Abstract
Stopover territoriality, i.e., the defense and broadcast of an ephemeral home range during avian migration, is rare or perhaps overlooked. However, settling on a territory may allow for easier access to resources and therefore promote faster refueling despite the added costs of territory defense. We examined the occurrence of stopover territoriality and its relationship to body condition and refueling rates. We color-ringed Western Subalpine Warblers (Curruca iberiae) mapped their home ranges and measured associated habitat characteristics during a spring stopover in the Sahara. We simulated territory intrusions via song playback to quantify territorial behavior. Males aggressively defended and broadcasted their stopover territories in response to intruders. Initial body condition in males was negatively related to home range vegetation density, suggesting that males in higher condition are selective for certain territory features. Low vegetation density does not support habitat selection for foraging, but perhaps does support social monitoring or predator vigilance as its functions. Fuel deposition rates in recaptured males were unrelated to home range habitat characteristics. Female Subalpine Warblers did not use repeated home ranges yet had similar refueling rates, body conditions, and stopover durations as territorial males. Therefore, while stopover territories were selected based on body condition, territory characteristics did not influence refueling rates. Subalpine Warblers expressed reduced migratory characteristics, making prolonged stopovers of up to 27 days with low refueling rates but maintaining body condition while defending territories. These results suggest that some individuals are staging for non-energetic reasons, such as pre-breeding preparations, or transitioning between a migratory and a breeding life history stage.
Significance statement
Territoriality in birds is commonly associated with breeding activities, less so with the wintering period and rarely with migratory periods. However, territoriality during migration may be energetically beneficial. During a trans-Saharan stopover, male warblers establish and defend temporary territories by singing and engaging in physical defense of home ranges, and those in better condition occupied territories with sparser vegetation. Females were not territorial but refueled at similar rates as males. Stopover territoriality did not increase access to food-rich patches, but perhaps aided in efficient social monitoring and preparations for the approaching breeding season. This is the first detailed documentation of birds defending ephemeral territories during migration, and its potential functions, costs and benefits may inform studies of full-annual cycles and life histories of many migratory species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Avian migration is characterized as a series of flights and stopovers, where birds rest, recover, and refuel for subsequent flights (e.g., Salewski et al. 2007; Skrip et al. 2015; Eikenaar et al. 2020b). Details regarding activity, behavior, and habitat selection during stopover may be one of the most consequential but often neglected aspects of migration ecology (Moore et al. 1995; Cohen et al. 2017, Cohen and Satterfield 2020; Schmaljohann et al. 2022). Specifically, stopover territoriality has received little attention; yet, it may help overcome some ecological and energetic challenges while en route, such as reliably acquiring food while reducing exposure to predators and unfavorable weather conditions (Davies and Houston 1981; Cuadrado 1997; Arizaga et al. 2011). Territoriality is the defense and broadcast of a restricted home range by an individual against potential intruders (Brown 1969). Therefore, territoriality occurs when both the (a) spatial (restricted movement; hereafter termed “home range”) and (b) behavioral (defense and broadcast) components are observed in concert (Noble 1939; Emlen 1957; Stamps 1990; Chernetsov and Mukhin 2006). Importantly, we do not use the terms “home range” and “territory” synonymously, but by our aforementioned definitions to differentiate between a physical space, and a physical space that is defended, respectively (Brown 1969; Stamps 1990). Stopover territoriality simply specifies its occurrence at an ephemerally occupied stopover site, in the midst of migration. It has very seldom been documented and evaluated as a migratory strategy (e.g., Rappole and Warner 1976; Kodric-Brown and Brown 1978; Bibby and Green 1980, 1981; Carpenter et al. 1993), suggesting it may be rare, overlooked, or difficult to discern.
From an energetic standpoint, territories that are economically defensible, where the costs of maintenance, monitoring, and defense are lower than the energetic benefits accrued over the duration of occupancy are beneficial investments (Kodric-Brown and Brown 1978; Davies and Houston 1981). This is commonly referred to as the “good feeding patch” hypothesis, and it stands as a strong candidate to potentially explain stopover territoriality (Davies and Houston 1981; Snow and Snow 1984; Cuadrado 1997). Defending a resource-rich territory during stopover may increase fueling efficiency (Rappole and Warner 1976; Bibby and Green 1980; Carpenter et al. 1993), and reduce energy expenditure from “floating” among potentially suboptimal habitat patches (Arizaga et al. 2011). Additional resources that might warrant defense include access to shelter from predators and weather, vigilance locations for predators and competitors, and access to water sources (Cuadrado 1997; Arizaga et al. 2011). Any resource that increases condition or improves chances for a successful migration could warrant defense during migration. Indeed, preferential access to higher quality stopover territories may be mediated by aggression and dominance, which is related to body condition in birds (Carrascal et al. 1998; Marra 2000; Marra and Holmes 2001; Catry et al. 2004; Jonart et al. 2007; Beltrão et al. 2021). Alternatively, individuals unable or unwilling to establish a home range but remaining at the stopover site for some days (termed “floaters” following Rappole and Warner 1976) may briefly intrude into a number of occupied territories for foraging and predator avoidance.
We studied the stopover ecology of the Western Subalpine Warbler (Curruca iberiae; Fig. 1; “Western” is hereafter omitted) during a spring trans-Saharan stopover. Subalpine Warblers spend winters in the Sahelian vegetation belt just south of the Sahara Desert and breed on the Iberian Peninsula in the spring and summer (Morel and Morel 1992; Svensson 2013; Zuccon et al. 2020; BirdLife International 2021). They are territorial during their stationary breeding and wintering periods (Pons et al. 2008; Mostafa et al. 2021). Recently, the inornata subspecies (sensu Svensson 2013), which breeds entirely in North Africa, was synonymized with the Iberian group (Zuccon et al. 2020). However, because the two populations are separable by plumage characteristics and measurement criteria, we only included the Iberian population in this study (Shirihai and Svensson 2018; Zuccon et al. 2020). Subalpine Warblers, especially males, are conspicuously aggressive towards conspecifics during spring trans-Saharan stopovers and broadcast their positions with calls and songs (Salewski et al. 2007). Therefore, they may be territorial even during migration.
