Abstract
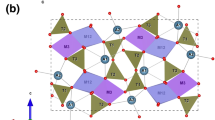
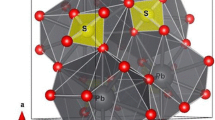
Characterization of the behavior of zeolites at high pressures is of interest both in fundamental science and for practical applications. For example, zeolites occur as a major mineral group in tuffaceous rocks (such as those at the Nevada Nuclear Security Site), and they play a key role in defining the high-pressure behavior of tuff in a nuclear explosion event. The crystal structure, Si/Al ratio, and type of pressure-transmitting media (PTM) used in high-pressure experiments influence the compressional behavior of a given zeolitic phase. The heulandite-type (HEU) zeolites, including heulandite and clinoptilolite, are isostructural but differ in their Si/Al ratios. Thus, HEU-type zeolites comprise an ideal system in unraveling the effects of Si/Al ratio and type of PTM on their pressure-induced structural behavior. In this study, we performed in situ high-pressure angle-dispersive powder synchrotron X-ray diffraction (XRD) experiments on a natural HEU zeolite, clinoptilolite, with a Si/Al ratio of 4.4, by compressing it in a diamond anvil cell (DAC) up to 14.65 GPa using a non-penetrating pressure-transmitting medium (KCl). Unit cell parameters as a function of pressure up to 9.04 GPa were obtained by Rietveld analysis. Unit cell volumes were fit to both a second and a third-order Birch–Murnaghan equation of state. The mean bulk modulus (K0) determined from all the fittings is 32.7 ± 0.9 GPa. The zero-pressure compressibility of the a-, b-, and c-axes for clinoptilolite are 10.6 (± 0.8) × 10–3 GPa–1, 5.3 (± 0.7) × 10–3 GPa–1, and 17.1 (± 1.8) × 10–3 GPa–1, respectively. The pressure–volume equations of states of this type of zeolite are important for characterizing high-pressure behavior of the broader family of microporous materials and for developing reliable geophysical signatures for underground nuclear monitoring.








Similar content being viewed by others
Data availability
All the data which is discussed in this manuscript is available to the read either in the maintext or is included in the electronic supplement.
References
Alberti A (1972) On the crystal structure of the zeolite heulandite. TMPM Tschermaks Mineral Und Petrogr Mitteilungen 18:129–146. https://doi.org/10.1007/BF01081798
Alberti A (1975) The crystal structure of two clinoptilolites. TMPM Tschermaks Mineral Und Petrogr Mitteilungen 22:25–37. https://doi.org/10.1007/BF01081301
Anderson OL (1995) Equations of state of solids for geophysics and ceramic science. Oxford University Press
Anderson DL, Anderson OL (1970) The bulk modulus-volume relationship for oxides. J Geophys Res 75:3494–3500
Anderson DL (2007) Elasticity and solid-state geophysics elastic constants of isotropic solids. In: New theory of the earth. Cambridge University Press, pp 233–245
Angel RJ (2000) Equations of state. Rev Mineral Geochem 41:35–59. https://doi.org/10.2138/rmg.2000.41.2
Arcoya A, González JA, Travieso N, Seoane XL (1994) Physicochemical and catalytic properties of a modified natural clinoptilolite. Clay Miner 29:123–131. https://doi.org/10.1180/claymin.1994.029.1.14
Arletti R, Ferro O, Quartieri S et al (2003) Structural deformation mechanisms of zeolites under pressure. Am Mineral 88:1416–1422. https://doi.org/10.2138/am-2003-1004
Armbruster T, Gunter ME (1991) Stepwise dehydration of heulandite-clinoptilolite from Succor Creek Oregon, USA: a single-crystal X-ray study at 100 K. Am Mineral 76:1872–1883
Armbruster T, Gunter ME (2001) Crystal structures of natural zeolites. Rev Mineral Geochem 45:1–67. https://doi.org/10.2138/rmg.2001.45.1
Birch F (1978) Finite strain isotherm and velocities for single-crystal and polycrystalline NaCl at high pressures and 300°K. J Geophys Res 83:1257. https://doi.org/10.1029/jb083ib03p01257
