Abstract
Background
The Omega-3 (ω-3) polyunsaturated fatty acids (PUFAs) generate bioactive lipid mediators that reduce inflammation. The present study evaluated the effect of SMOFlipid containing ω-3 PUFAs on wound healing.
Methods
Rats were divided into a SMOFlipid (SMOF) group and a 0.9% saline (placebo) group, with eight rats in each group. Wound excision was performed on the dorsal surface of each rat. In the SMOF group, 1 gm/kg SMOFlipid was dissolved in 3 mL saline as a treatment; in the placebo group, 3 mL saline was prepared as a treatment. The treatments were administered intravenously at an initial rate of 0.2 mL/kg body weight/h immediately after wounding, for 72 h. Blood samples were collected for white blood cell, tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-10 measurements at the baseline and at 1, 6, 12, 24, 48, and 72 h after intervention. Wound areas were measured over a 2-week period after excision, and a histological examination was performed.
Results
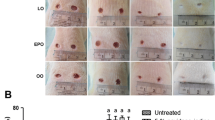
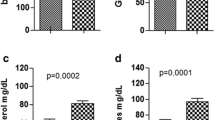
Compared with the placebo group, SMOFlipid supplementation engendered significant decreases in the wound area on day 3 (78.28 ± 5.25 vs. 105.86 ± 8.89%), day 5 (72.20 ± 4.31 vs. 96.39 ± 4.72%), day 10 (20.78 ± 1.28 vs. 39.80 ± 10.38%), and day 14 (7.56 ± 0.61 vs. 15.10 ± 2.42%). The placebo group had a higher TNF-α level than the SMOF group at 72 h. The IL-10 level was higher in the SMOF group than in the placebo group at 48 h. Histological analysis revealed a higher rate of fibroblast distribution and collagen fiber organization in the SMOF group (P = 0.01).
Conclusion
SMOFlipid enriched in ω-3 PUFA accelerates wound healing.




Similar content being viewed by others
References
Klein KC, Guha SC (2014) Cutaneous wound healing: current concepts and advances in wound care. Indian J Plast Surg 47(3):303–317
Soleimani Z, Hashemdokht F, Bahmani F et al (2017) Clinical and metabolic response to flaxseed oil omega-3 fatty acids supplementation in patients with diabetic foot ulcer: a randomized, double-blind, placebo-controlled trial. J Diabetes Complicat 31(9):1394–1400
de Castilho TJ, Campos AC, Mello EV (2015) Effect of omega-3 fatty acid in the healing process of colonic anastomosis in rats. Arg Bras Cir Dig 28(4):258–261
Baena-Gómez MA, Aguilar MJ, Mesa MD et al (2015) Changes in antioxidant defense system using different lipid emulsions in parenteral nutrition in children after hematopoietic stem cell transplantation. Nutrients 7(9):7242–7255
Ren T, Cong L, Wang Y et al (2013) Lipid emulsions in parenteral nutrition: current applications and future developments. Expert Opin Drug Deliv 10(11):1533–1549
Klek S, Chambrier C, Singer P et al (2013) Four-week parenteral nutrition using a third generation lipid emulsion (SMOFlipid)—a double-blind, randomised, multicentre study in adults. Clin Nutr 32(2):224–231
Uthaya S, Liu X, Babalis D et al (2016) Nutritional Evaluation and Optimisation in Neonates: a randomized, double-blind controlled trial of amino acid regimen and intravenous lipid composition in preterm parenteral nutrition. Am J Clin Nutr 103(6):1443–1452
Dai YJ, Sun LL, Li MY et al (2016) comparison of formulas based on lipid emulsions of olive oil, soybean oil, or several oils for parenteral nutrition: a systematic review and meta-analysis. Adv Nutr 7(2):279–286
Tian H, Yao X, Zeng R et al (2013) Safety and efficacy of a new parenteral lipid emulsion (SMOF) for surgical patients: a systematic review and meta-analysis of randomized controlled trials. Nutr Rev 71(12):815–821
Bolisetty S, Osborn D, Sinn J, Lui K (2012) Standardised neonatal parenteral nutrition formulations—an Australasian group consensus. BMC Pediatr 14:48–59
Li H, Ruan XZ, Powis SH et al (2005) EPA and DHA reduce LPS-induced inflammation responses in HK-2 cells: evidence for a PPAR-gamma-dependent mechanism. Kidney Int 67(3):867–874
Calder PC, Yaqoob P (2009) Understanding omega-3 polyunsaturated fatty acids. Postgrad Med 121(6):148–157