A Subalpine Warbler (Curruca iberiae; a) pictured here perched on a Tamarisk (Tamarix aphylla) in Yasmina, Morocco. This species breeds on the Iberian Peninsula (b) and spends winters in the Sahel Zone of central Africa (c). This study was carried out in Yasmina, Morocco (d), a stopover site in the Northern Sahara Desert, between March and May 2018, 2019, and 2022. This range map was generated using data from BirdLife International (BirdLife International 2021)
Here, we evaluate the occurrence of stopover territoriality and its relationship to body condition and refueling rates. First, we experimentally test the hypothesis that males broadcast and defend territories during stopover. We test this by mapping home ranges and measuring behavioral responses to a simulated territory intrusion. Second, we assessed which habitat characteristics are favored during stopover. Home range settlement in birds typically results from competitive exclusion, which is mediated by body condition (Marra 2000; Catry et al. 2004). Therefore, the most favorable home range characteristics should be selected by individuals in high body condition. For instance, we predict that individuals that stopover in higher body condition will select home ranges with higher vegetation density (a proxy for both food abundance and cover against predators; Haigh 1996; Cuadrado 1997; McGrath et al. 2009). Finally, we test the “good feeding patch” hypothesis during stopover. If home ranges are selected for their energetic value, then habitat characteristics indicative of food resources (such as vegetation density) should be related to fuel deposition rates of respective home range occupants. In sum, we evaluate territorial behavior during stopover, home range habitat selection, and relative energetic value of home ranges for migrating birds.
Methods
Study site and capture
We conducted this study at Yasmina Oasis, Morocco (31.213° N, 03.988° W; Fig. 1) in the Northern Sahara Desert, from March 5 to May 9, 2018, February 27 to May 10, 2019, and March 16 to April 25, 2022. The region is characterized by sand dunes and rocky desert with sparse vegetation. Yasmina Oasis is a dry lakebed surrounded by a shore of evergreen tamarisk trees (Tamarix aphylla). The lake itself remained dry for the duration of the study except for a small gray-water pond, which occasionally held some refuse water (115 m2, 1 m deep). We established twenty 12-m mist-nets within the vegetated areas and monitored them every hour to half-hour. We passively captured Subalpine Warblers (i.e., without playback) and fitted with an aluminum ring as well as a unique combination of color rings. After sexing and aging, a single observer (MI) measured the wing chord (mm), ninth primary (mm), fat score (0–8), and muscle score (0–3) and recorded the mass with an accuracy of 0.1 g (all following EURING guidelines, Bairlein 1995). The birds were then released back into the field site.
Spot-mapping, home range, and vegetation estimates
We conducted spot-mapping of color-ringed Subalpine Warblers nearly every hour by marking their positions (1 m accuracy) on an aerial field map along with time of day, interactions, and vocalizations. These data were entered into a QGIS database for spatial analysis (QGIS Development Team 2018).
Subalpine Warblers display obvious interspecific and intraspecific aggression during stopover (Salewski et al. 2007). In Yasmina, male Subalpine Warblers rarely tolerated conspecifics and did not share individual trees or bushes (AAA, pers. observation). In cases where two were seen together, a fight would almost certainly ensue and ultimately end in the displacement of one. Considering the obvious intolerance for sharing space, if an individual was seen in a tree, the measured vegetation characteristics of that tree (vegetation density, height, and planar area) were assigned as a component of its home range area. Home ranges regularly contained more than one tree and were sometimes separated by spaces of bare dirt or sand. Subalpine Warblers do not use bare exposed areas for foraging but rather to fly between trees. Therefore, an individual’s functional home range area was calculated as the sum of the vegetated areas it used as these contain valuable resources (food, shade, predation cover, and space). However, we included exposed area (the sum of non-vegetated area bounded by vegetation of a home range) as a separate and possibly confounding variable. We removed from the analyses resights of marked individuals made on the initial capture day because it may take at least a day to establish restricted home ranges even during migration (Aborn and Moore 1997; Chernetsov and Mukhin 2006). Rather, movements upon arrival are largely exploratory and directed towards gaining information about the unfamiliar stopover site (Németh and Moore 2014).
At the end of each season, we measured the vegetation density, height and planar area of every individual tamarisk tree in Yasmina. We consider one seasonal sample of each tamarisk sufficient because as evergreen trees, the vegetation structure was not expected or observed to change strongly. We used a cover pole to estimate vegetation density following but modifying procedures outlined in Robel et al. (1970). The cover pole was made of white plastic with black markers at each meter, placed in the center of the tree and read from a distance of 5 m by a single observer at eye level and always from 0° north. Percent visual obstruction (vegetation covering the pole) was estimated by 6 cover classes (1 = 0–5%, 2 = 5–25%, 3 = 25–50%, 4 = 50–75%, 5 = 75–95%, 6 = 95–100%) for every meter of height and converted to the midpoints of each cover class (2.5, 15.5, 37.5, 62.5, 85, and 97.5 respectively) following Daubenmire (1959). This method provides coarse but fast estimates and correlates positively with vegetation biomass (Robel et al. 1970). The percent visual obstruction of each meter strata was averaged for each tree yielding a relative vegetation density estimate to be compared across the site. The height of each tree was measured with a large, marked pole. The planar area of each tree was measured by manually converting treetops as seen from aerial imagery into bounded polygons in QGIS.
Because water can influence fueling in migratory birds (Sapir et al. 2004), we measured distances between home range centroids (calculated in QGIS) and the gray-water pond centroids as a potentially influential habitat characteristic. While we did not ever observe subalpine warblers drinking this water, it could influence insect abundance or vegetation properties.
Song recordings and playback of males
Male Subalpine Warblers regularly broadcasted chips and songs during stopover. We made audio recordings of singing Subalpine Warblers during March 2019 with a Sennheiser ME66 directional head, battery-powered K6 power module (Sennheiser, Wedemark, Germany), and Tascam DR-05 WAV recorder (Tascam, Montebello, California, USA). Song recordings coupled with decibel readings were made from 5 m of the focal individual (using a Voltcraft SL-10 digital decibel reader, Wollerau, Switzerland). We made recordings of ten unique individuals, which varied naturally in terms of length and intensity. We filtered all recordings to remove unwanted low noises below 750 Hz and normalized them to equal levels with Audacity 2.2.2 software (Audacity Team 2020). Longer recordings were cut to make shorter segments of singing males. We constructed 20 individual 10-min playback tracks. Each playback track consisted of alternating patterns of 30–45 s of song, followed by 30–45 s of silence. These were constructed based on the general patterns of the field recording. The placement and order of song segments were assorted randomly from the ten individuals to avoid pseudoreplication, but they were always kept as alternating between song and silence. We also constructed control sequences with identical structure as the playback tracks except that song segments were swapped for static white noise generated with Audacity 2.2.2. White noise was used to control for potential disturbances from the observer and noise from the speaker. All tracks were played through a Sandisk Clipsport WAV player (Sandisk, Milpitas, California, USA) and broadcast from a Roland Minicube amplifying speaker (Roland Corp., Hamamatsu, Shizuoka, Japan). The speaker volume was set so that the intensity from 5 m was 40 dB, the decibel readings of real Subalpine Warblers singing from that distance.
