Abstract
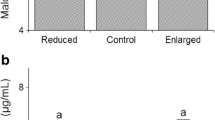
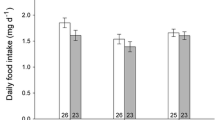
Predator presence and shortage of food resources can alter behavior and cause chronic physiological stress, with long-term detrimental effects on the prey. Recently, it has been hypothesized that cellular damage via oxidative stress could be associated with such effects. Variation in food availability and predation risk could modify the oxidative/antioxidant status of prey. However, data from wild populations is scarce and, importantly, the interactive effects of these major ecological factors on oxidative/antioxidant status are poorly understood. We present the first study in a wild vertebrate, addressing the combined effects of experimental food supplementation and increased predation risk (cues of Mustela erminea) on antioxidant status of female pied flycatchers (Ficedula hypoleuca). We tested the effects during breeding, assuming that such variation could modify the antioxidant cost of reproduction. Supplementary food increased female body mass, independently of predator treatment. However, timing of breeding and reproductive investment remained unaffected by the treatments. Principal component 1 (PC 1) of antioxidant levels [reflecting superoxidase dismutase (SOD), glutathione peroxidase (GPx), and glutathione (tGSH)] showed lower values in the food-supplemented than non-food-supplemented group. Neither PC 2 [reflecting catalase (CAT) and GSH/GSSG ratio] nor PC 1 was affected by predation risk, and further, predation risk did not alter the effect of supplementary food on either PC. These results suggest that food resources may have a stronger impact than predation risk on female antioxidant status during breeding. This is the first fully experimental study focusing on the interactive effects of key environmental variables on antioxidant status in wild vertebrates; more studies are however needed to understand the role of these factors in shaping the cost of reproduction.
Significance statement
Predator presence and shortage of food resources may modify animal behavior and cause chronic physiological stress response with long-term detrimental effects on the prey. Recently, it has been hypothesized that cellular damage via oxidative stress (i.e., imbalance between pro-oxidants and antioxidant defenses) could be associated with such effects, but evidence from wild populations is scarce. Importantly, the interactive effects of food and predation are not well understood. For the first time in a vertebrate, we experimentally studied the combined effects of food availability and (simulated) predation risk on antioxidant status, using wild breeding passerines. Higher food availability, but not increased perceived predation risk, lowered the antioxidant defenses. Predation risk did not alter the effect of supplementary food. The role of such interactions in shaping the cost of reproduction (via antioxidant/oxidative status) should be further studied.


Similar content being viewed by others
References
Alan RR, McWilliams SR (2013) Oxidative stress, circulating antioxidants, and dietary preferences in songbirds. Comp Biochem Physiol 164:185–193
Amo L, Galvan I, Tomas G, Sanz JJ (2008) Predator odour recognition and avoidance in a songbird. Funct Ecol 22:289–293
Amo L, Visser ME, van Oers K (2009) Smelling out predators is innate in birds. Ardea 99:177–184
Beaulieu M, Ropert-Coudert Y, Le Maho Y, Ancel A, Criscuolo F (2010) Foraging in an oxidative environment: relationship between delta C-13 values and oxidative status in Adelie penguins. Proc R Soc Lond B 277:1087–1092
Blount JD, Vitikainen EIK, Stott I, Cant MA (2016) Oxidative shielding and the cost of reproduction. Biol Rev 91:483–497
Boonstra R (2013) Reality as the leading cause of stress: rethinking the impact of chronic stress in nature. Funct Ecol 27:11–23
Boonstra R, Hik D, Singleton GR, Tinnikov A (1998) The impact of predator-induced stress on the snowshoe hare cycle. Ecol Monogr 68:371–394