Bish DL, Carey JW (2001) Thermal behavior of natural zeolites. Rev Mineral Geochem 45:403–452. https://doi.org/10.2138/rmg.2001.45.13
Breck DW (1974) Zeolite molecular sieves. John Wiley and Sons, New York
Buldakov MA, Cherepanov VN, Korolev BV, Matrosov II (2003) Role of intramolecular interactions in Raman spectra of N2 and O2 molecules. J Mol Spectrosc 217:1–8. https://doi.org/10.1016/S0022-2852(02)00012-7
Cadar O, Senila M, Hoaghia MA et al (2020) Effects of thermal treatment on natural clinoptilolite-rich zeolite behavior in simulated biological fluids. Molecules 25:1–12. https://doi.org/10.3390/molecules25112570
Chen F, Ewing RC, Clark SB (1999) The Gibbs free energies and enthalpies of formation of U6+ phases: an empirical method of prediction. Am Mineral 84:650–664. https://doi.org/10.2138/am-1999-0418
Chidester BA, Thompson EC, Fischer RA et al (2021) Experimental thermal equation of state of B2-KCl. Phys Rev B 104:5–10. https://doi.org/10.1103/PhysRevB.104.094107
Cockreham CB, Zhang X, Goncharov VG et al (2020) Dehydration pathway of CoF2∙4H2O revisited by integrated ex situ and in situ calorimetric and structural studies. J Phys Chem C 124:3551–3556. https://doi.org/10.1021/acs.jpcc.9b08175
Comodi P, Gatta GD, Zanazzi PF (2001) High-pressure structural behaviour of heulandite. Eur J Mineral 13:497–505. https://doi.org/10.1127/0935-1221/2001/0013-0497
Comodi P, Gatta GD, Zanazzi PF (2002) High-pressure structural behaviour of scolecite. Eur J Mineral 14:567–574. https://doi.org/10.1127/0935-1221/2002/0014-0567
Coombs DS, Alberti A, Armbruster T et al (1997) Recommended nomenclature for zeolite minerals: report of the subcommittee on zeolites of the international mineralogical association, commission on new minerals and mineral names. Can Mineral 35:1571–1606
Costa GCC, Xu H, Navrotsky A (2013) Thermochemistry of barium hollandites. J Am Ceram Soc 96:1554–1561
Dewaele A, Belonoshko AB, Garbarino G et al (2012) High-pressure-high–temperature equation of state of KCl and KBr. Phys Rev B 85:1–7. https://doi.org/10.1103/PhysRevB.85.214105
Fletcher WH, Rayside JS (1974) High resolution vibration Raman spectrum of oxygen. J Raman Spectrosc 2:3–14
Fourney W, Dick R, Taylor SR, Weaver TN (1994) An analysis of three nuclear events in P-tunnel. Los Alamos National Lab., Los Alamos
Gatta GD, Lee Y (2014) Zeolites at high pressure: a review. Mineral Mag 78:267–291. https://doi.org/10.1180/minmag.2014.078.2.04
Gatta GD, Comodi P, Zanazzi PF (2003) New insights on high-pressure behaviour of microporous materials from X-ray single-crystal data. Microporous Mesoporous Mater 61:105–115. https://doi.org/10.1016/S1387-1811(03)00359-7
Gillet P, Malézieux JM, Itié JP (1996) Phase changes and amorphization of zeolites at high pressures: the case of scolecite and mesolite. Am Mineral 81:651–657. https://doi.org/10.2138/am-1996-5-612
Gonzalez-Platas J, Alvaro M, Nestola F, Angel R (2016) EosFit7-GUI: a new graphical user interface for equation of state calculations, analyses and teaching. J Appl Crystallogr 49:1377–1382. https://doi.org/10.1107/S1600576716008050
Guo X, Tiferet E, Qi L et al (2016) U(v) in metal uranates: a combined experimental and theoretical study of MgUO4, CrUO4, and FeUO4. Dalt Trans 45:4622–4632. https://doi.org/10.1039/c6dt00066e
Hazen RM (1977) Temperature, pressure, and composition: structurally analogous variables. Phys Chem Miner 1:83–94
Helean KB, Navrotsky A, Lumpkin GR et al (2003) Enthalpies of formation of U-, Th-, Ce-brannerite: implications for plutonium immobilization. J Nucl Mater 320:231–244. https://doi.org/10.1016/S0022-3115(03)00186-7
Helean KB, Ushakov SV, Brown CE et al (2004) Formation enthalpies of rare earth titanate pyrochlores. J Solid State Chem 177:1858–1866. https://doi.org/10.1016/j.jssc.2004.01.009
