Abstract
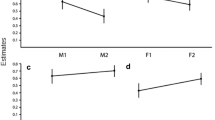
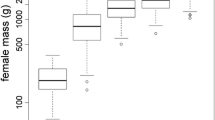
The age and parity of female primates could reflect their physical condition and reproductive experience. Consequently, the individual age-parity dependent condition of mothers and the survival of their offspring represents a fitness tradeoff. This reproductive tradeoff is expressed in the individual birth sex ratio, offspring mortality and interbirth interval (IBI). Maternal antenatal and postnatal investment hypotheses predict that more female offspring are produced by experienced mothers in poor condition (Trivers-Willard hypothesis), and that offspring mortality decreases and IBI shortens with parity (targeted reproductive effort hypothesis). Here we test the latter predictions in captive populations of the endangered Francois’ langur (Trachypithecus francoisi), using long-term demographic and reproductive data from 21 Chinese zoos and three breeding centers. In these captive populations, birth sex ratio changed slightly from male-biased to female-biased as parity increased above five offspring in experienced mothers, consistent with the Trivers-Willard hypothesis. Although mortality of male offspring was greater than female offspring, overall offspring mortality did not vary with maternal parity. There was no significant difference in duration of postnatal care or IBI between male and female offspring. However, IBI for both female and male offspring shortened overall with increasing parity, as predicted by the targeted reproductive effort hypothesis. Females with more reproductive experience that produce more female offspring are critical to captive breeding programs that aim to increase the overall viable population size of this endangered species.
Significance statement
For primates with a slow life-history, it is important to understand how mothers age and parity affects their reproductive fitness. We investigated maternal investment strategies of captive Francois’ langurs, specifically the effect of age and parity on the birth sex ratio, offspring mortality and interbirth interval. Mothers relied more on parity (i.e., their experience) to adjust the birth sex ratio, and had more sons in the first few parities. Mothers also spent significantly less time taking care of offspring as their reproductive experience increased and physical condition was likely declining. Thus, Francois’ langurs are able to adjust their investment in reproduction as they become more experienced (greater parity) to optimize reproductive fitness.




Similar content being viewed by others
Data availability
All data generated or analysed during this study are available in Figshare with the identifier https://doi.org/10.6084/m9.figshare.24125259.
Code availability
R code is available at https://doi.org/10.6084/m9.figshare.24125259.
References
Alberts SC (2019) Social influences on survival and reproduction: Insights from a long-term study of wild baboons. J Anim Ecol 88:47–66. https://doi.org/10.1111/1365-2656.12887
Altmann J, Gesquier L, Galbany J, Onyango PO, Alberts SC (2010) Life history context of reproductive aging in a wild primate model. Ann NY Acad Sci 1204:127–138. https://doi.org/10.1111/j.1749-6632.2010.05531.x
Anderson CM (1986) Female age: Male preference and reproductive success in primates. Int J Primatol 7:305–326. https://doi.org/10.1007/BF02736394
Bercovitch FB (2002) Sex-biased parental investment in primates. Int J Primatol 23:905–921. https://doi.org/10.1023/A:1015585117114
Bercovitch FB, Lebron MR, Martinez HS, Kessler MJ (1998) Primigravidity, body weight, and costs of rearing first offspring in rhesus macaques. Am J Primatol 46:135–144. https://doi.org/10.1002/(SICI)1098-2345(1998)46:2%3c135::AID-AJP3%3e3.0.CO;2-X
Bercovitch FB, Widdig A, Nürnberg P (2000) Maternal investment in rhesus macaques (Macaca mulatta): reproductive costs and consequences of raising sons. Behav Ecol Sociobiol 48:1–11. https://doi.org/10.1007/s002650000204
Boesch (1997) Evidence for dominant wild female chimpanzees investing more in sons. Anim Behav 54:811–815. https://doi.org/10.1006/anbe.1996.0510
Borries C, Koenig A, Winkler P (2001) Variation of life history traits and mating patterns in female langur monkeys (Semnopithecus entellus). Behav Ecol Sociobiol 50:391–402. https://doi.org/10.1007/s002650100391
