Abstract
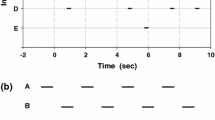
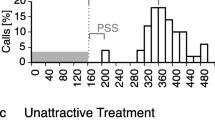
When acoustically advertising animals call in dense clusters, problems in signal efficacy often arise. These problems are particularly acute in species where females ignore males who call immediately following a neighbor and males adjust call timing to avoid broadcasting following calls: Males may forego such adjustments and produce many ineffective calls, they may attend to all neighbors and call at a reduced rate, or they may selectively attend to certain neighbors, likely those who are nearby and/or more intense. We studied the problem of group calling in Ephippiger diurnus, a European bush cricket distributed in genetically isolated populations that vary considerably in male song and chorusing and in female preference for male song. Female E. diurnus ignore following male calls, and males adjust their call timing but only with respect to several loud neighbors. We found that males were more selective in attending to only their nearest neighbor in a population where chorusing yields sound during a high proportion of a collective singing bout, and more indiscriminate in attending to several neighbors where chorusing yields more intermittent sound. Such fine tuning can maintain a relatively high calling rate and may be generated by positive feedback loops operating between individual and group-level calling traits.
Significance statement
When animals sing in the company of conspecifics, individual singers often adjust their call timing such that they do not immediately follow neighbors. These adjustments become problematic in dense choruses, as adjusting for all neighbors could lead to a marked reduction in call rate. Consequently, some degree of selective attention, most likely to nearby and/or loud neighbors, is expected. We confirmed this expectation in the chorusing bush cricket E. diurnus, but we also found that the degree of attention varied among populations. In particular, singers were most selective, attending to only a single neighbor, where chorusing generated rather continuous sound and more indiscriminate attention would have led to sporadic calling. Thus, choruses appear to be finely tuned and controlled by feedback loops in which individual singers generate a collective display that, in turn, influences the singing behavior of those very same individuals.





Similar content being viewed by others
References
Amichai E, Blumrosen G, Yovel Y (2015) Calling louder and longer: how bats use biosonar under severe acoustic interference from other bats. Proc R Soc Lond B 282:20152064. doi:10.1098/rspb.2015.2064
Barbosa F, Rebar D, Greenfield MD (2016a) Reproduction and immunity trade-offs constrain mating signals and nuptial gift size in a bushcricket. Behav Ecol 27:109–117
Barbosa F, Rebar D, Greenfield MD (2016b) Female preference functions drive inter-population divergence in male signaling: call diversity in the bushcricket Ephippiger diurnus. J Evol Biol (in press)
Buck J, Buck E, Case JF, Hanson FE (1981a) Control of flashing in fireflies. V. Pacemaker synchronization in Pteroptyx cribellata. J Comp Physiol A 144:287–298
Buck J, Buck E, Hanson FE, Case JF, Mets L, Atta GJ (1981b) Control of flashing in fireflies. IV. Free run pacemaking in synchronic Pteroptyx. J Comp Physiol A 144:277–286
Camazine S, Deneubourg J-L, Franks N, Sneyd J, Theraulaz G, Bonabeau E (2001) Self-organization in biological systems. Princeton University Press, Princeton, p. 560
Duijm M (1990) On some song characteristics in Ephippiger (Orthoptera, Tettigonioidea) and their geographic variation. Neth J Zool 40:428–453
Dyson ML, Passmore NI (1988) Two-choice phonotaxis in Hyperolius marmoratus (Anura: Hyperoliidae): the effect of temporal variation in presented stimuli. Anim Behav 36:648–652
Greenfield MD (1994a) Cooperation and conflict in the evolution of signal interactions. Annu Rev Ecol Syst 25:97–126
Greenfield MD (1994b) Synchronous and alternating choruses in insects and anurans: common mechanisms and diverse functions. Am Zool 34:605–615
Greenfield MD (2005) Mechanisms and evolution of communal sexual displays in arthropods and anurans. Adv Study Behav 35:1–61
Greenfield MD (2015) Signal interactions and interference in insect choruses: singing and listening in the social environment. J Comp Physiol A 201:143–154 Special issue: Insect hearing: From physics to ecology
Greenfield MD, Rand AS (2000) Frogs have rules: selective attention algorithms regulate chorusing in Physalaemus pustulosus (Leptodactylidae). Ethology 106:331–347
Greenfield MD, Roizen I (1993) Katydid synchronous chorusing is an evolutionarily stable outcome of female choice. Nature 364:618–620
Greenfield MD, Schul J (2008) Mechanisms and evolution of synchronous chorusing: emergent properties and adaptive functions in Neoconocephalus katydids (Orthoptera: Tettigoniidae). J Comp Psychol 122:289–297
Greenfield MD, Snedden WA (2003) Selective attention and the spatio-temporal structure of orthopteran choruses. Behaviour 140:1–26
Greenfield MD, Esquer-Garrigos Y, Streiff R, Party V (2016) Animal choruses emerge from receiver psychology. Sci Rep, in review.