Simulated male-male territorial intrusion experiments
In order to measure the extent of male-male territory defense to a simulated territory intrusion (STI), we selected individuals appearing to be settled on restricted home ranges. We randomly assigned individuals to experimental or control treatments, corresponding to song playback or white noise respectively. We conducted STIs for each individual over 3 days in the early morning (prior to 0900). The protocol began with placing the speaker in the middle of the territory and walking away to 15–20 m and allowing 10 min of silence. After this, the playback was activated (either a song or a white noise track). The playback tracks were randomly selected using the shuffle feature of the WAV player. A single observer (AAA) made a 10-min observation measuring number of chips, number of songs, latency to sing, latency to approach the speaker, nearest approach to speaker (0.2 m accuracy within 5 m; 1 m accuracy outside 5 m), and time spent within 5 m of the speaker. Blind observations were not possible as all individuals were marked; however, we actively attempted to minimize potential biases by using a Silva Starter stopwatch (Silva Sweden AB, Bromma, Sweden), a click counter, and distance-marking sticks at one and five meters around the speaker for all trials. The trials were repeated identically over three successive days for the same individuals to evaluate changes during stopover.
Body condition, fuel deposition rate and home range characteristics
Calculations and statistical analyses were achieved using R version 3.6.2 (R Core Team 2019). We calculated a size-corrected body mass by generating a scaled mass index (SMI; Eq. 2 in Peig and Green 2009). The SMI controls for body length by incorporating a scaling relationship (standardized major axis) which was calculated with a regression of mass against wing chord. Scaling relationships of mass and wing chord differed significantly between years (adj. r2 = 0.217, F3, 601 = 56.680, P < 0.001). Therefore, standardized major axes were calculated separately according to the average wing length from their respective year. The SMI is a good predictor of energy reserves relative to other body condition indices (Peig and Green 2010). Accordingly, SMI calculations from our data significantly predicted fat (r2 = 0.342, F1, 468 = 245.1, P < 0.001); the primary endogenous energy source used during migration (McWilliams et al. 2004). Therefore, we interpret SMI as a direct measure of body condition; hence, we use these terms synonymously.
We passively netted all recaptured individuals, i.e., without the use of song playback or other attractants. Fuel deposition rate (FDR; defined as change in mass over time) was calculated by multiple linear regressions. We calculated a coefficient for change in SMI between final and initial capture against (a) days between capture events and (b) time between captures events, and an intercept of zero (Schaub and Jenni 2000). An FDR model was generated for each year separately to account for potential global differences in fueling. The resulting models were FDR2018 = (0.271* Δ SMI/hour) + (0.434* Δ SMI/day) (adj. r2 = 0.3348, F2, 45 = 12.830, P < 0.001); FDR2019 = (0.011* Δ SMI/hour) + (0.604* Δ SMI/day) (adj. r2 = 0.435, F2, 95 = 36.600, P < 0.001); and FDR2022 = (0.068* Δ SMI/hour) + (0.895* Δ SMI/day) (adj. r2 = 0.469, F2, 42 = 18.54, P < 0.001).
We used multiple linear regressions to model home range habitat characteristics. Dependent variables were vegetation height, vegetation density, distance to water, roundness, and exposed area (i.e., bare dirt). Roundness, calculated as the perimeter of a circle with the same area as a home range polygon divided by the home range perimeter, was shown to be an important breeding territory characteristic in Curruca warblers (Pons et al. 2008). Independent variables used to model habitat parameters were body condition, arrival date (first capture), age class (after hatch year or after second year), and year. Finally, conspecific density (net captures of males and both sexes) on initial capture date was included to assess relative “contender pressure” on home range characteristics (Myers et al. 1979).
Additionally, we used a linear regression to model the effects of selected home range characteristics on FDR. Independent variables for FDR were functional home range area, roundness, exposed area, vegetation height, vegetation density, and distance to water. Terms for stopover schedule such as arrival date, stopover duration (last day resighted-first day captured, following Dierschke et al. 2005; Maggini et al. 2015), and individual characteristics such as age (after hatch year or after second year) initial body condition and year sampled (2018, 2019, or 2022), were also incorporated in the FDR model.
In order to evaluate our models and select the best performers, we used a multiple model inference approach (Burnham and Anderson 2002) using the R package ‘MuMIn’ (Barton 2010). We fit the full models and reduced parameters in a stepwise approach, ranking competing models by Akaike’s information criterion for small samples size (AICc).
Playback experiment statistical analysis
Playback responses were not normally distributed; therefore, nearest approach distance and latency to chip were cube-root transformed. The time spent within 5 m of the speaker was processed with Yeo-Johnson transformation to achieve normality (Yeo and Johnson 2000). Playback responses were tested using mixed effect models including terms for treatment (white noise and song playback), day (1, 2, 3), an interaction term of treatment and day, and individual as a random factor to account for repeated measures over 3 days. Latency to sing and latency to approach were analyzed only within the song playback treatment because birds exposed to white noise controls never responded by singing or approaching the speaker. Therefore, they were tested only across days with individual as a random effect.
We checked all models for adherence to their respective assumptions. Linearity between quantiles (qq-plot) suggested normally distributed residuals. Homogeneity was affirmed by plotting fitted versus residual values. Model stability was assessed using DFFIT estimates and DFBETA in R. We found no evidence for influential cases on the fit or outcome of models, suggesting they were stable and robust.
Results
We captured 188 subalpine warblers in 2018 (107 males, 80 females, 1 unknown); 288 in 2019 (172 males, 114 females, 2 unknown); and 132 in 2022 (85 males, 46 females, 1 unknown). We resighted 176 color-ringed individuals (29% of the total) in the field. Birds that were never resighted were presumed to have flown away the night following first capture. There was no difference in body condition between birds that left the night following capture and those that made a stopover (2018, W = 3329, P = 0.567; 2019, W = 3345, P = 0.534; 2022, W = 1895, P = 0.544). Median fat and muscle scores were both 3, which indicates generally adequate energy stores upon arrival at Yasmina. Within birds that made a stopover and were resighted, average stopover duration was 10 days with a maximum of 27 days. There was no significant difference in stopover duration between sexes (t = − 0.852, df = 50.923, P = 0.398).
During their stopover, males often restricted their movements and settled on small home ranges composed of a single or a few trees (Fig. 2). We analyzed 97 home ranges of male subalpine warblers. Home ranges did not overlap with others within the same time period, and median functional home range area was 139 m2. Males often produced full songs or chips to broadcast their home range position, and aggressively defended their spaces by chasing and attacking intruders. Females rarely made such restricted settlements but rather behaved as floaters moving in and out of male-occupied areas (but see exception in Fig. 2 and discussion below). Female aggression was not apparent. Still, we analyzed home ranges of 27 females based on resights (see below).
