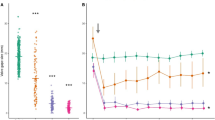
Abstract
Chemical alarm cues alert aquatic prey to the presence of an actively foraging predator. There is a large literature based upon responses to alarm cues derived from skin extract because it is anticipated that prey skin is damaged when prey are attacked by a predator. However, many predators feed by suction feeding whereby prey are quickly drawn into the buccal cavity and swallowed whole with little, if any, direct contact between the teeth of the predator and the skin of the prey. Here, we test if predation by suction feeding releases chemical information in sufficient quantity to elicit an antipredator response in conspecific prey. In tests of individual zebrafish Danio rerio, we found that odor of crushed zebrafish produced a clear antipredator behavioral response, but water collected immediately adjacent to staged predation events between a largemouth bass Micropterus salmoides (122–145 mm TL) and adult zebrafish (39 mm TL) did not elicit alarm behavior and did not differ from behavioral responses to blank water or bass odor (on a diet of earthworms). In a second experiment, zebrafish were swallowed by largemouth bass and then retrieved seconds later through gastric lavage, which produced zebrafish that were alive and completely intact with minimal epidermal damage. Published relationships between bass length, gape size, and the geometry of suction feeding suggest that in a hypothetical population of largemouth bass feeding on adult zebrafish or fathead minnows, the majority of predation events by piscivorous fish probably would not release detectable levels of prey alarm cue. Accounting for the role of feeding mechanics by fish predators requires a recalibration of the literature on risk assessment by small prey fishes. Chemically mediated antipredator behaviors against suction-feeding predators may occur primarily via post-ingestion dietary cues or disturbance cues released near the moment of attack.
Significance statement
Skin extract is commonly used to simulate predation and experimentally manipulate the perception of predation risk because it is generally assumed that epidermal tissue is damaged during predation. The resulting chemicals released by damaged skin induce nearby prey to engage in antipredator behavior. This study attempted to demonstrate behavioral responses to alarm cue released during staged predation events but was unable to do so, likely because of the mode of ingestion by the predator—suction feeding. Prey retrieved by gastric lavage from the gut of predators immediately after ingestion confirmed that the epidermis is largely intact when prey are swallowed by suction feeding. Because most fish predators employ suction feeding, this study calls for a recalibration of how chemical cues mediate predator–prey interactions in aquatic ecosystems. Release of alarm cues by fish predation cannot be assumed to occur in most cases. Chemical labeling by dietary cues or disturbance cues likely plays a larger role than previously appreciated for acquired predator recognition.


taken from Fernando et al. (2018) and data sources reported therein (Lawrence 1958; Shireman 1978; Johnson and Post 1996; Timmerman et al. 2000; Hill et al. 2004), data for individual models shown in grey lines. The bold black line is the average of these models, defined by gape (mm) = 0.0001 TL2 + 0.0868 TL – 0.1083, R2 = 0.9998. Ingested volume of water (IVW) for suction feeding was calculated from the equation reported by Kane and Higham (2014): height = 0.670 + (0.018 × ram speed) + (1.311 × gape), using a ram speed of 0. Because the dimensions of the envelope of ingested water are greater than the gape, we used gape size to calculate minimize length of bass capable of swallowing whole prey such as zebrafish and fathead minnows
Similar content being viewed by others
Data availability
Data are available at https://doi.org/10.5061/dryad.51c59zw8z.