Broussard DR, Risch TS, Dobson FS, Murie JO (2003) Senescence and age-related reproduction of female Columbian ground squirrels. J Anim Ecol 72:212–219. https://doi.org/10.1046/j.1365-2656.2003.00691.x
Cameron EZ, Linklater WL, Stafford KJ, Minot EO (2000) Aging and improving reproductive success in horses: declining residual reproductive value or just older and wiser? Behav Ecol Sociobiol 47:243–249. https://doi.org/10.1007/s002650050661
Campos FA, Altmann J, Cords M et al (2022) Female reproductive aging in seven primate species: Patterns and consequences. P Natl Acad Sci USA 119:e2117669119. https://doi.org/10.1073/pnas.2117669119
Caro TM, Sellen DW, Parish A, Frank R, Mulder MB (1995) Termination of reproduction in nonhuman and human female primates. Int J Primatol 16:205–220. https://doi.org/10.1007/BF02735478
Clutton-Brock TH, Albon SD (1982) Parental investment in male and female offspring in mammals. In: Group KCS (ed) Current Problems in Sociobiology. Cambridge University Press, Cambridge, pp 223–248
Colas S (1999) Evidence for sex-biased behavioral maternal investment in the gray mouse lemur (Microcebus murinus). Int J Primatol 20:911–926. https://doi.org/10.1023/A:1020878618941
Cui LW, Sheng AH, He SC, Xiao W (2006) Birth seasonality and interbirth interval of captive Rhinopithecus bieti. Am J Primatol 68:457–463. https://doi.org/10.1002/ajp.20241
Dolhinow P, McKenna JJ, Laws JVH (1979) Rank and reproduction among female langur monkeys: Aging and improvement (They’re not just getting older, they’re getting better). Aggressive Behav 5:19–30. https://doi.org/10.1002/1098-2337(1979)5:1%3c19::AID-AB2480050104%3e3.0.CO;2-7
Dubuc C, Ruiz-Lambides A, Widdig A (2014) Variance in male lifetime reproductive success and estimation of the degree of polygyny in a primate. Behav Ecol 25:878–889. https://doi.org/10.1093/beheco/aru052
Fairbanks LA (1996) Individual differences in maternal style: causes and consequences for mothers and offspring. Adv Stud Behav 25:579–611. https://doi.org/10.1016/S0065-3454(08)60343-5
Fan PL, He X, Yang YZ, Liu XF, Zhang HB, Yuan L, Chen W, Liu D, Fan PF (2021) Reproductive parameters of captive female northern white-cheeked (Nomascus leucogenys) and yellow-cheeked (Nomascus gabriellae) gibbons. Int J Primatol 42:49–63. https://doi.org/10.1007/s10764-020-00187-y
Fedigan LM, Carnegie SD, Jack KM (2008) Predictors of reproductive success in female white-faced capuchins (Cebus capucinus). Am J Phys Anthropol 137:82–90. https://doi.org/10.1002/ajpa.20848
Gadgil M, Bossert WH (1970) Life historical consequences of natural selection. Am Nat 104:1–24. https://doi.org/10.1086/282637
Gao XF (2014) Status of captive francois’s leaf monkey in China. Chin J Wildl 35:267–270. https://doi.org/10.19711/j.cnki.issn2310-1490.2014.03.005
Gibson MA, Mace R (2003) Strong mothers bear more sons in rural Ethiopia. Proc R Soc Lond B 270:S108–S109. https://doi.org/10.1098/rsbl.2003.0031
Gloneková M, Brandlová K, Pluháček J (2020) Higher maternal care and tolerance in more experienced giraffe mothers. Acta Ethol 23:1–7. https://doi.org/10.1007/s10211-019-00328-4
Green WCH (1990) Reproductive effort and associated costs in bison (Bison bison): do older mothers try harder? Behav Ecol 1:148–160. https://doi.org/10.1093/beheco/1.2.148
Green WCH (1993) Social effects of maternal age and experience in bison: pre-and post-weaning contact maintenance with daughters. Ethology 93:146–160. https://doi.org/10.1111/j.1439-0310.1993.tb00985.x
Groves C (2001) Primate Taxonomy. Smithsonian Institution Press, Washington DC
Guan CY, Chen Z, Huang CM, Zhou QH (2022) Analysis of fragmentation in landscape pattern for the Francois’s langur’s habitats in Guangxi and its influence on population. Acta Ecol Sin 42:1203–1212. https://doi.org/10.5846/stxb202101010003
Guo AX, Li W, Zhang W, Tian ML, Xie XC (2020) Study on reproduction technology of captive francois’s langurs (Trachypithecus francoisi) and growth and development of the young monkeys. Anim Husb Vet Med 52:125–128
Hoffman CL, Higham JP, Mas-Rivera A, Ayala JE, Maestripieri D (2010) Terminal investment and senescence in rhesus macaques (Macaca mulatta) on Cayo Santiago. Behav Ecol 21:972–978. https://doi.org/10.1093/beheco/arq098
