1312–4.
Filippiadis DK, Binkert C, Pellerin O, Hoffmann RT, Krajina A, Pereira PL. Cirse quality assurance document and standards for classification of complications: the cirse classification system. Cardiovasc Intervent Radiol. 2017;40(8):1141–6.
Savic LJ, Chapiro J, Geschwind J-FH. Intra-arterial embolotherapy for intrahepatic cholangiocarcinoma: update and future prospects. Hepatobiliary Surg Nutr. 2017; 6(1)
Kuhlmann JB, Euringer W, Spangenberg HC, et al. Treatment of unresectable cholangiocarcinoma: conventional transarterial chemoembolization compared with drug eluting bead-transarterial chemoembolization and systemic chemotherapy. Eur J Gastroenterol Hepatol. 2012;24(4):437–43.
Peng Z, Cao G, Hou Q, et al. The comprehensive analysis of efficacy and safety of callispheres drug-eluting beads transarterial chemoembolization in 367 liver cancer patients: a multiple-center. Cohort Study Oncol Res. 2020;28(3):249–71.
Laurent A, Wassef M, Chapot R, et al. Partition of calibrated tris-acryl gelatin microspheres in the arterial vasculature of embolized nasopharyngeal angiofibromas and paragangliomas. J Vasc Interv Radiol. 2005;16(4):507–13.
Lee K-H, Liapi E, Ventura VP, et al. Evaluation of different calibrated spherical polyvinyl alcohol microspheres in transcatheter arterial chemoembolization: VX2 tumor model in rabbit liver. J Vasc Interv Radiol. 2008;19(7):1065–9.
Lee K-H, Liapi E, Vossen JA, et al. Distribution of iron oxide-containing embosphere particles after transcatheter arterial embolization in an animal model of liver cancer: evaluation with MR imaging and implication for therapy. J Vasc Interv Radiol. 2008;19(10):1490–6.
Zhang S, Huang C, Li Z, et al. Comparison of pharmacokinetics and drug release in tissues after transarterial chemoembolization with doxorubicin using diverse lipiodol emulsions and CalliSpheres Beads in rabbit livers. Drug Deliv. 2017;24(1):1011–7.
Marquardt S, Kirstein MM, Brüning R, et al. Percutaneous hepatic perfusion (chemosaturation) with melphalan in patients with intrahepatic cholangiocarcinoma: european multicentre study on safety, short-term effects and survival. Eur Radiol. 2019;29(4):1882–92.
Sohal D, Mykulowycz K, Uehara T, et al. A phase II trial of gemcitabine, irinotecan and panitumumab in advanced cholangiocarcinoma. Ann Oncol. 2013;24(12):3061–5.
Higashi M, Yamada N, Yokoyama S, et al. Pathobiological implications of MUC16/CA125 expression in intrahepatic cholangiocarcinoma-mass forming type. Pathobiology. 2012;79(2):101–6.
Mosconi C, Solaini L, Vara G, et al. Transarterial chemoembolization and radioembolization for unresectable intrahepatic cholangiocarcinoma-a systemic review and meta-analysis. Cardiovasc Intervent Radiol. 2021;44(5):728–38.
Bagante F, Spolverato G, Merath K, et al. Intrahepatic cholangiocarcinoma tumor burden: a classification and regression tree model to define prognostic groups after resection. Surgery. 2019;166(6):983–90.
Asaoka T, Kobayashi S, Hanaki T, et al. Clinical significance of preoperative CA19-9 and lymph node metastasis in intrahepatic cholangiocarcinoma. Surg Today. 2020;50(10):1176–86.
Woo S, Chung JW, Hur S, et al. Liver abscess after transarterial chemoembolization in patients with bilioenteric anastomosis: frequency and risk factors. AJR Am J Roentgenol. 2013;200(6):1370–7.
Schicho A, Pereira PL, Pützler M, et al. Degradable starch microspheres transcatheter arterial chemoembolization (DSM-TACE) in intrahepatic cholangiocellular carcinoma (ICC): results from a national multi-center study on safety and efficacy. Med Sci Monit. 2017;23:796–800.
Carter S, Martin Ii RCG. Drug-eluting bead therapy in primary and metastatic disease of the liver. HPB. 2009;11(7):541–50.
Aliberti C, Benea G, Tilli M, Fiorentini G. Chemoembolization (TACE) of unresectable intrahepatic cholangiocarcinoma with slow-release doxorubicin-eluting beads: preliminary results. Cardiovasc Intervent Radiol. 2008;31(5):883–8.
Poggi G, Quaretti P, Minoia C, et al. Transhepatic arterial chemoembolization with oxaliplatin-eluting microspheres (OEM-TACE) for unresectable hepatic tumors. Anticancer Res. 2008;28:3835–42.
Tsilimigras DI, Sahara K, Wu L, et al. Very early recurrence after liver resection for intrahepatic cholangiocarcinoma: considering alternative treatment approaches. JAMA Surg. 2020;155(9):823–31.
Goyal L, Sirard C, Schrag M, et al. Phase I and biomarker study of the Wnt pathway modulator DKN-01 in combination with gemcitabine/cisplatin in advanced biliary tract cancer. Clin Cancer Res. 2020;26(23):6158–67.
Colyn L, Bárcena-Varela M, Álvarez-Sola G, et al. Dual targeting of G9a and DNMT1 for the treatment of experimental cholangiocarcinoma. Hepatology. 2020;
Meadows V, Francis H. Doublecortin-like kinase protein 1 in cholangiocarcinoma: is this the biomarker and target we have been looking for? Hepatology. 2021;73(1):4–6.
Funding
This work was supported by grants from the Research Fund for Interventional Oncology of Beijing Medical Award Foundation (No. XM2018_011_0006_01) and Capital's Funds for Health Improvement and Research (NO. Z181100001718075).
Author information
Authors and Affiliations
Contributions
XH: Conceptualization, substantial contributions to conception, formulation or evolution of overarching research goals and aims. LD, WJ: Methodology and Writing- original draft preparation, substantial contributions to Development of methodology and original draft preparation. MZ, ZN, ZY: Resources and Methodology, substantial contributions to samples collection, acquisition of data and analysis. YX, WZ: Investigation, substantial contributions to investigation process, specifically performing the experiments and data collection.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Ethical Approval
The study was approved by the ethics committee, and each patient and guardian provided informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, D., Wang, J., Ma, Z. et al. Treatment of unresectable intrahepatic cholangiocarcinoma using transarterial chemoembolisation with irinotecan-eluting beads: analysis of efficacy and safety. Cardiovasc Intervent Radiol 45, 1092–1101 (2022). https://doi.org/10.1007/s00270-022-03108-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-022-03108-z