Abbreviations
- AC:
-
Alarm cue derived from crushed zebrafish
- BAC:
-
Alarm cue collected from water surrounding a bass that had eaten a zebrafish
- BEW:
-
Tank water from bass fed a diet of earthworms
- DH2O:
-
Distilled water
- Pre VD:
-
Pres-stimulus vertical distribution
- Post VD:
-
Post-stimulus vertical distribution
- Change in VD:
-
Post-pre vertical distribution
- Pre activity:
-
Pre-stimulus number of lines crossed
- Post activity:
-
Post-stimulus number of lines crossed
- Change in activity:
-
Post-pre activity
- Pre SH:
-
Pre-stimulus time in shelter
- Post SH:
-
Post-stimulus time in shelter
- Change in SH:
-
Post-pre shelter use
- Bass TL:
-
Total length (mm) of largemouth bass, mouth gape-at-length data from regression equations from the various sources shown as column headings and cited in full in the manuscript (Each individual data set was plotted, and we also averaged these together to produce an overall gape-length relationship plotted in Fig. 2 as a bold line and the height of the ingested volume of water (IVW Ht))
References
Bairos-Novak KR, Ferrari MCO, Chivers DP (2019) A novel alarm signal in aquatic prey: familiar minnows coordinate group defences against predators through chemical disturbance cues. J Anim Ecol 88:1281–1290. https://doi.org/10.1111/1365-2656.12986
Brown GE, Chivers DP, Smith RJF (1995) Fathead minnows avoid conspecific and heterospecific alarm pheromones in the faeces of northern pike. J Fish Biol 47:387–393. https://doi.org/10.1111/j.1095-8649.1995.tb01908.x
Brown GE, Chivers DP, Smith RJF (1996) Effects of diet on localized defecation by northern pike, Esox Lucius. J Chem Ecol 22:467–475. https://doi.org/10.1007/BF02033649
Brown GE, Adrian JC, Shih ML (2001) Behavioural responses of fathead minnows to hypoxanthine-3-N-oxide at varying concentrations. J Fish Biol 58:1465–1470. https://doi.org/10.1111/j.1095-8649.2001.tb02301.x
Chivers DP, Mirza RS (2001) Predator diet cues and the assessment of predation risk by aquatic vertebrates: a review and prospectus. In: Marchlewska-Koj A, Lepri JJ, Müller-Schwarze D (eds) Chemical Signals in Vertebrates, vol 9. Plenum Press, New York, pp 277–284
Chivers DP, Smith RJF (1994) Fathead minows, Pimephales promelas, acquire predator recognition when alarm substance is associated with the sight of unfamiliar fish. Anim Behav 48:597–601. https://doi.org/10.1006/anbe.1994.1279
Chivers DP, Smith RJF (1998) Chemical alarm signalling in aquatic predator-prey systems: a review and prospectus. Écoscience 5:338–352. https://doi.org/10.1080/11956860.1998.11682471
Chivers DP, Brown GE, Smith RJF (1996) The evolution of chemical alarm signals: attracting predators benefits alarm signal senders. Am Nat 148:649–659. https://doi.org/10.1086/285945
Chivers DP, Wisenden BD, Hindman CJ et al (2007) Epidermal ‘alarm substance’ cells of fishes are maintained by non-alarm functions: possible defence against pathogens, parasites and UVB radiation. Proc R Soc Lond B 274:2611–2619. https://doi.org/10.1098/rspb.2007.0709
Faulkner AE, Holstrom IE, Molitor SA, Hanson ME, Shegrud WR, Gillen JC, Willard SJ, Wisenden BD (2017) Field verification of chondroitin sulfate as a putative component of chemical alarm cue in wild populations of fathead minnows (Pimephales promelas). Chemoecology 27:233–238. https://doi.org/10.1007/s00049-017-0247-z
Fernando AV, Hecke KB, Eggleton MA (2018) Length, body depth, and gape relationships and inference on piscivory among common North American centrarchids. Southwest Nat 17:309–326. https://doi.org/10.1656/058.017.0214
Ferrari MCO, Wisenden BD, Chivers DP (2010) Chemical ecology of predator-prey interactions in aquatic ecosystems: a review and prospectus. Can J Zool 88:698–724. https://doi.org/10.1139/Z10-029
Gelowitz CM, Mathis A, Smith RJF (1993) Chemosensory recognition of northern pike (Esox lucius) by brook stickleback (Culaea inconstans): population differences and the influence of predator diet. Behaviour 127:105–118. http://www.jstor.org/stable/4535145
Gidmark NJ, Pos K, Matheson B, Ponce E, Westneat MW (2019) Functional morphology and biomechanics of feeding in fishes. In: Bels V, Whishaw IQ (eds) Feeding in Vertebrates. Fascinating Life Sciences. Springer Nature, Cham, pp 297–332. https://doi.org/10.1007/978-3-030-13739-7_9
Goldman JA, Désormeaux IS, Brown GE (2020) Disturbance cues as a source of risk assessment information under natural conditions. Freshw Biol 65:981–986. https://doi.org/10.1111/fwb.13484
Hakala JP, Johnson FD (2004) Evaluation of a gastric lavage method for use on largemouth bass. N Am J Fish Manage 24:1398–1403. https://doi.org/10.1577/M03-092.1
