Abstract
Background
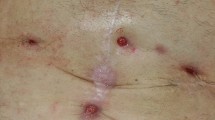
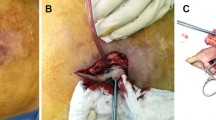
The goal of this article was to report the results about the efficacy of treatment of chronic mesh infection (CMI) after abdominal wall hernia repair (AWHR) in one-stage management, with complete mesh explantation of infected prosthesis and simultaneous reinforcement with a biosynthetic poly-4-hydroxybutyrate absorbable (P4HB) mesh.
Methods
This is a retrospective analysis of all patients that needed mesh removal for CMI between September 2016 and January 2019 at a tertiary center. Epidemiological data, hernia characteristics, surgical, and postoperative variables (Clavien–Dindo classification) of these patients were analyzed.
Results
Of the 32 patients who required mesh explantation, 30 received one-stage management of CMI. In 60% of the patients, abdominal wall reconstruction was necessary after the infected mesh removal: 8 cases (26.6%) were treated with Rives–Stoppa repair, 4 (13.3%) with a fascial plication, 1 (3.3%) with anterior component separation, and 1 (3.3%) with transversus abdominis release to repair hernia defects. Three Lichtenstein (10%) and 1 Nyhus repairs (3.3%) were performed in patients with groin hernias. The most frequent postoperative complications were surgical site occurrences: seroma in 5 (20%) patients, hematoma in 2 (6.6%) patients, and wound infection in 1 (3.3%) patient. During the mean follow-up of 34.5 months (range 23–46 months), the overall recurrence rate was 3.3%. Persistent, recurrent, or new CMIs were not observed.
Conclusions
In our experience, single-stage management of CMI with complete removal of infected prosthesis and replacement with a P4HB mesh is feasible with acceptable results in terms of mesh reinfection and hernia recurrence.



Similar content being viewed by others
References
Pande T, Naidu CS (2020) Mesh infection in cases of polypropylene mesh hernioplasty. Hernia 24(4):849–856
Plymale MA, Davenport DL, Walsh-Blackmore S et al (2020) Costs and complications associated with infected mesh for ventral hernia repair. Surg Infect (Larchmt) 21(4):344–349
Yang H, Xiong Y, Chen J et al (2020) Study of mesh infection management following inguinal hernioplasty with an analysis of risk factors: a 10-year experience. Hernia 24(2):301–305
Martin DP, Badhwar A, Shah DV et al (2013) Characterization of poly-4-hydroxybutyrate mesh for hernia repair applications. J Surg Res 184:766–773
Rognoni C, Bassi UA, Cataldo M et al (2018) Budget impact analysis of a biosynthetic mesh for incisional hernia repair. Clin Ther 40:1830–1844
Buell JF, Sigmon D, Ducoin C et al (2017) Initial experience with biologic polymer scaffold (poly-4-hydroxybuturate) in complex abdominal wall reconstruction. Ann Surg 266:185–188
Muysoms FE, Miserez M, Berrevoet F et al (2009) Classification of primary and incisional abdominal wall hernias. Hernia 13:407–414
Torregrosa-Gallud A, Sancho Muriel J, Bueno-Lledó J et al (2017) Modified components separation technique: experience treating large, complex ventral hernias at a university hospital. Hernia 21:601–608
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications. a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Liang MK, Li LT, Nguyen MT et al (2014) Abdominal reoperation and mesh explantation following open ventral hernia repair with mesh. Am J Surg 208:670–676
Boullenois H, Moszkowicz D, Poghosyan T et al (2016) Surgical management of chronic mesh infection following incisional hernia repair. J Visc Surg 153(6):461–464
Stremitzer S, Bachleitner-Hofmann T, Gradl B et al (2010) Mesh graft infection following abdominal hernia repair: risk factor evaluation and strategies of mesh graft preservation. a retrospective analysis of 476 operations. World J Surg 34:1702–1709
Shubinets V, Carney MJ, Colen DL et al (2018) Management of infected mesh after abdominal hernia repair: systematic review and single-institution experience. Ann Plast Surg 80(2):145–153
Bueno-Lledó J, Torregrosa-Gallud A, Sala-Hernandez A et al (2017) Predictors of mesh infection and explantation after abdominal wall hernia repair. Am J Surg 213:50–57
Kao AM, Arnold MR, Augenstein VA et al (2018) Prevention and treatment strategies for mesh infection in abdominal wall reconstruction. Plast Reconstr Surg 42(3 Suppl):149S–155S
Krpata DM, Blatnik JA, Novitsky YW et al (2013) Evaluation of high-risk comorbid patients undergoing open ventral hernia repair with synthetic mesh. Surgery 153:120–125
Birolini C, de Miranda JS, Tanaka EY et al (2020) The use of synthetic mesh in contaminated and infected abdominal wall repairs: challenging the dogma-a long-term prospective clinical trial. Hernia 24:307–323
Kao AM, Arnold MR, Otero J et al (2020) Comparison of outcomes after partial versus complete mesh excision. Ann Surg 272(1):177–182