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protien utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brinck C, Erlinge S, Sandell M (1983) Anal sac secretion in mustelids—a comparison. J Chem Ecol 9:727–745
Christensen LL, Selman C, Blount JD, Pilkington JG, Watt KA, Pemberton JM, Reid JM, Nussey DH (2015) Plasma markers of oxidative stress are uncorrelated in a wild mammal. Ecol Evol 5:5096–5108
Christians JK (2002) Avian egg size: variation within species and inflexibility within individuals. Biol Rev 77:1–26
Clinchy M, Zanette L, Boonstra R, Wingfield JC, Smith JNM (2004) Balancing food and predator pressure induces chronic stress in songbirds. Proc R Soc Lond B 271:2473–2479
Clinchy M, Zanette L, Charlier TD, Newman AEM, Schmidt KL, Boonstra R, Soma KK (2011) Multiple measures elucidate glucocorticoid responses to environmental variation in predation threat. Oecologia 166:607–614
Cohen AA, McGraw KJ, Robinson WD (2009) Serum antioxidant levels in wild birds vary in relation to diet, season, life history strategy, and species. Oecologia 161:673–683
Costantini D (2008) Oxidative stress in ecology and evolution: lessons from avian studies. Ecol Lett 11:1238–1251
Costantini D, Verhulst S (2009) Does high antioxidant capacity indicate low oxidative stress? Funct Ecol 23:506–509
Costantini D, Cardinale M, Carere C (2007) Oxidative damage and anti-oxidant capacity in two migratory bird species at a stop-over site. Comp Biochem Physiol 144:363–371
Costantini D, Dell’Ariccia G, Lipp HP (2008) Long flights and age affect oxidative status of homing pigeons (Columba livia). J Exp Biol 211:377–381
Costantini D, Rowe M, Butler MV, McGraw KJ (2010) From molecules to living systems: historical and contemporary issues in oxidative stress and antioxidant ecology. Funct Ecol 24:950–959
Costantini D, Marasco V, Møller AP (2011) A meta-analysis of glucocorticoids as modulators of oxidative stress in vertebrates. J Comp Physiol 181:447–456
Costantini D, Casasole G, Eens M (2014) Does reproduction protect against oxidative stress? J Exp Biol 217:4237–4243
Costantini D, Casasole G, AdbElgawad H, Asard H, Eens M (2016) Experimental evidence that oxidative stress influences reproductive decisions. Funct Ecol 30:1169–1174
Eggers S, Griesser M, Nystrand M, Ekman J (2006) Predation risk induces changes in nest-site selection and clutch size in the Siberian jay. Proc R Soc Lond B 273:701–706
Ercal N, Gurer-Orhan H, Aykin-Burns N (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal induced oxidative damage. Curr Topics Med Chem 1:529–539
Feoli AM, Siqueira I, Almeida LMV, Tramontina AC, Battu C, Wofchuk ST, Gottfried C, Perry ML, Goncalves CA (2006a) Brain glutathione content and glutamate uptake are reduced in rats exposed to pre- and postnatal protein malnutrition. J Nutr 136:2357–2361
Feoli AM, Siqueira IR, Almeida L, Tramontina AC, Vanzella C, Sbaraini S, Schweigert ID, Netto CA, Perry MLS, Goncalves CA (2006b) Effects of protein malnutrition on oxidative status in rat brain. Nutrition 22:160–165
Fletcher QE, Selman C, Boutin S, McAdam AG, Woods SB, Seo AY, Leeuwenburgh C, Speakman JR, Humphries MM (2013) Oxidative damage increases with reproductive energy expenditure and is reduced by food-supplementation. Evolution 67:1527–1536
Forsman JT, Mönkkönen M, Korpimäki E, Thomson RL (2013) Mammalian nest predator feces as a cue in avian habitat selection decisions. Behav Ecol 24:262–266
Giordano M, Costantini D, Pick JL, Tschirren B (2013) Female oxidative status, egg antioxidant protection and eggshell pigmentation: a supplemental feeding experiment in great tits. Behav Ecol Sociobiol 69:777–785
Guerra C, Zenteno-Savin T, Maeda-Martinez AN, Abele D, Philipp EER (2013) The effect of predator exposure and reproduction on oxidative stress parameters in the Catarina scallop Argopecten ventricosus. Comp Biochem Physiol 165:89–96