Hall D, Suboski MD (1995) Visual and olfactory stimuli in learned release of alarm reactions by zebra danio fish (Brachydanio rerio). Neuro Learn Mem 63:229–240. https://doi.org/10.1006/nlme.1995.1027
Hazlett BA (1985) Disturbance pheromones in crayfish Orconectes virilis. J Chem Ecol 11:1695–1711. https://doi.org/10.1007/BF01012121
Hazlett BA (1990) Source and nature of disturbance-chemical system in crayfish. J Chem Ecol 16:2263–2275. https://doi.org/10.1007/BF01026936
Higham TE, Day SW, Wainwright PC (2006) Multidimensional analysis of suction feeding performance in fishes: fluid speed, acceleration, strike accuracy and the ingested volume of water. J Exp Biol 209:2713–2725. https://doi.org/10.1242/jeb.02315
Hill JE, Nico LG, Cichra CE, Gilbert CR (2004) Prey vulnerability to peacock cichlids and largemouth bass based on predator gape and prey body-depth. Proc Southeast Assoc Fish Wildl Agencies 58:47–56
Hume JB, Wagner M (2018) A death in the family: sea lamprey (Petromyzon marinus) avoidance of confamilial alarm cues diminishes with phylogenetic distance. Ecol Evol 8:3751–3762. https://doi.org/10.1002/ece3.3930
Johnson JM, Post DM (1996) Morphological constraints on intracohort cannibalism in age-0 largemouth bass. Trans Am Fish Soc 125:809–812. https://doi.org/10.1577/1548-8659(1996)125%3c0809:MCOICI%3e2.3.CO;2
Kane EA, Higham TE (2014) Modelled three-dimensional suction accuracy predicts capture success in three species of centrarchid fishes. J R Soc Interface 11:20140223. https://doi.org/10.1098/rsif.2014.0223
Kasumyan AO, Ponomarev VY (1987) Biochemical features of alarm pheromone in fish of the order Cypriniformes. J Evol Biochem Phys 23:20–24
Lawrence JM (1958) Estimated sizes of various forage fishes largemouth bass can swallow. Proc Southeast Assoc Fish Wildl Agencies 11:220–225
Lawrence BJ, Smith RJF (1989) Behavioral response of solitary fathead minnows, Pimephales promelas, to alarm substance. J Chem Ecol 15:209–219. https://doi.org/10.1007/BF02027783
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Magurran AE (1989) Acquired recognition of predator odour in the European minnow (Phoxinus phoxinus). Ethology 82:216–223. https://doi.org/10.1111/j.1439-0310.1989.tb00501.x
Manek AK, Ferrari MCO, Pollock RJ, Vicente D, Weber LP, Chivers DP (2013) Within and between population variation in epidermal club cell investment in a freshwater prey fish: a cautionary tale for evolutionary ecologists. PLoS ONE 8:e56689. https://doi.org/10.1371/journal.pone.0056689
Mathis A, Smith RJF (1993a) Fathead minnows, Pimephales promelas, learn to recognize northern pike, Esox lucius, as predators on the basis of chemical stimuli from minnows in the pike’s diet. Anim Behav 46:645–656. https://doi.org/10.1006/anbe.1993.1241
Mathis A, Smith RJF (1993b) Chemical labeling of northern pike (Esox lucius) by the alarm pheromone of fathead minnows (Pimephales promelas). J Chem Ecol 19:1967–1979. https://doi.org/10.1007/BF00983800
Mathuru AS, Kibat C, Cheong WF, Shui G, Wenk MR, Friedrich RW, Jesuthasan S (2012) Chondroitin fragments are odorants that trigger fear behavior in fish. Curr Biol 22:538–544. https://doi.org/10.1016/j.cub.2012.01.061
Mirza RS, Chivers DP (2001) Learned recognition of heterospecific alarm signals: the importance of a mixed predator diet. Ethology 107:1007–1018. https://doi.org/10.1046/j.1439-0310.2001.00728.x
Mirza RS, Chivers DP (2002) Behavioural responses to conspecific disturbance chemicals enhance survival of juvenile brook charr, Salvelinus fontinalis, during encounters with predators. Behaviour 139:1099–1110. https://doi.org/10.1163/15685390260437272
Mirza RS, Chivers DP (2003a) Fathead minnows learn to recognize heterospecific alarm cues they detect in the diet of a known predator. Behaviour 140:1359–1370
Mirza RS, Chivers DP (2003b) Predator diet cues and the assessment of predation risk by juvenile brook charr: do diet cues enhance survival? Can J Zool 81:126–132. https://doi.org/10.1139/z02-225
Pandey S, Stockwell CA, Snider MR, Wisenden BD (2021) Epidermal club cells in fishes: a case for ecoimmunological analysis? Int J Mol Sci 22:1440. https://www.mdpi.com/1422-0067/22/3/1440
Parra KV, Adrian JC Jr, Gerlai R (2009) The synthetic substance hypoxanthine 3-N-oxide elicits alarm reactions in zebrafish (Danio rerio). Behav Brain Res 205:336–341. https://doi.org/10.1016/j.bbr.2009.06.037
Pfeiffer W (1977) The distribution of fright reaction and alarm substance cells in fishes. Copeia 1977:653–665. https://doi.org/10.2307/1443164
