Abstract
Background
We previously showed comparable volume effects of injections of acellular adipose matrix (AAM), an adipose tissue-derived extracellular matrix, and conventional fat grafting in a murine model. Thus, AAM could be a novel allogenic injectable product. However, its retention rate poses a concern, as repeated AAM injections may be required in some cases. This study investigated the biological properties and therapeutic value of stored AAM and compared them with those of fresh AAM, in a murine model.
Methods
AAM was manufactured from fresh human abdominoplasty fat. Fresh and stored injectable AAM was prepared within 24 h and 3 months after generation, respectively. Either fresh or stored injectable AAM was injected into the scalp of athymic nude mice (0.2 mL/sample, n = 6 per group). After 8 weeks, graft retention was assessed through weight measurement, and histological analysis was performed, including immunofluorescence staining for CD31 and perilipin.
Results
Retention rate was significantly reduced in the stored compared to the fresh injectable AAM group. Nevertheless, histological analysis revealed comparable inflammatory cell presence, with minimal capsule formation, in both groups. Adipogenesis occurred in both groups, with no significant difference in the blood vessel area (%) between groups.
Conclusions
Although the volume effects of stored AAM for soft tissue reconstruction were limited compared to those of fresh injectable AAM, stored AAM had similar capacity for adipogenesis and angiogenesis. This promising allogeneic injectable holds the potential to serve as an effective “off-the-shelf” alternative for repeated use within a 3-month storage period.
No Level Assigned
This journal requires that authors assign a level of evidence to each submission to which Evidence-Based Medicine rankings are applicable. This excludes Review Articles, Book Reviews, and manuscripts that concern Basic Science, Animal Studies, Cadaver Studies, and Experimental Studies. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors https://link.springer.com/journal/00266.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Soft tissue augmentation is a significant requirement in plastic and reconstructive surgery. The number of commercially available soft tissue fillers has increased. Hyaluronic acid-based filler is the most commonly used owing to its excellent immune profile and minimal allergy concerns [1, 2]. However, it does not replicate all characteristics of living tissue and has a volume effect of only up to 1 year; hence, repeated injections are required to maintain the desired outcome. Autologous fat grafting can be considered as a standard for filling and rejuvenation [3]; however, it has a significant drawback in that graft retention is unpredictable. A meta-analysis study involving 1011 patients who underwent facial fat grafting showed fat graft retention ranging from 26 to 83% [4]. Ideal material for soft tissue augmentation should be biocompatible, avoid immune complications, and offer long-term volume maintenance by encouraging new fat formation.
Recently, acellular adipose matrix (AAM) was introduced as a potential soft tissue reconstruction material. This is an extracellular matrix isolated from adipose tissue. Briefly, fat tissue undergoes mechanical, chemical, and biological processes to remove cellular components completely [5]. AAM can potentially act as a scaffold that not only supports stem cells in proliferation and differentiation but also induce adipogenesis and angiogenesis [5, 6]. AAM has minimal immunogenicity [6, 7], serving as an off-the-shelf biocompatible filler in plastic and reconstructive surgery and tissue engineering [8]. Comparable volume effects for injectable AAM and lipoaspirate have been shown previously in a murine model; thus, AAM could be a promising allogenic injectable product [9].
However, the retention rate of AAM is a concern, and repeated injections of AAM may be required in some cases [10]. This study investigated the biological properties and therapeutic value of stored AAM, compared to fresh AAM (i.e., AAM used within 24 hours after generation) in a murine model.