Baldan CS, Masson IF, Esteves Junior I et al (2015) Inhibitory effects of low-level laser therapy on skin-flap survival in a rat model. Plast Surg 23(1):35–39
Abramov Y, Golden B, Sullivan M et al (2007) Histologic characterization of vaginal vs. abdominal surgical wound healing in a rabbit model. Wound Repair Regen 15(1):80–86
Khoshmohabat H, Dalfardi B, Dehghanian A et al (2016) The effect of CoolClot hemostatic agent on skin wound healing in rats. J Surg Res 200(2):732–737
Kamer E, Recai Unalp H, Gundogan O et al (2010) Effect of ascorbic Acid on incisional wound healing in streptozotocin-induced diabetic rats. Wounds 22(2):27–31
McDaniel JC, Massey K, Nicolaou A (2011) Fish oil supplementation alters levels of lipid mediators of inflammation in microenvironment of acute human wounds. Wound Repair Regen 19(2):189–200
Kendall AC, Nicolaou A (2013) Bioactive lipid mediators in skin inflammation and immunity. Prog Lipid Res 52(1):141–164
Jafari Naveh HR, Taghavi MM, Shariati M et al (2011) Both omega-3 and omega-6 polyunsaturated fatty acids stimulate foot wound healing in chronic diabetic rat. Afr J Pharm Pharmacol 5(14):1713–1717
Lee RP, Wang D, Lin NT et al (2002) Physiological and chemical indicators for early and late stages of sepsis in conscious rats. J Biomed Sci 9(6 Pt 2):613–621
McDaniel JC, Belury M, Ahijevych K et al (2008) Omega-3 fatty acids effect on wound healing. Wound Repair Regen 16(3):337–345
Lu Y, Tian H, Hong S (2010) Novel 14,21-dihydroxy-docosahexaenoic acids: structures, formation pathways, and enhancement of wound healing. J Lipid Res 51(5):923–932
McDaniel JC, Ahijevych K, Belury M (2010) Effect of omega-3 oral supplements on the omega-6/omega-3 ratio in young adults. West J Nurs Res 32(1):64–80
Fontes JA, Rose NR, Čiháková D (2015) The varying faces of IL-6: from cardiac protection to cardiac failure. Cytokine 74(1):62–68
Choi JH, Jun JH, Kim JH et al (2014) Synergistic effect of interleukiomega-6 and hyaluronic acid on cell migration and ERK activation in human keratinocytes. J Korean Med Sci 29(Suppl 3):S210–S216
Muldoon MF, Laderian B, Kuan DC et al (2016) Fish oil supplementation does not lower C-reactive protein or interleukiomega-6 levels in healthy adults. J Intern Med 279(1):98–109
Hankenson KD, Watkins BA, Schoenlein IA et al (2000) Omega-3 fatty acids enhance ligament fibroblast collagen formation in association with changes in interleukiomega-6 production. Proc Soc Exp Biol Med 223(1):88–95
Stenvinkel P, Ketteler M, Johnson RJ et al (2005) IL-10, IL-6, and TNF-alpha: central factors in the altered cytokine network of uremia—the good, the bad, and the ugly. Kidney Int 67(4):1216–1233
Balaji S, King A, Marsh E et al (2015) The role of interleukin-10 and hyaluronan in murine fetal fibroblast function in vitro: implications for recapitulating fetal regenerative wound healing. PLoS ONE 10(5):e0124302
Kieran I, Knock A, Bush J et al (2013) Interleukin-10 reduces scar formation in both animal and human cutaneous wounds: results of two preclinical and phase II randomized control studies. Wound Repair Regen 21(3):428–436
de Miranda Torrinhas RS, Santana R, Garcia T et al (2013) Parenteral fish oil as a pharmacological agent to modulate post-operative immune response: a randomized, double-blind, and controlled clinical trial in patients with gastrointestinal cancer. Clin Nutr 32(4):503–510
Acknowledgements
This work was partially supported by the Tzu Chi University (TCIRP 101002-01Y1). This manuscript was edited by Wallace Academic Editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors declare any conflict of interest.
Rights and permissions
About this article
Cite this article
Peng, YC., Yang, FL., Subeq, YM. et al. Lipid Emulsion Enriched in Omega-3 PUFA Accelerates Wound Healing: A Placebo-Controlled Animal Study. World J Surg 42, 1714–1720 (2018). https://doi.org/10.1007/s00268-017-4404-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-017-4404-x