Hill RA, Esherick P, Owyoung A (1983) High-resolution stimulated Raman spectroscopy of O2. J Mol Spectrosc 100:119–133. https://doi.org/10.1016/0022-2852(83)90029-2
Klotz S, Chervin JC, Munsch P, Le Marchand G (2009) Hydrostatic limits of 11 pressure transmitting media. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/42/7/075413
McCusker LB, Olson DH, Baerlocher C (2007) Atlas of zeolite framework types. Elsevier Science
Momma K, Izumi F (2011) VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J Appl Crystallogr 44:1272–1276. https://doi.org/10.1107/S0021889811038970
Mumpton FA (1960) Clinoptilolite redefined. Am Mineral 45:351–369
Navrotsky A (2001) Systematic trends and prediction of enthalpies of formation of refractory lanthanide and actinide ternary oxide phases. Ceram Trans 119:137–146
Navrotsky A (2005) Thermochemical insights into refractory ceramic materials based on oxides with large tetravalent cations. J Mater Chem 15:1883–1890. https://doi.org/10.1039/b417143h
Neil CW, Hjelm RP, Hawley ME et al (2020) Small-angle neutron scattering (SANS) characterization of clay-and carbonate-rich shale at elevated pressures. Energy Fuels 34:8178–8185. https://doi.org/10.1021/acs.energyfuels.0c01009
Parikin DM, Jahja AK et al (2018) Crystal structure investigation of ferritic 73Fe24Cr2Si0.8Mn0.1Ni steel for multi-purpose structural material applications. Int J Technol 9:78–88. https://doi.org/10.14716/ijtech.v9i1.247
Passaglia E, Sheppard RA (2001) The crystal chemistry of zeolites. Rev Mineral Geochem 45:68–116. https://doi.org/10.2138/rmg.2001.45.2
Petrov DV, Matrosov II, Sedinkin DO, Zaripov AR (2018) Raman spectra of nitrogen, carbon dioxide, and hydrogen in a methane environment. Opt Spectrosc 124:8–12. https://doi.org/10.1134/S0030400X18010137
Prescher C, Prakapenka VB (2015) DIOPTAS: a program for reduction of two-dimensional X-ray diffraction data and data exploration. High Press Res 35:223–230. https://doi.org/10.1080/08957959.2015.1059835
Qi J, Guo X, Mielewczyk-Gryn A, Navrotsky A (2015) Formation enthalpies of LaLn׳O3 (Ln׳=Ho, Er, Tm and Yb) interlanthanide perovskites. J Solid State Chem 227:150–154. https://doi.org/10.1016/j.jssc.2015.03.026
Seryotkin YV (2015) Influence of content of pressure-transmitting medium on structural evolution of heulandite: single-crystal X-ray diffraction study. Microporous Mesoporous Mater 214:127–135. https://doi.org/10.1016/j.micromeso.2015.05.015
Seryotkin YV (2016) High-pressure behavior of HEU-type zeolites: X-ray diffraction study of clinoptilolite-Na. Microporous Mesoporous Mater 235:20–31. https://doi.org/10.1016/j.micromeso.2016.07.048
Seryotkin YV, Bakakin VV (2018) Structure of K-substituted zeolite clinoptillolite and its behavior upon compression in penetrating and non-penetrating media. J Struct Chem 59:1392–1399. https://doi.org/10.1134/S0022476618060203
Seryotkin YV, Bakakin VV, Likhacheva AY, Rashchenko SV (2012) High-pressure diffraction study of zeolites stilbite and stellerite. J Struct Chem 53:S26–S34. https://doi.org/10.1134/S0022476612070049
Shannon RD (1976) Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr Sect A 32:751–766. https://doi.org/10.1107/S0567739476001563
Sheppard RA, Gude AJ 3rd (1982) Mineralogy, chemistry, gas adsorption, and NH4+-exchange capacity for selected zeolitic tuffs from the Western United States. US Geological Survey
Strzelecki AC, Reece ME, Zhao X et al (2022a) Thermodynamics of mixing HREE in xenotime solid solution (Er(x)Yb(1–x)PO4). ACS Earth Sp Chem. https://doi.org/10.1021/acsearthspacechem.2c00052
Strzelecki AC, Ren Y, Chong S et al (2022b) Structure and thermodynamics of calcium rare earth silicate oxyapatites, Ca2RE8(SiO4)6O2 (RE = Pr, Tb, Ho, Tm). Phys Chem Miner. https://doi.org/10.1007/s00269-022-01187-5