Patients and Methods
Adipose Tissue Harvesting and Injectable AAM Processing
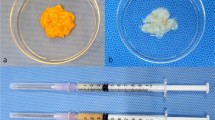
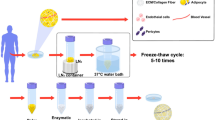
The study protocol was approved by the relevant institutional review board. Abdominal adipose tissue samples were sourced from discarded tissues of patients aged 30–40 years undergoing transverse rectus abdominis myocutaneous flap surgery for breast reconstruction. Fat tissue was used to generate AAM using mechanical, chemical, and biological processes, following previously published protocols [9]. Adipose tissue underwent five freeze-thawing cycles and was then centrifuged at 1500×g for 10 min [11]. After washing, the fatty portion was discarded, and the tissue was treated with polar solvent extraction using 99.9% isopropanol over 8 h [12]. After washing, the processed tissue was incubated in a solution of 0.05% trypsin, 0.05% EDTA, 10 ng/ml DNAse I (Sigma–Aldrich; Merck KGaA, Darmstadt, Germany), and 10 ng/ml RNAse (Sigma-Aldrich; Merck KGaA) for 2 h, with slow rotation, in an incubator at 37 °C. Finally, the tissue was washed four times with phosphate-buffered saline (PBS) for 30 min each time and subsequently treated with 1% penicillin (Sigma-Aldrich; Merck KGaA) and streptomycin (Sigma-Aldrich; Merck KGaA) for 12 h at 4 °C [13]. It was then dried under ultraviolet light for 4 h at room temperature. To fabricate the “fresh injectable AAM,” the final material was meticulously minced with sharp scissors prior to use within 24 h. AAM was stored at 4 °C in PBS for 3 months prior to use as the “stored injectable AAM” [14]. It was then dried and minced in the same manner as the “fresh injectable AAM”.
Scanning Electron Microscopy
Fresh and stored AAM samples were analyzed by scanning electron microscopy (SEM) to confirm cellular components and to investigate the matrix architecture. Samples were fixed in 2.5% glutaraldehyde for 24 h at 4 °C. Samples were then moved to a cover glass slide and air-dried at room temperature. The surface morphology of the AAM was observed by SEM (JSM-7401F, JEOL Inc., Akashima, Japan) following coating with platinum at an accelerating voltage of 15 kV.
Animal Models and Procedures
Eight-week-old athymic nude mice (weight: 20–25 g) were purchased from Koatech (Gyeonggi, South Korea). The mice were housed in a temperature-controlled environment at 24 ± 2 °C, with an artificial 12-h light/dark cycle. All applicable institutional and/or national guidelines for the care and use of animals were followed.
Twelve mice were anesthetized with 3% isoflurane in 100% oxygen at a delivery rate of 5 L/min for anesthesia induction. They were randomly divided into two groups (n = 6 per group): either fresh AAM or stored AAM samples were randomly injected into the mice at the supraperiosteal plane of the skull, using 18-gauge needles (0.2 mL/sample). At postoperative week 8, all mice were anesthetized and the grafts were extirpated. The grafts were evaluated using weight measurement. The graft portion was excised using a scalpel and scissors and subjected to macroscopic photography. Following macroscopic observation of each animal treated with injected fresh and stored AAM, five out of six samples were randomly subjected to histological analysis. All the anesthetized mice were sacrificed at the end of the experiment.
Histomorphometric Analysis
Five tissue samples in each group were fixed overnight in 4% paraformaldehyde (BD Biosciences, Franklin Lakes, NJ, USA), dehydrated, and embedded in paraffin. Slides of 4-μm thickness were prepared for hematoxylin–eosin staining, to compare the amounts of inflammatory cells, and Masson's trichrome staining, to delineate collagen distribution. Images were observed using a light microscope (Nikon ECLIPSE Ts2, Nikon, Tokyo, Japan). Five random fields were selected and evaluated for inflammation independently by two investigators, based on a semiquantitative scale, ranging from 0 to 5, with 0 = none, 1 = minimal presence (< 20%), 2 = minimal to moderate presence (20–40%), 3 = moderate presence (40–60%), 4 = moderate to extensive presence (60–80%), and 5 = extensive presence (> 80%). The average values were used for analysis.
Unstained slides were deparaffinized with xylene, rehydrated, and boiled in citrate buffer (Agilent Technologies, Santa Clara, CA, USA) for antigen retrieval. Samples were then blocked with 5% goat serum (Jackson ImmunoResearch, West Grove, PA, USA.) for 1 h and incubated overnight at 4 °C with primary antibodies. After washing, samples were incubated with secondary antibodies for 2 h at room temperature. Slides were counterstained with 40,6-diamidino-2-phenylindole to visualize the cell nuclei. The primary antibodies used included rabbit anti-CD31 (PA5-16301; Invitrogen, Waltham, MA, USA) and rabbit anti-perilipin (PA5-72921; Invitrogen). The secondary antibodies used were fluorescein-conjugated goat anti-rabbit. Immunofluorescence-stained images were visualized using a confocal microscope (SP8; Leica, Wetzlar, Germany). Five 500 × 500 μm-sized random fields from each sample were analyzed using ImageJ (Fiji) software (National Institutes of Health, Bethesda, MD, USA) and the average % area values were determined.