Hu G (2007) Socioecology and behavioural flexibility of francois' langur (Trachypithecus francoisi) in Mayanghe Nature Reserve, Southwest China. Openresearch-repository.anu.edu.au. https://doi.org/10.25911/5d63c4ef22855
IUCN (2022) The IUCN Red List of Threatened Species. IUCN, Cambridge
Jin T, Wang DZ, Zhao Q, Yin LJ, Qin DG, Ran WZ, Pan WS (2009) Reproductive parameters of wild Trachypithecus leucocephalus: seasonality, infant mortality and interbirth interval. Am J Primatol 71:558–566. https://doi.org/10.1002/ajp.20688
Jin T, Wang DZ, Pan WS, Yao M (2015) Nonmaternal infant handling in wild white-headed langurs (Trachypithecus leucocephalus). Int J Primatol 36:269–287. https://doi.org/10.1007/s10764-015-9823-7
Jones JH (2011) Primates and the evolution of long, slow life histories. Curr Biol 21:R708–R717. https://doi.org/10.1016/j.cub.2011.08.025
Künkele J, Kenagy G (1997) Inefficiency of lactation in primiparous rats: the costs of first reproduction. Physiol Zool 70:571–577. https://doi.org/10.1086/515862
Mace GM (1990) Birth sex ratio and infant mortality rates in captive western lowland gorillas. Folia Primatol 55:156–165. https://doi.org/10.1159/000156511
Maestripieri D (2002) Maternal dominance rank and age affect offspring sex ratio in pigtail macaques. J Mammal 83:563–568. https://doi.org/10.1644/1545-1542(2002)083%3c0563:MDRAAA%3e2.0.CO;2
Ostner J, Borries C, Schülke O, Koenig A (2005) Sex allocation in a colobine monkey. Ethology 111:924–939. https://doi.org/10.1111/j.1439-0310.2005.01102.x
Parga JA, Lessnau RG (2005) Female age-specific reproductive rates, birth seasonality, and infant mortality of ring-tailed lemurs on St. Catherines Island: 17-year reproductive history of a free-ranging colony. Zoo Biol 24:295–309. https://doi.org/10.1002/zoo.20062
Paul A, Kuester J, Podzuweit D (1993) Reproductive senescence and terminal investment in female Barbary macaques (Macaca sylvanus) at Salem. Int J Primatol 14:105–124. https://doi.org/10.1007/BF02196506
Pollet TV, Fawcett TW, Buunk AP, Nettle D (2009) Sex-ratio biasing towards daughters among lower-ranking co-wives in Rwanda. Biol Lett 5:765–768. https://doi.org/10.1098/rsbl.2009.0394
Qi XG, Li BG, Ji WH (2008) Reproductive parameters of wild female Rhinopithecus roxellana. Am J Primatol 70:311–319. https://doi.org/10.1002/ajp.20480
R Development Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org
Rapaport LG, Kloc B, Warneke M, Mickelberg JL, Ballou JD (2013) Do mothers prefer helpers? Birth sex-ratio adjustment in captive callitrichines. Anim Behav 85:1295–1302. https://doi.org/10.1016/j.anbehav.2013.03.018
Robbins AM, Robbins MM, Gerald-Steklis N, Steklis HD (2006) Age-related patterns of reproductive success among female mountain gorillas. Am J Phys Anthropol 131:511–521. https://doi.org/10.1002/ajpa.20474
Roof KA, Hopkins WD, Izard MK, Hook M, Schapiro SJ (2005) Maternal age, parity, and reproductive outcome in captive chimpanzees (Pan troglodytes). Am J Primatol 67:199–207. https://doi.org/10.1002/ajp.20177
Saltz D, Rubenstein DI (1995) Population dynamics of a reintroduced Asiatic wild ass (Equus hemionus) herd. Ecol Appl 5:327–335. https://doi.org/10.2307/1942025
Setchell JM, Lee PC, Wickings EJ, Dixson AF (2002) Reproductive parameters and maternal investment in mandrills (Mandrillus sphinx). Int J Primatol 23:51–68. https://doi.org/10.1023/A:1013245707228
Sheldon BC, West SA (2004) Maternal dominance, maternal condition, and offspring sex ratio in ungulate mammals. Am Nat 163:40–54. https://doi.org/10.1086/381003
Shelmidine N, Borries C, McCann C (2009) Patterns of reproduction in Malayan silvered leaf monkeys at the Bronx Zoo. Am J Primatol 71:852–859. https://doi.org/10.1002/ajp.20712
Shen FJ, Zhang ZH, He W, Yue BS, Zhang AJ, Zhang L, Hou R, Wang CD, Watanabe T (2009) Microsatellite variability reveals the necessity for genetic input from wild giant pandas (Ailuropoda melanoleuca) into the captive population. Mol Ecol 18:1061–1070. https://doi.org/10.1111/j.1365-294X.2009.04086.x
Silk JB (1988) Maternal investment in captive Bonnet macaques (Macaca radiata). Am Nat 132:1–19. https://doi.org/10.1086/284834