Home ranges of Subalpine Warblers (Curruca iberiae) during a trans-Saharan stopover between March 15 and March 25, 2018 in Yasmina, Morocco. Each color demarks a color-ringed male’s home range across 10 days except the orange home range in the Southeastern area of the plot, which belonged to a female. White line segments show locations of nets
Simulated territory intrusion experiment
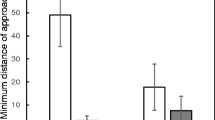
Male Subalpine Warblers responded aggressively to simulated territory intrusions. Males spent significantly more time within five meters of the speaker (χ2 = 32.091, df = 1, P < 0.001; Fig. 3a) approached closer to the speaker (χ2 = 19.407, df = 1, P < 0.001; Fig. 3b) and began chipping sooner (χ2 = 11.0217, df = 1, P < 0.001; Fig. 3c) in response to a playback of a conspecific song over a control playback of white noise. None of these responses significantly changed over the three days of experiments, relative to controls, nor was there evidence for interaction between treatment and days. Singing occurred during every simulated territory intrusion experiment (30 trial-days) and almost never during white noise trials (1 out of 21 trial days). Latency to begin singing did not change over three days (P = 0.584). Birds made obvious approaches to the speaker 29 out of 30 times and latency to approach the speaker significantly reduced over the three-day trials (χ2 = 32.717, df = 1, P < 0.001; Fig. 3d).
Results from simulated male-male territory intrusion experiments. Time spent within 5 m of the speaker (a), nearest approach to the speaker (b), latency to begin chipping (c), and latency to approach the speaker (d) in response to conspecific simulated territory intrusion playback (song; n = 10) versus a white noise playback (control; n = 7) across three consecutive days by Subalpine Warblers (Curruca iberiae) during a trans-Saharan stopover March–May 2019 in Yasmina, Morocco. Error bars show 95% confidence intervals
Relationships among home ranges, body condition, and fuel deposition rate
Some habitat features were related to initial body condition, but only for males. The best performing habitat model explained vegetation density of a home range by initial body condition of its occupant, and year of sampling (adj. r2 = 0.230, F3, 93 = 10.540, P < 0.001; Table 1; Fig. 4). Higher body condition was associated with home ranges of lower vegetation density (t = − 3.452, P < 0.001; Fig. 4). Total home range area (i.e., vegetated and exposed bare dirt) was negatively related to date of sampling and year, with home ranges earlier in the season being larger than later ones (adj. r2 = 0.144, F3, 93 = 6.389, P < 0.001). Models for vegetation height, home range roundness, distance to water, and exposed area were not supported by individual or stopover variables such as arrival date and stopover duration (P > 0.05).
Relationship between body condition (scaled mass index in grams) and home range vegetation density (percent cover) among years (2018, 2019, and 2022) of male Subalpine Warblers (Curruca iberiae) during a springtime trans-Saharan stopover in Yasmina, Morocco. The shaded areas show 95% confidence intervals
Median FDR ± SE for males that had home ranges and were recaptured was 0.058 ± 0.045 g per day (n = 56). All combinations and relevant interactions of independent variables failed to explain FDR in males (i.e., year, home range area, roundness, exposed area, vegetation density, vegetation height, distance to water, arrival date, stopover duration, age, and conspecific density). Additionally, male FDR was not explained by initial body condition (P = 0.221).
We ran an identical analysis for female body condition and FDR. Most notably to our questions, female body condition did not explain any habitat characteristics as it did in males (P > 0.05). Indeed, the only significant model predicted that older females selected denser vegetation than younger ones (adj. r2 = 0.358, F3, 23 = 5.837, P = 0.004). FDR was also unrelated to any measured habitat characteristics in females (P > 0.05). FDR was not significantly different between females and males (female median FDR ± SE = 0.072 ± 0.064; W = 476, P = 0.710).
Discussion
Our results show that male Subalpine Warblers make prolonged stopovers (> 7 days, sensu Alerstam 1991) on ephemeral but restricted home ranges, which are discretely used and not shared with male conspecifics. We also show that home ranges during stopover are reinforced by vocal broadcasts and physical defense with some evidence for increased defense over time. Therefore, we report that stopover territoriality is a common behavior expressed by male Subalpine Warblers in the midst of their spring trans-Saharan migration. To our knowledge, this is the first study of stopover territoriality where behavior, energetics, and habitat selection are closely examined together.
Behavioral responses to territory intrusion
Subalpine Warblers males spent more time in proximity to the intruder, approached more closely, and made swifter approaches during intrusions than in control experiments. Therefore, we confirm that males defend territories during stopover. Indeed, latency to approach the intruder was significantly shorter with each successive day. This partially supports that territorial investment, measured by urgency to respond, also increases with days at stopover. One explanation could be the “value asymmetry hypothesis,” where individuals that are already aware of a territory’s value are more willing to fight for it over intruders, which have limited to no knowledge about the contested area (Krebs 1982; Forstmeier 2002).
At the onset of territory intrusion (within 2 min) territory owners responded sharply by singing and chipping. This demonstrates the willingness to engage in territorial reinforcement via acoustic signaling. Males always sang in response to intruders, but latency to begin singing did not change across days. After loud vocalizations, a dive into the vegetation surrounding the speaker typically followed. The territory owner would then approach the speaker quietly in a stalking position with its tail raised.
We suggest that vocal broadcasts during stopover serve to deter territory intrusion, as they do in breeding contexts (Kroodsma and Byers 1991). However, once an intruder has infiltrated the territory, physical defense should be the primary mode of territorial mediation. Our playback experiment results clearly show that individuals invest in defending territories during stopover by increased vocal warnings and willingness to seek and approach the intruder. Territoriality is particularly interesting and unusual during spring migration, because there is no chance for reproduction, and still males invest time and energy towards aggressive defense of stopover territories. Therefore, we discuss the potential energetics of this behavior.
Territory selection by body condition
We found strong support that stopover habitat selection is related to body condition in males. While we did not directly score dominance patterns, we assume that body condition is positively related to dominance, as widely described for songbirds (Carrascal et al. 1998; Marra 2000; Marra and Holmes 2001; Jonart et al. 2007; Beltrão et al. 2021). Therefore, males in higher condition are likely more competitive at securing preferred territories at stopover, as they do at breeding and wintering sites (Carrascal et al. 1998; Marra 2000). Male Subalpine Warblers that arrived in higher body condition at stopover settled on home ranges with lower vegetation density. This is the opposite pattern expected if feeding were the primary motivation for habitat selection. Insect biomass is correlated with vegetation density in tamarisk habitats, and therefore migrating songbirds preferentially select trees with higher leaf density during stopover (Haigh 1996; McGrath et al. 2009). However, at their breeding sites in the Iberian Peninsula, Subalpine Warblers select low-density vegetation, especially relative to five other sympatric Curruca species (Cody and Walter 1976). This suggests that low vegetation density is a preferred habitat feature across this species’ migratory range. However, we acknowledge that the requirements between migration and breeding habitats may be very different. Home range size was negatively correlated with date in males. This may be related to a lower number of migrants early in the season, and thus lower density-dependent forces acting on individual movements. As more Subalpine Warbler challengers arrive and settle, movements became more restricted and home ranges became smaller. Conspecific density (estimated by net captures) was not significant in explaining home range characteristics. However, all captured birds are not inherently competitive. Net captures do not necessarily provide a direct index of competition as stopover and departure decisions are variable and may depend on the specific function for which a given stopover was made (reviewed in Schmaljohann et al. 2022). Rather, a better metric for “contender pressure” may come from scoring the aggression or dominance patterns of newcomers and relating this to changes in home range characteristics (Stamps 1990).