Immunofluorescence staining was evaluated in five paraffin block samples from both groups by confocal microscopy. Perilipin staining was employed to detect adipogenesis in the graft materials. For a quantitative evaluation of adipogenesis, perilipin-positive adipocyte counts were measured in five 500 × 500 μm-sized fields.
Statistical Analysis
The results were analyzed using nonparametric Mann–Whitney U tests because the sample size was small, and data were not normally distributed. R version 4.0.3 (https://www.r-project.org/) was used for statistical analyses, and p-values < 0.05 were considered statistically significant.
Results
3D Microstructure of Fresh and Stored AAM
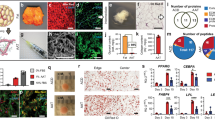
Microscopic examination of SEM images demonstrated that both fresh and stored AAM exhibited a finely organized, parallel fibrillar structure, devoid of cellular components. By comparing the SEM images of the two preparations, we observed that the fresh AAM consisted of organized nano-fibrous collagen 3D microstructure with more voluminous fibers and micropores. The stored AAM, in contrast, showed a nano-fibrous collagen 3D microstructure with more shrunken and compressed fibers and fewer micropores (Fig. 1).
Macroscopic Observation of Graft Retention
Extirpated graft samples in both groups were round and firm with microvasculature present on the surface, which could not be distinguished with the naked eye by two observers. The visible microvasculature highlighted the angiogenic effects of both the fresh and stored AAM (Fig 2).
Quantitative Analysis of Graft Retention
The mean initial weights from the three measurements of the material present in 0.2 ml of injectable AAM (both fresh and stored) were 0.175 g. The weights of the grafted materials at postoperative week 8 were measured. The retained sample weight (%) of the injected materials was larger in the fresh AAM group than in the stored AAM group (fresh AAM group: median = 44.9%, interquartile range = 35.4–49.1%; stored AAM group: median = 23.4%, interquartile range = 24.0–22.4%, p = 0.013, n = 6) (Fig. 3).
Histological Findings of Graft Retention
Five randomly selected graft samples from each group were subjected to histological analysis. Inflammatory cell infiltrations are observed in hematoxylin–eosin-stained sections and collagenous fibrous tissue component are observed in Masson’s trichrome-stained sections (Fig. 4). Inflammatory cell infiltration in the grafted materials showed no statistically significant difference between the fresh and stored AAM groups (fresh AAM group: median = 1, interquartile range = 1–2; stored AAM group: median = 1, interquartile range = 1–2, p = 0.905, n = 5).
Histological findings of a section of extirpated mass of the fresh (a, c) and stored (b, d) AAM group. Inflammatory cell infiltrations are observed in hematoxylin–eosin-stained sections (a, b) and collagenous fibrous tissue component are observed in Masson’s trichrome-stained sections (c, d). Magnification 200 ×, scale bar, 50 µm. AAM, acellular adipose matrix
For a quantitative evaluation of collagen proportion, %area values, representing the collagen %area, were measured in five 500 × 500 μm-sized fields. We found no significant difference in collagen proportion between the fresh and stored AAM groups (fresh AAM group: median = 84.7%, interquartile range = 71.7–85.4%; stored AAM group: median = 73.1%, interquartile range = 71.2–74.4%, p = 0.403, n = 5) (Fig. 5).
Analysis of Adipogenesis and Angiogenesis
We found no significant difference in perilipin-positive adipocyte number between the fresh and stored AAM groups (fresh AAM group: median = 17.6, interquartile range = 4.4–31.6; stored AAM group: median = 14.8, interquartile range = 12.4–17.2, p = 0.999, n = 5) (Figs. 6 and 7).
CD31 staining was employed to identify angiogenesis, a marker of endothelial cells in blood vessels. Many perilipin-positive cells were observed in both fresh and stored AAM groups (Fig. 8). For a quantitative evaluation of angiogenesis, the %area values, representing the blood vessel area (%) in five 500 × 500 μm-sized fields were randomly determined. CD31-positive staining did not differ between the fresh and stored AAM groups (fresh AAM group: median = 1.61%, interquartile range = 1.42–1.72%; stored AAM group: median = 1.24%, interquartile range = 1.13–1.35%, p = 0.2101, n = 5) (Fig. 9).