Sverjensky D, Shock EL, Helgeson HC (1997) Prediction of the thermodynamic properties of aqueous metal complexes to 1000 C and 5 kb. Geochim Cosmochim Acta 61:1359–1412. https://doi.org/10.1016/S0016-7037(97)00009-4
Toby BH, Von Dreele RB (2013) GSAS-II: The genesis of a modern open-source all purpose crystallography software package. J Appl Crystallogr 46:544–549. https://doi.org/10.1107/S0021889813003531
Tsai YL, Huang E, Li YH et al (2021) Raman spectroscopic characteristics of zeolite group minerals. Minerals 11:1–14. https://doi.org/10.3390/min11020167
Vezzalini G, Quartieri S, Sani A, Levy D (2001) Structural modifications induced by high pressure in scolecite and heulandite: in-situ synchrotron X-ray powder diffraction study. Stud Surf Sci Catal 135:289. https://doi.org/10.1016/S0167-2991(01)81639-6
Xu H, Navrotsky A, Balmer ML, Su Y (2002) Crystal chemistry and phase transitions in substituted pollucites along the CsAlSi2O6-CsTiSi2O6.5 join: a powder synchrotron X-ray diffractometry study. J Am Ceram Soc 85:1235–1242
Xu H, Zhao Y, Zhang J et al (2009) Anisotropic elasticity of jarosite: a high-P synchrotron XRD study. Am Mineral 95:19–23. https://doi.org/10.2138/am.2010.3280
Zhang J, Celestian A, Parise JB, Xu H, Heaney PJ (2002) A new polymorph of eucryptite (LiAlSiO4), ε-eucryptite, and thermal expansion of α- and ε-eucryptite at high pressure. Am Mineral 87:566–571
Zhang X, Strzelecki AC, Cockreham CB et al (2021) Thermodynamics of molybdenum trioxide encapsulated in zeolite Y. AIChE J. https://doi.org/10.1002/aic.17464
Acknowledgements
We are grateful to the two anonymous reviewers for helpful comments. We thank Dr. Blake T. Sturtevant for allowing us to use the laboratory facilities of Dynamic Experiments Division at Los Alamos National Laboratory to prepare our diamond anvil cell. This research was funded by the Defense Nuclear Nonproliferation’s Nonproliferation Research and Development (NA-22) of the National Nuclear Security Administration (NNSA) and the Department of Energy (DOE). Los Alamos National Laboratory, an affirmative action/equal opportunity employer, is operated by Triad National Security, LLC, for the NNSA of U.S. DOE (Contract No. 89233218CNA00000). This work was also performed at GeoSoilEnviroCARS (The University of Chicago, Sector 13), Advanced Photon Source (APS), Argonne National Laboratory. GeoSoilEnviroCARS is supported by the National Science Foundation-Earth Sciences (EAR-1634415) and the DOE-GeoSciences (DE-FG02-94ER14466). This research further used resources of the Advanced Photon Source; a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357. Los Alamos National Laboratory strongly supports academic freedom and a researcher’s right to publish; as an institution, however, the Laboratory does not endorse the viewpoint of a publication or guarantee its technical correctness.
Author information
Authors and Affiliations
Contributions
HX, HB, and ACS designed the project. CBC and ACS performed the TG-DSC measurements. ACS was assisted by BAC in preparing the diamond anvil cell. Synchrotron XRD measurements were performed by ACS with SC and VP supporting these measurements. Rietveld analyses were performed by ACS with HX and XG supporting these analyses. This manuscript was written through contributions of all authors. All authors have given approval to the final version of this manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Strzelecki, A.C., Chariton, S., Cockreham, C.B. et al. Determination of P–V equation of state of a natural clinoptilolite using high-pressure powder synchrotron X-ray diffraction. Phys Chem Minerals 49, 45 (2022). https://doi.org/10.1007/s00269-022-01224-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00269-022-01224-3