Halliwell B, Gutteridge JMC (2007) Free radicals in biology and medicine. Oxford University Press, New York
Hawlena D, Schmitz OJ (2010) Physiological stress as a fundamental mechanism linking predation to ecosystem functioning. Am Nat 176:537–556
Hoffman DJ (2002) Role of selenium toxicity and oxidative stress in aquatic birds. Aquat Toxicol 57:11–26
Isaksson C, Oernborg J, Stephensen E, Andersson S (2005) Plasma glutathione and carotenoid coloration as potential biomarkers of environmental stress in great tits. EcoHealth 2:138–146
Janssens L, Stoks R (2013) Predation risk causes oxidative damage in prey. Biol Lett 9:20130350
Janssens L, Stoks R (2014) Chronic predation risk reduces escape speed by increasing oxidative damage: a deadly cost of an adaptive antipredator response. PLoS One 9:e101273
Korpimäki E, Norrdahl K, Rintajaskari T (1991) Response of stoats and least weasels to fluctuating food abundences—is the low phase of the vole cycle due to mustelid predation. Oecologia 88:552–561
Li J, Wang H, Stoner GD, Bray TM (2002) Dietary supplementation with cysteine prodrugs selectively restores tissue glutathione levels and redox status in protein-malnourished mice. J Nutr Biochem 13:625–633
Lilley TM, Ruokolainen L, Meierjohann A, Kanerva M, Stauffer J, Laine VN, Atosuo J, Lilius EM, Nikinmaa M (2013) Resistance to oxidative damage but not immunosuppression by organic tin compounds in natural populations of Daubenton’s bats (Myotis daubentonii). Comp Biochem Physiol 157:298–305
Lundberg A, Alatalo R (1992) The pied flycatcher. Poyser, London
Meija-Carmona GE, Gosselink KL, de la Rosa LA, Pérez-Ishiwara G, Martínez-Martínez A (2014) Evaluation of antioxidant enzymes in response to predator odor stress in prefrontal cortex and amygdala. Neurochem J 8:125–128
Meister A, Anderson ME, Hwang O (1986) Intracellular cysteine and glutathione delivery systems. J Am Coll Nutr 5:137–151
Metcalfe NB, Alonso-Alvarez C (2010) Oxidative stress as a life-history constraint: the role of reactive oxygen species in shaping phenotypes from conception to death. Funct Ecol 24:984–996
Monaghan P, Metcalfe NB, Torres R (2009) Oxidative stress as a mediator of life history trade-offs: mechanisms, measurements and interpretation. Ecol Lett 12:75–92
Mönkkönen M, Forsman JT, Kananoja T, Ylönen H (2009) Indirect cues of nest predation risk and avian reproductive decisions. Biol Lett 5:176–178
Moreno J (1989) Strategies of mass change in breeding birds. Biol J Linn Soc 37:297–310
Morosinotto C, Thomson RL, Korpimäki E (2010) Habitat selection as an antipredator behaviour in a multi-predator landscape: all enemies are not equal. J Anim Ecol 79:327–333
Morosinotto C, Thomson RL, Hänninen M, Korpimäki E (2012) Higher nest predation risk in association with a top predator: mesopredator attraction? Oecologia 170:507–515
Morosinotto C, Ruuskanen S, Thomson RL, Siitari H, Korpimäki E, Laaksonen T (2013) Predation risk affects the levels of maternal immune factors in avian eggs. J Avian Biol 44:427–436
Morosinotto C, Thomson RL, Ruuskanen SR, Korpimäki E, Lehikoinen E, Möstl E, Laaksonen T (2016) Maternal transfer of androgens in eggs is affected by food supplementation but not by predation risk. J Avian Biol 47:629–641
Orell M, Ojanen M (1979) Mortality rates of the great tit Parus major in northern population. Ardea 67:130–133
Pick J, Hutter P, Ebneter C, Ziegler A-K, Giardano M, Tschirren B (2016) Artifical selection reveals the energeticc expense of producing larger eggs. Front Zool 13:38
Rainio MJ, Kanerva M, Salminen J-P, Nikinmaa M, Eeva T (2013) Oxidative status in nestlings of three small passerine species exposed to metal pollution. Sci Total Environ 454–455:466–473
Rainio MJ, Eeva T, Lilley T, Stauffer J, Ruuskanen S (2015) Effects of early-life lead exposure on oxidative status and phagocytosis activity in great tits (Parus major). Comp Biochem Physiol 167:24–34