Discussion
Although injectable AAM has shown effectiveness comparable to conventional fat grafting, the issue of effectiveness depending on storage period had to be identified [9]. To increase the utility value of a product as a “off-the-shelf” merchandise, its effectiveness should be maintained as much as possible throughout the storage period. Therefore, additional research was planned considering that the product should be utilizable during its storage period. Here, we investigated the biological properties and therapeutic value of stored AAM, as compared to fresh AAM (i.e., AAM used within 24 h after generation) in a murine model. The retention rate was significantly reduced in the stored compared to the fresh injectable AAM group. Nevertheless, histological analysis revealed comparable inflammatory cell presence, adipogenesis, and angiogenesis, with minimal capsule formation, in both the fresh and stored injectable groups.
A previous study showed that the volume effect of AAM is similar to that of clinically well-known conventional fat grafting [9]. Furthermore, AAM has potential clinical benefit because of its convenience. AAM can be used more easily in patients than lipoaspirate, which should be harvested from donor sites of the patients prior to injection procedures [15]. This material could reduce not only time but also physical and psychological strain of the patients due to its unique advantages and convenience [8], and could avoid donor site morbidity. Thus, although using AAM as an “off-the-shelf” product may incur a financial burden, it remains an attractive and competitive material from a cost-effectivity point of view.
In various previous basic studies related to AAM material, the prepared AAM was usually stored in a 1% penicillin and streptomycin solution at 4 °C [16,17,18,19,20]. Although the impact of the storage method has not been accurately analyzed, we followed this previous storage method as an up-to-date reference.
In the in vitro study, the macroscopic and microscopic structure of the product was preserved after storage for 3 months, although there was a difference in the SEM microstructure. The density of the fresh and stored material remained similar. The in vivo study revealed that the volume effect was reduced in the stored AAM group; however, we showed, both qualitatively and quantitatively, that the unique properties of AAM, i.e., adipogenesis and angiogenesis, were preserved. Therefore, it can be assumed that the volume effect partially decreases depending on the storage period due to factors other than these two main characteristics. Additional research will be needed to confirm the mechanism.
Moreover, this study revealed that unlike conventional autologous fat, the effectiveness of AAM is limited if it is stored for a certain period of time after production. Nevertheless, although it is better to use the product as soon as possible after production, it can be stored for a while. This could increase the ease of distribution of this material that can be manufactured from discarded human fat. Furthermore, this could be a clinical merit as compared to conventional fat grafting, as an individual patient could then store the remaining product after the first use and could then repeat the procedure after 3 months when partial absorption occurred or to increase the volume in soft tissue reconstruction, and repeatedly refine the contouring process. This would therefore increase cost-effectiveness.
This study had a few limitations. First, as it was not a clinical study, the same product may lead to different results in clinical settings. Second, only a few mice were included in each group, which might have led to random errors or a low statistical power. Third, unintentional performance bias might have occurred while injecting AAM or extirpating the grafted mass sample in the two groups, as this was a labelled-group experiment. Additionally, serial evaluations, such as 1-month, 6-month, or longer follow-up evaluations were not performed. Finally, there was no precise evaluation of safety and complications according to the storage period.
Conclusion
We showed that 3-month-stored injectable AAM demonstrated a limited volume effect in comparison to that of fresh injectable AAM in soft tissue reconstruction. However, the ability of adipogenesis and angiogenesis of the fresh and stored AAM was similarly preserved. This promising allogeneic injectable holds the potential to serve as an effective “off-the-shelf” alternative for repeated use for soft tissue reconstruction within a 3-month storage period. Further studies are needed to assess changes in AAM characteristics and utility over time and to determine the safe storage period for this material.