Female home ranges
Almost, no females used a restricted home range repeatedly but rather appeared to remain subordinate to the aggressive males. Here, they took on a floater strategy, whereby they sneaked into occupied territories (or were momentarily tolerated) to forage until they were expelled. Similar sex-specific strategies have been reported for hummingbirds during migration (Carpenter et al. 1993; Zenzal and Moore 2019). The one exception in our study was a female in the far Southeastern corner of the plot (orange polygons in Fig. 2). Perhaps, this individual was able to secure a restricted home range because of reduced traffic by males in this area. We also note that this exact area was occupied by a different female the following year. Of note, however, is that females had similar refueling rates, body conditions, and stopover durations as males despite drastically different behavioral strategies. This may reflect compensation by feeding at higher rates or reduced energy expenditure compared to actively territorial males (Dierschke et al. 2005). We suggest females would also benefit from home ranges, but male presence and aggression during this stage constrains establishments. Additionally, the potential for seasonal variation in inter-sexual behavioral dynamics at stopover should be examined.
Refueling benefits to stopover territoriality
Stopover territoriality as a strategy did not reveal obvious refueling benefits in male Subalpine Warblers. Territorial males had similar FDRs as non-territorial females. This supports that floating between feeding sites can yield similar FDRs as resource defense, and that such strategies may be sex-dependent (Carpenter et al. 1993; Zenzal and Moore 2016, 2019). As part of a similar study, color-ringed Common Redstarts (Phoenicurus phoenicurus) occasionally displayed conspecific aggression in Yasmina; however, they did not appear to settle on restricted home ranges but also had similar FDRs as Subalpine Warblers (AAA, unpublished data). Therefore, stopover territoriality is most likely not a ubiquitous strategy among species.
Other studies did find refueling benefits from stopover territoriality (e.g., Rappole and Warner1976; Bibby and Green 1980; Carpenter et al. 1993). Because we found no relationships between FDR and any home range characteristics, our data do not support the good feeding patch hypothesis (sensu Davies and Houston 1981). Vegetation and spatial characteristics of home ranges did not explain FDR or stopover duration. Similar results were found for a study on Reed Warblers (Acrocephalus scirpaceus) that defended stopover territories; yet,fueling, food abundance, and food distribution did not vary during stopover (Bibby and Green 1981). In the present study, the decision to make a stopover was independent of body condition. This is consistent with a previous study at Yasmina, where Subalpine Warblers had adequate body condition to continue their migration, yet still made stopovers (Arizaga et al. 2013). These patterns suggest that the function of stopover in this system is not primarily for fast refueling.
Subalpine Warblers may be employing an energy-minimizing migration strategy (sensu Alerstam and Lindström 1990; Maggini et al. 2015). This strategy predicts longer stopovers, slower refueling, slower migration speed, and prioritizes arrival at breeding sites in the best body condition rather than early arrival (as predicted for a time-minimizing strategy). Therefore, if indeed speed of migration is a secondary consideration, and migratory speed is correlated with FDR (Alerstam and Lindström 1990), then the rate of fuel deposition does not strongly affect behavioral strategies during stopover in the Subalpine Warbler.
In sum, male median FDR was only slightly above zero (0.058 g per day; 0.137% body mass per day), it was similar to females, and prolonged stopover duration or initial body condition did not explain refueling rates. These stopover patterns recall behavioral features of the breeding stage such as remaining sedentary, aggressive territorially, and frequent singing, as opposed to typical migratory stage patterns such as high mobility, cycles of hyperphagia, and rapid fuel accumulation (Ramenofsky and Wingfield 2006; Aamidor et al. 2011). Subalpine Warblers appear to be simply maintaining their already adequate body condition, rather than improving it or resuming migratory flights.
While food could be differentially distributed and correlated with vegetation biomass, we suggest it is probably not limiting at this stopover site regardless of variable vegetation density, and that it is ubiquitously available. Indeed, evenly distributed food sources should promote territoriality as a strategy in birds (Brown 1969; Rappole and Warner 1976; Chernetsov 2012; but see Ostfeld 1985 for the inverse relationship in rodents), especially at a compact stopover site, where space is limited but not necessarily food. Following this, we suggest that territoriality is not related to defense of food-rich patches for this species as reported for migratory hummingbirds (Carpenter et al. 1993; Zenzal and Moore 2019). Importantly, although we did not measure food sources, the lack of variation in fueling rates across vegetation densities supports this presumption. Also, supporting this is that pecking rates of Subalpine Warblers did not vary significantly across different zones at Yasmina (IM, unpublished data). This invites alternative explanations as to why males in the highest conditions (and presumably most dominant) would select lower vegetation density for their temporary territories.
Stopover territoriality for predation avoidance and social monitoring
Previous studies of wintering migratory birds have rejected the “good feeding patch” hypothesis but found support for an anti-predator function to territory selection (Cuadrado 1997). Territories that allowed for easier concealment from predators were selected over those that provided greater foraging opportunities (Cuadrado 1997; Arizaga et al. 2011). In contrast, Subalpine Warbler males with high body condition selected areas with lower vegetation density, an opposite pattern expected for purely anti-predator advantages (Dickman 1992). Furthermore, unlike wintering Robins, Subalpine Warbler males at stopover often sing in an exposed fashion, but they do regularly forage in the hidden recesses of the vegetation. However, while low vegetation density could increase detection from predators, it may simultaneously increase vigilance capacity against predators and competitors (Ebensperger and Hurtado 2005). Regardless, having intimate familiarity with a territory should inherently be safer because a territory owner would know the best places to seek refuge and the fastest routes to escape danger (Davies and Houston 1981; Stamps 1987; Cuadrado 1997).
In general appearance, predation on birds at Yasmina was low. Woodchat Shrikes (Lanius senator) are common quasi-predators in Yasmina, but only prey on migratory birds that are already in dying condition (Aispuro et al. 2019). Feral cats were uncommon, and snakes were never observed so we suggest home range selection is not strongly influenced by predators.
Alternatively, the selection of low-density vegetation could support a social monitoring strategy. Detailed familiarity of a home range’s structure, coupled with low visual obstruction, movement, and maneuverability due to the low vegetation density could provide for greater vigilance and defense capacity of intruders (Stamps 1987; Ebensperger and Hurtado 2005). Additionally, monitoring of neighbors may provide cues of potential danger, which may be enhanced in territories with low visual obstruction (Caine and Marra 1988; Elgar 1989). Therefore, selection of territories with low vegetation density may contain subtle benefits, but they remain to be examined at stopover. Indeed, birds in lower body condition may also benefit from their relegation to denser vegetation as it offers greater concealment from predators and dominant conspecific aggressors (Catry et al. 2004).