Bueno-Lledo J, Torregrosa-Gallud A, Carreño-Saenz O et al (2017) Partial versus complete removal of the infected mesh after abdominal wall hernia repair. Am J Surg 214:47–52
Atema JJ, de Vries FE, Boermeester MA (2016) Systematic review and meta-analysis of the repair of potentially contaminated and contaminated abdominal wall defects. Am J Surg 212:982–995
Köckerling F, Alam NN, Antoniou SA et al (2018) What is the evidence for the use of biologic or biosynthetic meshes in abdominal wall reconstruction? Hernia 22:249–269
Baldan N, Munegato G, Di Leo A et al (2020) Lessons learned from 227 biological meshes used for the surgical treatment of ventral abdominal defects. Hernia 24(1):57–65
Scott JR, Deeken CR, Martindale RG et al (2016) Evaluation of a fully absorbable poly-4-hydroxybutyrate/absorbable barrier composite mesh in a porcine model of ventral hernia repair. Surg Endosc 30:3691–3701
Miserez M, Jairam AP, Boersema GSA et al (2019) Resorbable synthetic meshes for abdominal wall defects in preclinical setting: a literature review. J Surg Res 237:67–75
Deeken CR, Matthews BD (2013) Characterization of the mechanical strength, resorption properties, and histologic characteristics of a fully absorbable material (Poly-4-hydroxybutyrate-PHASIX Mesh) in a porcine model of hernia repair. ISRN Surg 28:2013
Abdelmoaty WF, Dunst CM, Filicori F et al (2019) Combination of surgical technique and bioresorbable mesh reinforcement of the crural repair leads to low early hernia recurrence rates with laparoscopic paraesophageal hernia repair. J Gastrointest Surg. https://doi.org/10.1007/s11605-019-04358-y[Epub ahead of print]
Levy AS, Bernstein JL, Xia JJ et al (2020) Poly-4-hydroxybutyric acid mesh compares favorably with acellular dermal matrix in tissue expander-based breast reconstruction. Ann Plast Surg. https://doi.org/10.1097/SAP.0000000000002339[Epub ahead of print]
Lake SP, Stoikes NFN, Badhwar A et al (2019) Contamination of hybrid hernia meshes compared to bioresorbable Phasix™ mesh in a rabbit subcutaneous implant inoculation model. Ann Med Surg 46:12–16
Petro CC, Rosen MJ (2018) A current review of long-acting resorbable meshes in abdominal wall reconstruction. Plast Reconstr Surg 142:84–91
Kim M, Oommen B, Ross SW et al (2014) The current status of biosynthetic mesh for ventral hernia repair. Surg Technol Int 25:114–121
Williams SF, Rizk S, Martin DP (2013) Poly-4-hydroxybutyrate (P4HB): a new generation of resorbable medical devices for tissue repair and regeneration. Biomed Tech 58:439–452
Montgomery A (2013) The battle between biological and synthetic meshes in ventral hernia repair. Hernia 17:3–11
Warren J, Desai SS, Boswell ND et al (2020) Safety and efficacy of synthetic mesh for ventral hernia repair in a contaminated field. J Am Coll Surg 230:405–413
Plymale MA, Davenport DL, Dugan A et al (2018) Ventral hernia repair with poly-4-hydroxybutyrate mesh. Surg Endosc 32:1689–1694
Buell JF, Sigmon D, Ducoin C et al (2017) Initial experience with biologic polymer scaffold (Poly-4-hydroxybuturate) in complex abdominal wall reconstruction. Ann Surg 266:185–188
Roth JS, Anthone GJ, Selzer DJ et al (2018) Prospective evaluation of poly-4-hydroxybutyrate mesh in CDC class I/high-risk ventral and incisional hernia repair: 18-month follow-up. Surg Endosc 32:1929–1936
Messa CA, Kozak G, Broach RB et al (2019) When the mesh goes away: an analysis of poly-4-hydroxybutyrate mesh for complex hernia repair. Plast Reconstr Surg Glob Open 7(11):e2576
Pakula A, Skinner R (2020) Outcomes of open complex ventral hernia repairs with retromuscular placement of poly-4-hydroxybutyrate bioabsorbable mesh. Surg Innov 27:32–37
Levy AS, Bernstein JL, Premaratne ID et al (2020) Poly-4-hydroxybutyrate (Phasix™) mesh onlay in complex abdominal wall repair. Surg Endosc. https://doi.org/10.1007/s00464-020-07601-9
Van Rooijen MMJ, Jairam AP, Tollens T et al (2018) A post-market, prospective, multi-center, single-arm clinical investigation of Phasix™ mesh for VHWG grade 3 midline incisional hernia repair: a research protocol. BMC Surg 20:104
Acknowledgements
We would like to thank Santiago Bonafé, Providencia Garcia-Pastor, Victoria Pareja, José Iserte and Raquel Blasco of our surgical group for their helpful discussions.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Bueno-Lledó has been speaker for BD surgical and had grants with BD surgical (workshops). These disclosures do not impact the topics reported herein. All other authors declare no disclosures relevant to this article or research. Drs. Ceno, Perez-Alonso, Martinez-Hoed, Torregrosa-Gallud and Pous-Serrano have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bueno-Lledó, J., Ceno, M., Perez-Alonso, C. et al. Biosynthetic Resorbable Prosthesis is Useful in Single-Stage Management of Chronic Mesh Infection After Abdominal Wall Hernia Repair. World J Surg 45, 443–450 (2021). https://doi.org/10.1007/s00268-020-05808-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-020-05808-4