Abstract
Background
Simultaneous bilateral thoracoscopic lung resection (SBTLR) has been shown to be a feasible and efficacious approach for a wide range of pulmonary conditions. Our aim was to evaluate the impact of different procedures on surgical outcomes in patients receiving SBTLR.
Methods
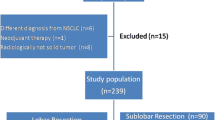
Between 2012 and 2021, 207 patients with bilateral lung neoplasms who underwent SBTLR were retrospectively reviewed. Fifty-one patients received ipsilateral plus contralateral lobectomy or sublobectomy (lobar group), whilst 156 patients received bilateral sublobectomy (sublobar group). Propensity scores were calculated and matched. Perioperative and clinicopathologic outcomes were compared.
Results
The lobar group had a greater mean age (64.5 vs. 60.0 years, p = 0.008), longer operative time (254 vs. 205 min, p < 0.001), and more blood loss (74 vs. 46 ml, p < 0.001). The sublobar group had fewer complications (6.4 vs. 19.6%, p = 0.006), shorter hospital stay (4.8 vs. 7.4 days, p < 0.001), and lower hospital costs (p = 0.03). Among 50 pairs of matched groups, significant differences were found only in operative time, hospital stay, and costs. Maximum tumor size and pathological features differed significantly before and after matching (all p < 0.05), with the lobar group consistently demonstrating a larger main tumor (median, 2.5 cm) and a higher percentage of primary lung cancer (84%). Multivariate logistic regression analysis showed that a longer operative time was the factor associated with more complications (OR: 1.01; 95% CI 1.00–1.02, p = 0.002).
Conclusions
With regard to SBTLR, our data suggests that sublobectomy may reduce the prolonged recovery, hospital costs, and complications incurred by lobectomy, without compromising oncological outcomes.




Similar content being viewed by others
References
Feldman HA, Zhou N, Antonoff MB et al (2021) Simultaneous versus staged resections for bilateral pulmonary metastases. J Surg Oncol 123:1633–1639
Liu YW, Chou A, Chou SH (2022) Experience of simultaneous bilateral open surgery and VATS for pulmonary metastasectomy. Thorac Cardiovasc Surg. https://doi.org/10.1055/s-0042-1744476
Matsubara T, Toyokawa G, Kinoshita F et al (2018) Safety of simultaneous bilateral pulmonary resection for metastatic lung tumors. Anticancer Res 38:1715–1719
Liu YW, Chang PC, Chang SJ et al (2020) Simultaneous bilateral thoracoscopic blebs excision reduces contralateral recurrence in patients undergoing operation for ipsilateral primary spontaneous pneumothorax. J Thorac Cardiovasc Surg 159:1120-1127.e3
Zhang Y, Wang Y, Lv C et al (2018) Clinical analysis of 56 cases of simultaneous bilateral video-assisted thoracoscopic surgery for bilateral synchronous multiple primary lung adenocarcinoma. J Thorac Dis 10:6452–6457
Xu G, Fu X (2019) One-stage video-assisted thoracic surgery for bilateral multiple pulmonary nodules. J Thorac Dis 11:535–541
Cai H, Xie D, Al Sawalhi S et al (2020) Subxiphoid versus intercostal uniportal video-assisted thoracoscopic surgery for bilateral lung resections: a single-institution experience. Eur J Cardiothorac Surg 57:343–349
Lin S, Yang C, Guo X et al (2021) Simultaneous uniportal video-assisted thoracic surgery of bilateral pulmonary nodules. J Cardiothorac Surg 16(1):42
Huang C, Sun Y, Wu Q et al (2021) Simultaneous bilateral pulmonary resection via single-utility port VATS for multiple pulmonary nodules: a single-center experience of 16 cases. Thorac Cancer 12:525–533
Zheng H, Peng Q, Xie D et al (2021) Simultaneous bilateral thoracoscopic lobectomy for synchronous bilateral multiple primary lung cancer-single center experience. J Thorac Dis 13(3):1717–1727