Stopover territoriality and non-energetic staging
It is likely that most birds recover during stopover (i.e., repair oxidative damage and immune functioning, sleep, balance water budget, and others reviewed in Schmaljohann et al. 2022). However, features of physiological recovery such as reduction in oxidative lipid damage as well as an increase in immune function have been shown to occur within two or three days of stopover (Skrip et al. 2015; Eikenaar et al. 2020a, b). Therefore, recovery does not fully explain the average 10-day stopover in our system.
While the specific functions of stopover territoriality are unclear, we suggest that the resource that males are defending is simply the space itself where basic ecological constraints (e.g., food limitation, predation, competition), can be minimized in a relatively controlled setting while maintaining an adequate body condition. After energetic demands relating to survival have been met, other important non-energetic benefits might be accrued.
For example, migratory birds face an additional challenge in that they must transition from a migratory phenotype into a breeding phenotype (Jacobs and Wingfield 2000; Ramenofsky and Wingfield 20062017). For some species, this transition likely occurs during spring migration, evidenced by studies where increasing testes size and hormonal upregulations are correlated with proximity to breeding sites (Bauchinger et al. 2007; Covino et al. 2017). We suggest that stopover, as a period of time and as a physical space, is the most likely interval and location for life history transitions to occur. If so, the platform for this transition is in itself a valuable resource worthy of territorial defense.
While we did not investigate this theme directly, it is possible that stopover territoriality may in part be an indication of a life history transition, for example, an overlap between the termination of the migratory phenotype, and the development of the breeding phenotype (Jacobs and Wingfield 2000). For birds that migrate between the Sahel and Iberian Peninsula, the northern Sahara may be a strategic location for this transition to occur or accelerate, because the lengthiest and most arduous desert passage has already been crossed. Life history stage transitions may be gauged by monitoring hormone activity reflective of a life history continuum during stopover, such as testosterone in males (e.g., Covino et al. 2015, 2017). This is a prospective avenue to elucidate non-energetic functions of stopover territoriality and its influences or interactions with life history events.
The duration of territory occupancy and extent of defense during stopover probably vary according to species, sex, body condition, location, timing, and habitat suitability among other factors. We have thus far documented these variables for the Subalpine Warbler in the Sahara, and we propose that stopover behavioral strategies, such as territoriality, may be consequential in describing full-annual cycles in birds and integral to understanding their life histories.
Data availability
Data is archived in the Phaidra repository hosted by the University of Veterinary Medicine, Vienna, Austria. Available at: https://phaidra.vetmeduni.ac.at/o:1205.
References
Aamidor SE, Bauchinger U, Mizrahy O, McWilliams SR, Pinshow B (2011) During stopover, migrating blackcaps adjust behavior and intake of food depending on the content of protein in their diets. Integr Comp Biol 51:385–393. https://doi.org/10.1093/icb/icr054
Aborn DA, Moore FR (1997) Pattern of movement by summer tanagers (Piranga rubra) during migratory stopover: a telemetry study. Behaviour 134:1077–1100. https://doi.org/10.1163/156853997X00412
Aispuro AA, Machowetz C, Illa M, Gargallo G, Maggini I (2019) Woodchat shrike Lanius senator predation on migrating songbirds during a trans-Saharan stopover. Ardeola 67:137–143. https://doi.org/10.13157/arla.67.1.2020.sc4
Alerstam T (1991) Bird flight and optimal migration. Trends Ecol Evol 6:210–215. https://doi.org/10.1016/0169-5347(91)90024-R
Alerstam T, Lindström Å (1990) Optimal bird migration: the relative importance of time, energy, and safety. In: Gwinner E (ed) Bird migration: physiology and ecophysiology. Springer, Berlin Heidelberg, New York, pp 331–351. https://doi.org/10.1007/978-3-642-74542-3_22
Arizaga J, Maggini I, Hama F, Crespo A, Gargallo G (2013) Site and species-specific fuel load of European-Afrotropical passerines on arrival at three oases of southeast Morocco during spring migration. Bird Study 60:11–21. https://doi.org/10.1080/00063657.2012.735222
Arizaga J, Schmaljohann H, Bairlein F (2011) Stopover behaviour and dominance: a case study of the Northern Wheatear Oenanthe. Ardea 99:157–165. https://doi.org/10.5253/078.099.0205
Audacity Team (2020) Audacity(R): free audio editor and recorder, version 2.4.2. https://audacityteam.org/. Accessed 10 Aug 2020
Bairlein F (1995) Manual of field methods. European-African songbird migration network. Institut für Vogelforschung, Willhelmshaven, Germany
Barton K (2010) MuMIn: multi-model inference. R package version 0.13.17, http://CRAN.R-project.org/package=MuMIn
Bauchinger U, Van’t Hof T, Biebach H (2007) Testicular development during long-distance spring migration. Horm Behav 51:295–305. https://doi.org/10.1016/j.yhbeh.2006.10.010
Beltrão P, Godinho C, Lourenço R, Pereira PF (2021) Behavioural repeatability in Sardinian warblers (Sylvia melanocephala): larger individuals are more aggressive. Acta Ethol 24:131–140. https://doi.org/10.1007/s10211-020-00358-3
Bibby CJ, Green RE (1980) Foraging behaviour of migrant pied flycatchers, Ficedula hypoleuca, on temporary territories. J Animal Ecol 49:507–521. https://doi.org/10.2307/4260
Bibby CJ, Green RE (1981) Autumn migration strategies of Reed and Sedge Warblers. Ornis Scand 12:1–12. https://doi.org/10.2307/3675898
BirdLife International (2021) Species factsheet: Sylvia cantillans, http://www.birdlife.org on 20/08/2021
Brown JL (1969) Territorial behavior and population regulation in birds: a review and re-evaluation. Wilson Bull 81:293–329
Burnham KP, Anderson DR (2002) Model selection and multimodel inference. A practical information-theoretic approach. Springer, Berlin. https://doi.org/10.1007/b97636
Caine NG, Marra SL (1988) Vigilance and social organization in two species of primates. Anim Behav 36:897–904. https://doi.org/10.1016/S0003-3472(88)80172-6
Carpenter FL, Hixon MA, Russell RW, Paton DC, Temeles EJ (1993) Interference asymmetries among age-sex classes of rufous hummingbirds during migratory stopovers. Behav Ecol Sociobiol 33:297–304. https://doi.org/10.1007/BF00172927