Greenfield MD, Siegfreid E, Snedden WA (2004) Variation and repeatability of female choice in a chorusing katydid, Ephippiger ephippiger: an experimental exploration of the precedence effect. Ethology 110:287–299
Greenfield MD, Tourtellot MK, Snedden WA (1997) Precedence effects and the evolution of chorusing. Proc R Soc Lond B 264:1355–1361
Hanson FE, Case JF, Buck E, Buck J (1971) Synchrony and flash entrainment in a New Guinea firefly. Science 174:161–164
Hartbauer M, Kratzer S, Steiner K, Römer H (2005) Mechanisms for synchrony and alternation in song interactions of the bushcricket Mecopoda elongata (Tettigoniidae: Orthoptera). J Comp Physiol A 191:175–188
Höbel G (2010) Interaction between signal timing and signal feature preferences: causes and implications for sexual selection. Anim Behav 79:1257–1266
Litovsky RY, Colburn HS, Yost WA, Guzman SJ (1999) The precedence effect. J Acoust Soc Am 106:1633–1654
Minckley RL, Greenfield MD (1995) Psychoacoustics of female phonotaxis and the evolution of male signal interactions in Orthoptera. Ethol Ecol Evol 7:235–243
Minckley RL, Greenfield MD, Tourtellot MK (1995) Chorus structure in tarbush grasshoppers: inhibition, selective phonoresponse, and signal competition. Anim Behav 50:579–594
Moiseff A, Copeland J (2010) Firefly synchrony: a behavioral strategy to minimize visual clutter. Science 329:181
Murphy MA, Thompson NL, Schul J (2016) Keeping up with the neighbor: a novel mechanism of call synchrony in Neoconocephalus ensiger katydids. J Comp Physiol A 202:225–234
Nityananda V, Balakrishnan R (2007) Synchrony during acoustic interactions in the bushcricket Mecopoda ’chirper’ (Tettigoniidae: Orthoptera) is generated by a combination of chirp-by-chirp resetting and change in intrinsic chirp rate. J Comp Physiol A 193:51–65
Nityananda V, Stradner J, Balakrishnan R, Römer H (2007) Selective attention in a synchronising bushcricket: physiology, behaviour and ecology. J Comp Physiol A 193:983–991
Party V, Brunel-Pons O, Greenfield MD (2014) Priority of precedence: receiver psychology, female preference for leading calls and sexual selection in insect choruses. Anim Behav 87:175–185
Party V, Streiff R, Marin-Cudraz T, Greenfield MD (2015) Group synchrony and alternation as an emergent property: elaborate chorus structure in a bushcricket is an incidental by-product of female preference for leading calls. Behav Ecol Sociobiol 69:1957–1973
Pollack GS (1988) Selective attention in an insect auditory neuron. J Neurosci 8:2635–2639
Rebar D, Barbosa F, Greenfield MD (2016) Acoustic experience influences male and female pre- and postcopulatory behaviors in a bushcricket. Behav Ecol 27:434–443
Römer H, Krusch M (2000) A gain-control mechanism for processing of chorus sounds in the afferent auditory pathway of the bushcricket Tettigonia viridissima (Orthoptera; Tettigoniidae). J Comp Physiol A 186:181–191
Sismondo E (1990) Synchronous, alternating, and phase-locked stridulation by a tropical katydid. Science 249:55–58
Snedden WA, Greenfield MD, Jang Y (1998) Mechanisms of selective attention in grasshopper choruses: who listens to whom? Behav Ecol Sociobiol 43:59–66
Spooner LJ, Ritchie MG (2006) An unusual phylogeography in the bushcricket Ephippiger ephippiger from southern France. Heredity 97:398–408
Ulanovsky N, Fenton MB, Tsoar A, Korine C (2004) Dynamics of jamming avoidance in echolocating bats. Proc R Soc Lond B 271:1467–1475
Walker TJ (1969) Acoustic synchrony: two mechanisms in the snowy tree cricket. Science 166:891–894
Zollinger SA, Brumm H (2011) The Lombard effect. Curr Biol 21:R614–R615
Zurek PM (1987) The precedence effect. In: Yost WA, Gourevitch G (eds) Directional hearing. Springer-Verlag, New York, pp. 85–105
Acknowledgments
We thank Guy Bourdais, Séverine Devers, Yann Emmelin, Alicia Jarrige, Virginie Party, and Justine Penin for technical assistance in the laboratory, and the Agence Nationale de la Recherche de France (contrat ANR-11-BSV7-025-01; EVOLCHOR), the Centre National de la Recherche Scientifique (CNRS), and the Université François Rabelais de Tours for their financial support. Virginie Party, Darren Rebar, and several referees provided valuable criticisms of earlier versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was funded by grant ANR-11-BSV7-025-01 from the Agence Nationale de la Recherche de France. The authors declare that they have no conflicts of interest pertaining to the research reported in this manuscript. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Communicated by S. Sakaluk
Rights and permissions
About this article
Cite this article
Marin-Cudraz, T., Greenfield, M.D. Finely tuned choruses: bush crickets adjust attention to neighboring singers in relation to the acoustic environment they create. Behav Ecol Sociobiol 70, 1581–1589 (2016). https://doi.org/10.1007/s00265-016-2166-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-016-2166-3