Paul S, Altorki NK, Sheng S et al (2010) Thoracoscopic lobectomy is associated with lower morbidity than open lobectomy: a propensity-matched analysis from the STS database. J Thorac Cardiovasc Surg 139:366–378
Yan TD, Black D, Bannon PG et al (2009) Systematic review and meta-analysis of randomized and nonrandomized trials on safety and efficacy of video-assisted thoracic surgery lobectomy for early-stage non-small-cell lung cancer. J Clin Oncol 27:2553–2562
Bendixen M, Jørgensen OD, Kronborg C et al (2016) Postoperative pain and quality of life after lobectomy via video-assisted thoracoscopic surgery or anterolateral thoracotomy for early stage lung cancer: a randomised controlled trial. Lancet Oncol 17:836–844
Detterbeck F, Arenberg D, Asamura H et al (2016) IASLC Staging and Prognostic Factors Committee; Advisory Boards; Multiple Pulmonary Sites Workgroup. The IASLC lung cancer staging project: background data and proposed criteria to distinguish separate primary lung cancers from metastatic foci in patients with two lung tumors in the forthcoming eighth edition of the TNM classification for lung cancer. J Thorac Oncol 11:651–665
Wu MH, Wu HY (2022) One-stage thoracoscopic resection of bilateral bronchiectasis. Asian Cardiovasc Thorac Ann 30(2):190–194
Wu MH, Wu HY (2018) Pleural drainage using drainage bag for thoracoscopic lobectomy. Asian Cardiovasc Thorac Ann 26(3):212–217
de Angelis P, Tan KS, Chudgar NP et al (2022) Operative time is associated with postoperative complications after pulmonary lobectomy. Ann Surg. https://doi.org/10.1097/SLA.0000000000005696
Martini N, Melamed MR (1975) Multiple primary lung cancers. J Thorac Cardiovasc Surg 70(4):606–612
Nakata M, Sawada S, Yamashita M et al (2004) Surgical treatments for multiple primary adenocarcinoma of the lung. Ann Thorac Surg 78(4):1194–1199
Shimada Y, Saji H, Otani K et al (2015) Survival of a surgical series of lung cancer patients with synchronous multiple ground-glass opacities, and the management of their residual lesions. Lung Cancer 88(2):174–180
Nie Y, Wang X, Yang F et al (2021) Surgical prognosis of synchronous multiple primary lung cancer: systematic review and meta-analysis. Clin Lung Cancer 22(4):341-350.e3
Subramanian M, McMurry T, Meyers BF et al (2018) Long-term results for clinical stage IA lung cancer: comparing lobectomy and sublobar resection. Ann Thorac Surg 106(2):375–381
Saji H, Okada M, Tsuboi M et al (2022) Segmentectomy versus lobectomy in small-sized peripheral non-small-cell lung cancer (JCOG0802/WJOG4607l): a multicentre, open-label, phase 3, randomised, controlled, non-inferiority trial. Lancet 399(10335):1607–1617
Zhang Y, Hu H, Wang R et al (2014) Synchronous non-small cell lung cancers: diagnostic yield can be improved by histologic and genetic methods. Ann Surg Oncol 21(13):4369–4374
Qu R, Ye F, Tu D et al (2022) Clinical features and surgical treatment of synchronous multiple primary lung adenocarcinomas with different EGFR mutations. Front Oncol 13(11):785777
Tian H, Bai G, Yang Z, Chen P et al (2023) Multiple primary lung cancer: updates of clinical management and genomic features. Front Oncol 22(13):1034752
Acknowledgements
We thank Dr Jadzia Tin-Tsen Chou for her revision of the manuscript.
Funding
None declared.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Human and animal rights
The IRB approved this study and the requirement for written informed consent was waived (KMUHIRB-E(I)-20200228, KMUHIRB-E(II)-20220192, SCMHIRB No. 1110413).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, YW., Wu, MH., Kao, CN. et al. Lobectomy Versus Sublobar Resection in Simultaneous Bilateral Thoracoscopic Lung Resection. World J Surg 47, 2568–2577 (2023). https://doi.org/10.1007/s00268-023-07081-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-023-07081-7