Carrascal LM, Senar JC, Mozetich I, Uribe F, Domenech J (1998) Interactions among environmental stress, body condition, nutritional status, and dominance in great tits. Auk 115:727–738. https://doi.org/10.2307/4089420
Catry P, Campos AR, Almada VC, Cresswell W (2004) Winter segregation of migrant European robins Erithacus rubecula in relation to sex, age and size. J Avian Biol 35:204–209. https://doi.org/10.1111/j.0908-8857.2004.03266.x
Chernetsov N (2012) Passerine migration: stopovers and flight. Springer, Berlin. https://doi.org/10.1007/978-3-642-29020-6
Chernetsov N, Mukhin A (2006) Spatial behavior of European Robins during migratory stopovers: a telemetry study. Wilson J Ornithol 118:364–373. https://doi.org/10.1676/04-134.1
Cody ML, Walter H (1976) Habitat selection and interspecific interactions among Mediterranean sylviid warblers. Oikos 27:210–238. https://doi.org/10.2307/3543900
Cohen EB, Barrow WC Jr, Buler JJ et al (2017) How do en route events around the Gulf of Mexico influence migratory landbird populations? Condor 119:327–343. https://doi.org/10.1650/CONDOR-17-20.1
Cohen EB, Satterfield DA (2020) ‘Chancing on a spectacle:’co-occurring animal migrations and interspecific interactions. Ecography 43:1657–1671. https://doi.org/10.1111/ecog.04958
Covino KM, Jawor JM, Kelly JF, Moore FR (2017) Overlapping life-history stages in migrating songbirds: variation in circulating testosterone and testosterone production capacity. J Ornithol 158:203–212. https://doi.org/10.1007/s10336-016-1385-8
Covino KM, Morris SR, Moore FR (2015) Patterns of testosterone in three Nearctic-Neotropical migratory songbirds during spring passage. Gen Comp Endocrinol 224:186–193. https://doi.org/10.1016/j.ygcen.2015.08.012
Cuadrado M (1997) Why are migrant Robins (Erithacus rubecula) territorial in winter?: the importance of the anti-predatory behaviour. Ethol Ecol Evol 9:77–88. https://doi.org/10.1080/08927014.1997.9522904
Davies NB, Houston AI (1981) Owners and satellites the economics of territory defense in the Pied Wagtails Motacilla alba. J Anim Ecol 50:157–180. https://doi.org/10.2307/4038
Daubenmire RF (1959) Canopy coverage method of vegetation analysis. Northwest Sci 33:39–64
Dickman CR (1992) Predation and habitat shift in the house mouse, Mus domesticus. Ecology 73:313–322. https://doi.org/10.2307/1938742
Dierschke V, Mendel B, Schmaljohann H (2005) Differential timing of spring migration in northern wheatears Oenanthe: hurried males or weak females? Behav Ecol and Sociobiol 57:470–480. https://doi.org/10.1007/s00265-004-0872-8
Ebensperger LA, Hurtado MJ (2005) On the relationship between herbaceous cover and vigilance activity of degus (Octodon degus). Ethology 111:593–608. https://doi.org/10.1111/j.1439-0310.2005.01084.x
Eikenaar C, Hessler S, Hegemann A (2020) Migrating birds rapidly increase constitutive immune function during stopover. R Soc Open Sci 7:192031. https://doi.org/10.1098/rsos.192031
Eikenaar C, Winslott E, Hessler S, Isaksson C (2020b) Oxidative damage to lipids is rapidly reduced during migratory stopovers. Funct Ecol 34:1215–1222. https://doi.org/10.1111/1365-2435.13540
Elgar MA (1989) Predator vigilance and group size in mammals and birds: a critical review of the empirical evidence. Biol Rev 64:13–33. https://doi.org/10.1111/j.1469-185X.1989.tb00636.x
Emlen JT (1957) Defended area? A critique of the territory concept and of conventional thinking. Ibis 99:352
Forstmeier W (2002) Benefits of early arrival at breeding grounds vary between males. J Anim Ecol 71:1–9. https://doi.org/10.1046/j.0021-8790.2001.00569.x
Haigh SL (1996) Avian habitat use in southern Nevada riparian areas with varying amounts of Tamarix ramosissima. PhD thesis, University of Nevada. https://doi.org/10.25669/vsqs-jevv
Jacobs JD, Wingfield JC (2000) Endocrine control of life-cycle stages: a constraint on response to the environment? Condor 102:35–51. https://doi.org/10.1093/condor/102.1.35
Jonart LM, Hill GE, Badyaev AV (2007) Fighting ability and motivation: determinants of dominance and contest strategies in females of a passerine bird. Anim Behav 74:1675–1681. https://doi.org/10.1016/j.anbehav.2007.03.012
Kodric-Brown A, Brown JH (1978) Influence of economics, interspecific competition, and sexual dimorphism on territoriality of migrant rufous hummingbirds. Ecology 59:285–296. https://doi.org/10.2307/1936374
Krebs JR (1982) Territorial defence in the great tit (Parus major): do residents always win? Behav Ecol Sociobiol 11:185–194. https://doi.org/10.1007/BF00300061
Kroodsma DE, Byers BE (1991) The function(s) of bird song. Am Zool 31:318–328. https://doi.org/10.1093/icb/31.2.318
Maggini I, Hama F, Robson D, RguibiIdrissi H, Bairlein F, Gargallo G (2015) Foraging behavior of three species of songbirds during stopover in southeastern Morocco during spring migration. J Field Ornithol 86:266–276. https://doi.org/10.1111/jofo.12115
Marra PP (2000) The role of behavioral dominance in structuring patterns of habitat occupancy in a migrant bird during the nonbreeding season. Behav Ecol 11:299–308. https://doi.org/10.1093/beheco/11.3.299
Marra PP, Holmes RT (2001) Consequences of dominance-mediated habitat segregation in American Redstarts during the nonbreeding season. Auk 118:92–104. https://doi.org/10.1093/auk/118.1.92
McGrath LJ, Van Riper CIII, Fontaine JJ (2009) Flower power: tree flowering phenology as a settlement cue for migrating birds. J Anim Ecol 78:22–30. https://doi.org/10.1111/j.1365-2656.2008.01464.x
McWilliams SR, Guglielmo C, Pierce B, Klaassen M (2004) Flying, fasting, and feeding in birds during migration: a nutritional and physiological ecology perspective. J Avian Biol 35:377–393. https://doi.org/10.1111/j.0908-8857.2004.03378.x
Moore FR, Gauthreaux S Jr, Kerlinger P, Simons T, Martin T, Finch D (1995) Habitat requirements during migration: important link in conservation. In: Martin T, Finch D (eds) Ecology and management of neotropical migratory birds, a synthesis and review of critical issues. Oxford University Press, New York, pp 121–144
Morel GJ, Morel MY (1992) Habitat use by Palaearctic migrant passerine birds in West Africa. Ibis 134:83–88. https://doi.org/10.1111/j.1474-919X.1992.tb04737.x
Mostafa DAA, Willemoes M, Salewski V, Ortvad TE, Dabelsteen T, Thorup K (2021) Contrasting use of space by two migratory Afro-Palearctic warblers on their African non-breeding grounds. J Ornithol 162:813–821. https://doi.org/10.1007/s10336-021-01881-1