Ruffino L, Salo P, Koivisto E, Banks PB, Korpimäki E (2014) Reproductive responses of birds to experimental food supplementation: a meta-analysis. Front Zool 11:80
Sinclair ARE, Arcese P (1995) Population consequences of predation-sensitive foraging: the serengeti wildebeest. Ecology 76:882–891
Slos S, Stoks R (2008) Predation risk induces stress proteins and reduces antioxidant defense. Funct Ecol 22:637–642
Slos S, De Meester L, Stoks R (2009) Food level and sex shape predator-induced physiological stress: immune defence and antioxidant defence. Oecologia 161:461–467
Speakman JR, Garratt M (2014) Oxidative stress as a cost of reproduction: beyond the simplistic trade-off model. BioEssays 36:93–106
Thomson RL, Gustavo T, Forsman JT, Broggi J, Mönkkönen M (2010) Predator proximity as a stressor in breeding flycatchers: mass loss, stress protein induction, and elevated provisioning. Ecology 9:1832–1840
Toumi ML, Merzoug S, Baudin B, Tahraoui A (2013) Quercetin alleviates predator stress-induced anxiety-like and brain oxidative signs in pregnant rats and immune count disturbance in their offspring. Pharmacol Biochem Behav 107:1–10
Travers M, Clinchy M, Zanette L, Boonstra R, Williams TD (2010) Indirect predator effects on clutch size and the cost of egg production. Ecol Lett 13:980–988
Vaanholt LM, Milne A, Zheng Y, Hambly C, Mitchell SE, Valencak TG, Allison DB, Speakman JR (2016) Oxidative costs of reproduction: oxidative stress in mice fed standard and low antioxidant diets. Physiol Behav 154:1–7
van de Crommenacker J, Komdeur J, Burke T, Richardson DS (2011) Spatio-temporal variation in territory quality and oxidative status: a natural experiment in the Seychelles warbler (Acrocephalus sechellensis). J Anim Ecol 80:668–680
Walankiewicz W (2002) Nest predation as a limiting factor to the breeding population size of the collared flycatcher Ficedula albicollis in the Białowieza National Park (NE Poland). Acta Ornithol 2:91–106
Williams T (2012) Physiological adaptations for breeding in birds. Princeton University Press, Pinceton
Zanette LY, Clinchy M, Smith JNM (2006) Food and predators affect egg production in song sparrows. Ecology 87:2459–2467
Zanette LY, White AF, Allen MC, Clinchy M (2011) Perceived predation risk reduces the number of offspring songbirds produce per year. Science 334:1398–1340
Zanette LY, Clinchy M, Suraci JP (2014) Diagnosing predation risk effects on demography: can measuring physiology provide the means? Oecologia 176:637–651
Acknowledgments
We thank Mirella Kanerva, Miia Rainio, and Janina Stauffer for instructions on the antioxidant assays and Alexandre Villers, Ryan Miller, Stefan Siivonen, and Elina Ode for extensive help in the fieldwork. We are grateful to the Ähtäri zoo for providing the material necessary for the predator treatment. We thank the associate editor and anonymous reviewers for their constructive feedback, which significantly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Experiments and sample collection were conducted under licenses of the Animal Experiment Committee of the State Provincial Office of Southern Finland and the Environmental Centre of Southwest Finland (license number EPOELY/456/07.01/2012).
Conflict of interest
The authors declare that they have no conflict of interest.
Data accessibility
The data will be deposited in Dryad upon acceptance.
Funding
This study was financially supported by the Academy of Finland (grant nos. 123379, 136717, and 250709 to EK, 258419 to SR), South Ostrobothnia Regional Fund of the Finnish Cultural Foundation (to CM), and Erasmus Mundus EXPERT Asia program (to CPR).
Additional information
Communicated by B. Voelkl
Rights and permissions
About this article
Cite this article
Ruuskanen, S., Morosinotto, C., Thomson, R.L. et al. Food supplementation, but not predation risk, alters female antioxidant status during breeding. Behav Ecol Sociobiol 71, 69 (2017). https://doi.org/10.1007/s00265-017-2299-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-017-2299-z