References
Bogdan Allemann I, Baumann L (2008) Hyaluronic acid gel (Juvéderm) preparations in the treatment of facial wrinkles and folds. Clin Interv Aging 3:629–634
Verpaele A, Strand A (2006) Restylane SubQ, a non-animal stabilized hyaluronic acid gel for soft tissue augmentation of the mid- and lower face. Aesthet Surg J 26(1S):S10–S17
Coleman SR (2006) Structural fat grafting: more than a permanent filler. Plast Reconstr Surg 118(3 Suppl):108S–120S
Lv Q, Li X, Qi Y, Gu Y, Liu Z, Ma GE (2021) Volume retention after facial fat grafting and relevant factors: a systematic review and meta-analysis. Aesthet Plast Surg 45:506–520
Wu I, Nahas Z, Kimmerling KA, Rosson GD, Elisseeff JH (2012) An injectable adipose matrix for soft-tissue reconstruction. Plast Reconstr Surg 129:1247–1257
Kokai LE, Sivak WN, Schilling BK et al (2020) Clinical evaluation of an off-the-shelf allogeneic adipose matrix for soft tissue reconstruction. Plast Reconstr Surg Glob Open 8:e2574
Choi YC, Choi JS, Kim BS, Kim JD, Yoon HI, Cho YW (2012) Decellularized extracellular matrix derived from porcine adipose tissue as a xenogeneic biomaterial for tissue engineering. Tissue Eng Part C Methods 18:866–876
Nguyen MTN, Tran HLB (2021) Fabrication of an injectable acellular adipose matrix for soft tissue regeneration. J Sci Adv Mater Devices 6:1–10
Kim J, Tran VVT, Hong KY, Chang H (2024) Effect of injectable acellular adipose matrix on soft tissue reconstruction in a murine model. Aesthet Plast Surg 48:2210–2219. https://doi.org/10.1007/s00266-024-03924-3
Anderson AE, Wu I, Parrillo AJ et al (2022) An immunologically active, adipose-derived extracellular matrix biomaterial for soft tissue reconstruction: concept to clinical trial. NPJ Regen Med 7:6
Song M, Liu Y, Hui L (2018) Preparation and characterization of acellular adipose tissue matrix using a combination of physical and chemical treatments. Mol Med Rep 17:138–146
Flynn LE (2010) The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials 31:4715–4724
Zhao Y, Fan J, Bai S (2019) Biocompatibility of injectable hydrogel from decellularized human adipose tissue in vitro and in vivo. J Biomed Mater Res B Appl Biomater 107:1684–1694
Yang JZ, Qiu LH, Xiong SH et al (2020) Decellularized adipose matrix provides an inductive microenvironment for stem cells in tissue regeneration. World J Stem Cells 12:585–603
Xiong C, Yao W, Tao R et al (2024) Application of decellularized adipose matrix as a bioscaffold in different tissue engineering. Aesthet Plast Surg 48:1045–1053
Lin CY, Liu TY, Chen MH, Sun JS, Chen MH (2016) An injectable extracellular matrix for the reconstruction of epidural fat and the prevention of epidural fibrosis. Biomed Mater 11:035010
Turner AE, Flynn LE (2012) Design and characterization of tissue-specific extracellular matrix-derived microcarriers. Tissue Eng Part C Methods 18:186–197
Han TT, Toutounji S, Amsden BG, Flynn LE (2015) Adipose-derived stromal cells mediate in vivo adipogenesis, angiogenesis and inflammation in decellularized adipose tissue bioscaffolds. Biomaterials 72:125–137
Omidi E, Fuetterer L, Reza Mousavi S, Armstrong RC, Flynn LE, Samani A (2014) Characterization and assessment of hyperelastic and elastic properties of decellularized human adipose tissues. J Biomech 47:3657–3663
Kuljanin M, Brown CFC, Raleigh MJ, Lajoie GA, Flynn LE (2017) Collagenase treatment enhances proteomic coverage of low-abundance proteins in decellularized matrix bioscaffolds. Biomaterials 144:130–143
Funding
Open Access funding enabled and organized by Seoul National University. The authors have no financial or institutional interest in any of the drugs, materials, or devices described in this article.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Human and Animal Rights
The study protocol received approval from the institutional review board of our institution. All applicable institutional and/or national guidelines for the care and use of animals were followed. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
For this type of study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, J., Tran, V.V.T., Hong, K.Y. et al. Comparison of Stored and Fresh Injectable Acellular Adipose Matrix in Soft Tissue Reconstruction in a Murine Model. Aesth Plast Surg (2024). https://doi.org/10.1007/s00266-024-04175-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00266-024-04175-y