Myers JP, Connors PG, Pitelka FA (1979) Territory size in wintering sanderlings: the effects of prey abundance and intruder density. Auk 96:551–561. https://doi.org/10.1093/auk/96.3.551
Németh Z, Moore FR (2014) Information acquisition during migration: a social perspective. Auk 131:186–194. https://doi.org/10.1642/AUK-13-195.1
Noble GK (1939) The role of dominance in the social life of birds. Auk 56:263–273. https://doi.org/10.2307/4079047
Ostfeld RS (1985) Limiting resources and territoriality in microtine rodents. Am Nat 126:1–15. https://doi.org/10.1086/284391
Peig J, Green AJ (2009) New perspectives for estimating body condition from mass/length data: the scaled mass index as an alternative method. Oikos 118:1883–1891. https://doi.org/10.1111/j.1600-0706.2009.17643.x
Peig J, Green AJ (2010) The paradigm of body condition: a critical reappraisal of current methods based on mass and length. Funct Ecol 24:1323–1332. https://doi.org/10.1111/j.1365-2435.2010.01751.x
Pons P, Bas JM, Prodon R, Roura-Pascual N, Clavero M (2008) Territory characteristics and coexistence with heterospecifics in the Dartford warbler Sylvia undata across a habitat gradient. Behav Ecol Sociobiol 62:1217–1228. https://doi.org/10.1007/s00265-008-0550-3
QGIS Development Team (2018) QGIS geographic information system. https://qgis.org. Accessed 24 Sept 2020
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 1 Jan 2020
Ramenofsky M, Wingfield JC (2006) Behavioral and physiological conflicts in migrants: the transition between migration and breeding. J Ornithol 147:135–145. https://doi.org/10.1007/s10336-005-0050-4
Ramenofsky M, Wingfield JC (2017) Regulation of complex behavioural transitions: migration to breeding. Anim Behav 124:299–306. https://doi.org/10.1016/j.anbehav.2016.09.015
Rappole JH, Warner DW (1976) Relationships between behavior, physiology and weather in avian transients at a migration stopover site. Oecologia 26:193–212. https://doi.org/10.1007/BF00345289
Robel RJ, Briggs JN, Dayton AD, Hulbert LC (1970) Relationships between visual obstruction measurements and weight of grassland vegetation. Rangeland Ecol Manag 23:295–297. https://doi.org/10.2307/3896225
Salewski V, Almasi B, Heuman A, Thoma M, Schlageter A (2007) Agonistic behaviour of Palaearctic passerine migrants at a stopover site suggests interference competition. Ostrich 78:349–355. https://doi.org/10.2989/OSTRICH.2007.78.2.37.117
Sapir N, Tsurim I, Gal B, Abramsky Z (2004) The effect of water availability on fuel deposition of two staging Sylvia warblers. J Avian Biol 35:25–32. https://doi.org/10.1111/j.0908-8857.2004.03212.x
Schaub M, Jenni L (2000) Fuel deposition of three passerine bird species along the migration route. Oecologia 122:306–317. https://doi.org/10.1007/s004420050036
Schmaljohann H, Eikenaar C, Sapir N (2022) Understanding the ecological and evolutionary function of stopover in migrating birds. Biol Rev 97:1231–1252. https://doi.org/10.1111/brv.12839
Shirihai H, Svensson L (2018) Handbook of Western Palearctic Birds, Volume 1: Passerines: Larks to Warblers. Bloomsbury Publishing, London
Skrip MM, Bauchinger U, Goymann W, Fusani L, Cardinal M, Alan RR, McWilliams SR (2015) Migrating songbirds on stopover prepare for, and recover from, oxidative challenges posed by long-distance flight. Ecol Evol 5:3198–3209. https://doi.org/10.1002/ece3.1601
Snow BK, Snow DW (1984) Long-term defence of fruit by Mistle Thrushes Turdus viscivorus. Ibis 126:39–49. https://doi.org/10.1111/j.1474-919X.1984.tb03662.x
Stamps JA (1987) The effect of familiarity with a neighborhood on territory acquisition. Behav Ecol Sociobiol 21:273–277. https://doi.org/10.1007/BF00299964
Stamps JA (1990) The effect of contender pressure on territory size and overlap in seasonally territorial species. Am Nat 135:614–632 (https://www.jstor.org/stable/2462026)
Svensson L (2013) A taxonomic revision of the Subalpine Warbler Sylvia cantillans. Bull Br Ornithol Club 113:240–248
Yeo IK, Johnson RA (2000) A new family of power transformations to improve normality or symmetry. Biometrika 87:954–959. https://doi.org/10.1093/biomet/87.4.954
Zenzal TJ, Moore FR (2016) Stopover biology of Ruby-throated Hummingbirds (Archilochus colubris) during autumn migration. Auk 133:237–250. https://doi.org/10.1642/AUK-15-160.1
Zenzal TJ, Moore FR (2019) Resource use and defence by ruby-throated hummingbirds during stopover. Behaviour 156:131–153. https://doi.org/10.1163/1568539X-00003533
Zuccon D, Pons JM, Boano G et al (2020) Type specimens matter: new insights on the systematics, taxonomy and nomenclature of the subalpine warbler (Sylvia cantillans) complex. Zool J Linn Soc -Lond 190:314–341. https://doi.org/10.1093/zoolinnean/zlz169
Acknowledgements
We thank Clara Machowetz, Michele Pes, Christina Ninou, Barbara Kofler, Raül Aymí and Georg Gruber, along with many volunteers for assistance in the field. We thank Gabriel Gargallo, Abdeljebbar Qninba, Hamid Rguibi Idrissi, Aziz Chandi, Youssef Kraoui, and the staff at Hotel Kasbah Yasmina for planning and logistical support. We thank Wolfgang Wiltschko, Heiko Schmaljohann and an anonymous reviewer for their constructive reviews and discourse regarding this manuscript.
Funding
Open access funding provided by University of Veterinary Medicine Vienna This work was supported by the Austrian Academy of Sciences DOC fellowship 25133 granted to AAA. Also supported by start-up grants from the University of Veterinary Medicine, Vienna to AAA, IM and LF, as well as start-up funds to LF from the University of Vienna.
Author information
Authors and Affiliations
Contributions
AAA, IM, LF and VC conceived the ideas and designed methodology; AAA and MI collected the data; AAA analyzed the data and led the writing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Ethics approval
All research activities for this study were approved and permitted by the Moroccan Ministry of Agriculture and the High Commissioner of Water (reference nr. 18/2017; 06/2019; 10/2022). We adhered to all applicable international, national, and institutional guidelines for the use of animals in this study.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by W. Wiltschko.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aispuro, A.A., Canoine, V., Illa, M. et al. Stopover territoriality in songbirds crossing the Sahara: aggression and energetics during migration. Behav Ecol Sociobiol 77, 57 (2023). https://doi.org/10.1007/s00265-023-03336-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-023-03336-4