Pfeiffer W, Riegekbauer G, Meier G, Scheibler B (1985) Effect of hypoxanthine-3(N)-oxide and hypoxanthine-1(N)-oxide on central nervous excitation of the black tetra Gymnocorymbus ternetzi (Characidae, Ostariophysi, Pisces) indicated by dorsal light response. J Chem Ecol 11:507–523. https://doi.org/10.1007/BF00989562
Schutz F (1956) Vergleichende Untersuchungen über die Schreckreaktion bei Fischen und deren Verbreitung. Z Vergl Physiol 38:84–135. https://doi.org/10.1007/BF00338623
Shireman JV, Colle DE, Rottman RW (1978) Size limits to predation on grass carp by largemouth bass. Trans Am Fish Soc 107:213–215. https://doi.org/10.1577/1548-8659(1978)107%3c213:SLTPOG%3e2.0.CO;2
Smith RJF (1992) Alarm signals in fishes. Rev Fish Biol Fisher 2:33–63
Sutrisno R, Schotte PM, Wisenden BD (2014) Chemical arms race between predator and prey: a test of predator digestive counter-measures against chemical labeling by dietary cues of prey. J Freshw Ecol 29:17–23. https://doi.org/10.1080/02705060.2013.804886
Timmerman CM, Annett CA, Bailey CF (2000) Determination of factors limiting prey-size swallowed by larval and small juvenile largemouth bass. Trans Am Fish Soc 129:618–622. https://doi.org/10.1577/1548-8659(2000)129%3c0618:DOFLPS%3e2.0.CO;2
Vavrek MA, Elvidge CK, DeCaire R, Belland B, Jackson CD, Brown GE (2008) Disturbance cues in freshwater prey fishes: do juvenile convict cichlids and rainbow trout respond to ammonium as an ‘early warning’ signal? Chemoecology 18:255–261. https://doi.org/10.1007/s00049-008-0412-5
von Frisch K (1942) Über einen Schreckstoff der Fischhaut und seine biologische Bedeutung. Z Vergl Physiol 29:46–145
Wainwright P (1996) Ecological explanation through functional morphology: the feeding biology of sunfishes. Ecology 77:1336–1343. https://doi.org/10.2307/2265531
Wainwright P (2005) Functional morphology of the pharyngeal jaw apparatus. Fish Physiol 23:77–101. https://doi.org/10.1016/S1546-5098(05)23003-0
Wainwright P, Carroll AM, Collar DC, Day SW, Higham TE, Holzman RA (2007) Suction feeding mechanics, performance, and diversity in fishes. Integr Comp Biol 47:96–106. https://doi.org/10.1093/icb/icm032
Waldman B (1982) Quantitative and developmental analyses of the alarm reaction in the zebra danio, Brachydanio rerio. Copeia 1982:1–9. https://doi.org/10.2307/1444261
Wisenden BD (2000) Olfactory assessment of predation risk. Phil Trans R Soc B 355:1205–1208. https://doi.org/10.1098/rstb.2000.0668
Wisenden BD (2008) Active space of chemical alarm cue in natural fish populations. Behaviour 145:391–407. https://doi.org/10.1163/156853908783402920
Wisenden BD (2011) Quantifying anti-predator response to chemical alarm cues. In: Kalueff AV, Hart P, LaPorte J (eds) Zebrafish Behavioral Protocols. Humana Press, New York, Springer Science, pp 49–60
Wisenden BD (2015) Chemical cues that indicate risk of predation. In: Sorensen PW, Wisenden BD (eds) Fish pheromones and related cues. Wiley-Blackwell Press, Ames, pp 131–148
Wisenden BD, Chivers DP (2006) The role of public chemical information in antipredator behaviour. In: Ladich F, Collins SP, Moller P, Kapoor BG (eds) Fish Communication. Science Publisher, NH, pp 259–278
Wisenden BD, Smith RJF (1997) The effect of physical condition and shoal-mate familiarity on proliferation of alarm substance cells in the epidermis of fathead minnows. J Fish Biol 50:799–808. https://doi.org/10.1111/j.1095-8649.1997.tb01973.x
Wisenden BD, Chivers DP, Smith RJF (1995) Early warning in the predation sequence: a disturbance pheromone in Iowa darters (Etheostoma exile). J Chem Ecol 21:1469–1480. https://doi.org/10.1007/BF02035146
Acknowledgements
We are grateful to two anonymous reviewers for constructive suggestions for improving the manuscript.
Funding
Funding for this study was provided by a grant from the Judy Strong Undergraduate Research fund awarded to A.A.T. and J.D.U.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Experimental protocols used in this study were reviewed and approved by the Minnesota State University Moorhead Institutional Animal Care and Use Committee under protocol number 19-R/T-BIO-018-N-Y-C. The use of animals adheres to the guidelines set forth by the Animal Behavior Society/Association for the Study of Animal Behaviour.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by I. Hamilton
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wisenden, B.D., Taylor, A.A., Undem, J.D. et al. Moby-bass: suction feeding by predators limits direct release of alarm cues in fishes. Behav Ecol Sociobiol 76, 33 (2022). https://doi.org/10.1007/s00265-022-03146-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03146-0