Stiver KA (2021) Male reproductive variance. In: Shackelford TK, Weekes-Shackelford VA (eds) Encyclopedia of Evolutionary Psychological Science. Springer, Cham, 4721–4725. https://doi.org/10.1007/978-3-319-19650-3_1975
Swedell L, Leedom L, Saunders J, Pines M (2014) Sexual conflict in a polygynous primate: costs and benefits of a male-imposed mating system. Behav Ecol Sociobiol 68:263–273. https://doi.org/10.1007/s00265-013-1641-3
Tanaka Y, Fukano Y, Nakamura M (2019) Effect of paternal age on the birth sex ratio in captive populations of aye-aye (Daubentonia madagascariensis (Gmelin)). Zoo Biol 38:389–392. https://doi.org/10.1002/zoo.21487
Trivers RL, Willard DE (1973) Natural selection of parental ability to vary the sex ratio of offspring. Science 179:90–92. https://doi.org/10.1126/science.179.4068.90
Veller C, Haig D, Nowak MA (2016) The Trivers-Willard hypothesis: sex ratio or investment? Proc R Soc B 283:20160126. https://doi.org/10.1098/rspb.2016.0126
Weladji RB, Mysterud A, Holand Ø, Lenvik D (2002) Age-related reproductive effort in reindeer (Rangifer tarandus): evidence of senescence. Oecologia 131:79–82. https://doi.org/10.1007/s00442-001-0864-6
Wich SA, Steenbeek R, Sterck EH, Korstjens AH, Willems EP, van Schaik CP (2007) Demography and life history of Thomas langurs (Presbytis thomasi). Am J Primatol 69:641–651. https://doi.org/10.1002/ajp.20386
Williams GC (1957) Pleiotropy, natural selection, and the evolution of senescence. Evolution 11:398–411. https://doi.org/10.1126/sageke.2001.1.c
Williams GC (1966) Natural selection, the costs of reproduction, and a refinement of Lack’s principle. Am Nat 100:687–690. https://doi.org/10.1086/282461
Wu AK, Luo Y, Wang SL, Chen ZR, Wang B (2006) Preliminary study on breeding periodicity of wild Francois’ langurs (Trachypithecus francoisi) in Mayanghe Nature Reserve Guizhou Guizhou. Acta Theriologica Sinica 26:303–306. https://doi.org/10.16829/j.slxb.2006.03.015
Zhao D, Li B, Watanabe K (2011a) Impact of group size on female reproductive success of free-ranging Rhinopithecus roxellana in the Qinling Mountains, China. Folia Primatol 82:1–12. https://doi.org/10.1159/000326058
Zhao Q, Borries C, Pan WS (2011b) Male takeover, infanticide, and female countertactics in white-headed leaf monkeys (Trachypithecus leucocephalus). Behav Ecol Sociobiol 65:1535–1547. https://doi.org/10.1007/s00265-011-1163-9
Zhao X, Ren B, Li D, Garber PA, Zhu P, Xiang Z, Grueter CC, Liu Z, Li M (2019) Climate change, grazing, and collecting accelerate habitat contraction in an endangered primate. Biol Conserv 231:88–97. https://doi.org/10.1016/j.biocon.2019.01.007
Acknowledgements
We thank the editor and reviewers for their helpful comments and suggestions. We also thank all staff, that managed François’ s langurs in 21 zoos and three breeding centers in China for their industrious work and excellent standards of husbandry to the Francois’ langurs.
Funding
This work was supported by Key Project of Guangxi Natural Science Foundation of China (Grant number 2022GXNSFDA080004) (PF); National Natural Science Foundation of China (Grant numbers 32270504, 31900335) (CH and PF); Key Laboratory of Ecology of Rare and Endangered Species and Environmental Protection (Guangxi Normal University), Ministry of Education, China (Grant number ERESEP2022Z01) (PF).
Author information
Authors and Affiliations
Contributions
PF and QZ designed the study; SW, XL and YL collected the data; BY analyzed the data; PF, BY and MJL wrote the manuscript; all authors revised the manuscript and gave final approval for publication.
Corresponding authors
Ethics declarations
Ethical approval
This study was approved by the animal ethics committee of the Guangxi Normal University, China. All applicable international, national and/or institutional guidelines for the use of animals were followed.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by K. Langergraber
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Communicated by Qihai Zhou and Penglai Fan.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yi, B., Wang, S., Sun, T. et al. Maternal parity influences the birth sex ratio and birth interval of captive Francois’ langur (Trachypithecus francoisi). Behav Ecol Sociobiol 77, 140 (2023). https://doi.org/10.1007/s00265-023-03408-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-023-03408-5