Abstract
Background
Luminal and Basal are the primary intrinsic subtypes of muscle-invasive bladder cancer (MIBC). The presence of CD8+ T cells infiltration holds significant immunological relevance, potentially influencing the efficacy of antitumor responses. This study aims to synergize the influence of molecular subtypes and CD8+ T cells infiltration in MIBC.
Methods
This study included 889 patients with MIBC from Zhongshan Hospital, The Cancer Genome Atlas, IMvigor210 and NCT03179943 cohorts. We classified the patients into four distinct groups, based on the interplay of molecular subtypes and CD8+ T cells and probed into the clinical implications of these subgroups in MIBC.
Results
Among patients with Luminal-CD8+Thigh tumors, the confluence of elevated tumor mutational burden and PD-L1 expression correlated with a heightened potential for positive responses to immunotherapy. In contrast, patients featured by Luminal-CD8+Tlow displayed a proclivity for deriving clinical advantages from innovative targeted interventions. The Basal-CD8+Tlow subgroup exhibited the least favorable three-year overall survival outcome, whereas their Basal-CD8+Thigh counterparts exhibited a heightened responsiveness to chemotherapy.
Conclusions
We emphasized the significant role of immune-molecular subtypes in shaping therapeutic approaches for MIBC. This insight establishes a foundation to refine the process of selecting subtype-specific treatments, thereby advancing personalized interventions for patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bladder cancer is one of the most common genitourinary malignancies with a high mortality risk [1]. Within this context, approximately a quarter of cases are identified as muscle-invasive bladder cancer (MIBC), carrying an unfavorable prognosis [2]. Over the past decades, platinum-based chemotherapy has been regarded as the standard care for MIBC patients [3]. For those patients ineligible or resistant to platinum-based treatment, certain immune checkpoint inhibitors have gained clinical benefit and have been approved for MIBC treatment in recent years. Despite this, the favorable therapeutic outcomes have remained confined to a limited patient subset [4]. This therapeutic landscape has facilitated the development of a more precise, biology-driven classification of MIBC, aimed at enhancing clinical management for patients.
The molecular classification of bladder cancer has emerged as a pivotal approach to understanding the diversity of MIBC. Previous studies shown that two primary intrinsic subtypes of MIBC have emerged—Luminal and Basal [5,6,7]. These intrinsic subtypes hold immediate potential in guiding both prognosis and treatment strategies for MIBC. Notably, it has come to light that the MIBC with the basal feature is linked to reduced disease-free survival (DFS) and overall survival (OS) in comparison with the luminal subtypes [6]. Furthermore, a significant proportion of Basal MIBC cases could get benefit from chemotherapy, contrasting with the resistance observed in Luminal subtype tumors [6]. However, the responses to immune therapies were observed not only in metastatic urothelial carcinoma (mUC) patients with luminal features but also in those with basal features [8].
Beyond the genetic and molecular attributes, the infiltration and functional status of immune cells exert a considerable influence on patient prognosis and treatment responses. Notably, the CD8+ T cells have emerged as a pivotal immune parameter with the potential to decisively impact effective antitumor responses [9,10,11]. Numerous researches have demonstrated the correlation between CD8+ T cells infiltration and improved prognosis as well as enhanced therapeutic effectiveness [12, 13]. Our previous investigations also emphasize the significant heterogeneity of intratumoral CD8+ T cells in the context of MIBC. These intratumoral CD8+ T cells exhibit diverse phenotypes that ultimately influence the direction of the anti-tumor immune response [14,15,16].
In this study, we established an immunomolecular subtype classification, achieved by integrating and categorizing molecular subtypes alongside CD8+ T cells infiltration into four distinct groups within MIBC: Luminal-CD8+Thigh, Luminal-CD8+Tlow, Basal-CD8+Thigh and Basal-CD8+Tlow. Subsequently, we conducted an in-depth analysis encompassing clinicopathological, genomic, and immunophenotypic attributes across these four stratified subgroups. Moreover, we investigated how this novel classification influences both chemotherapy and PD-L1 blockade therapy outcomes in patients with MIBC.
Materials and methods
Study cohorts
The study utilized data from four independent patient cohorts, consisting of a total of 889 patients (Supplementary Fig. 1A). For our local Zhongshan Hospital Affiliated to Fudan University (ZSHS) cohort, with the approval of the Clinical Research Ethics Committee of Zhongshan Hospital affiliated to Fudan University, 142 patients who were diagnosed with MIBC and underwent radical cystectomy at Zhongshan Hospital from 2002 to 2014 were followed up regularly till July 2016. Among them, 7 patients were excluded from the analysis due to dot loss on tissue microarray (TMA). Of these, 64 patients received adjuvant chemotherapy to reduce the risk of disease relapse according to the guidelines and the preferences of the patients, while 71 did not receive any neoadjuvant or adjuvant therapy. In clinical settings, platinum-based chemotherapy remains the standard for treating advanced and metastatic bladder cancer [17]. Our local ZSHS cohort maintained consistency in systemic therapy over a 12-year period, with patients being administered platinum-based chemotherapy rather than immunotherapy. The Cancer Genome Atlas (TCGA) cohort encompasses 391 MIBC patients with comprehensive clinical profiles. Initially, a pool of 412 patients diagnosed with bladder cancer was sourced from the TCGA network using TCGA-Assembler 2.0.6 as of July 2021. Subsequently, 21 cases were excluded from this pool due to reasons such as non-muscle-invasive bladder cancer (NMIBC) pathologic diagnosis (n = 4), absence of survival or mRNA data (n = 7), or prior neoadjuvant therapy (n = 10). Regarding the IMvigor210 cohort, a phase II clinical trial that comprises 348 individuals diagnosed with mUC treated with the PD-L1 inhibitor atezolizumab, clinical and RNA-seq data were procured from http: //research-pub.gene.com/IMvigor210CoreBiologies utilizing the "IMvigor210CoreBiologies" R package. As for the phase II clinical trial NCT03179943 cohort, it encompassed 21 patients who underwent treatment with guadecitabine and atezolizumab. Ultimately, the mRNA data of 15 patients were incorporated into this study. Detailed clinical and pathological attributes are presented in Supplementary Tables 1–3.
Processing of genomic and transcriptomic data
For transcriptomic analyses, RNA-seq data were normalized through the formula Log2(FPKM + 1). Immune cells infiltration signature including CD8+ T cells signature [18], effector CD8+T signature [4], exhausted CD8+T signature [19] and immune function signature including IFN-γ-related signature [20], pan fibroblast TGF-β signature [21] were calculated by Single sample gene set enrichment analysis (ssGSEA) algorithms based on related genes expression as previously reported (genes are listed in Supplementary Table 4). ssGSEA algorithms was accomplished by a ‘single sample’ extension of GSEA that allows one to define an enrichment score that represents the degree of absolute enrichment of a gene set in each sample within a given data set. The gene expression values for a given sample were rank-normalized, and an enrichment score was produced using the empirical cumulative distribution functions (ECDF) of the genes in the signature and the remaining genes [22].
For genomic analyses, tumor mutation burden (TMB) was defined as total non-silent somatic mutation per megabase (mut/Mb) which was acquired from https://portal.gdc.cancer.gov/. The copy number variants (CNV) data in TCGA cohort originated from http://www.cbioportal.org. Amplified or deleted regions of the genome were identified using the Genomic Identification of Significant Targets in Cancer (GISTIC) 2.0 algorithm, which divided into five discrete steps: (1) accurately defining the copy-number profile of each cancer sample; (2) identifying the somatic copy number alterations (SCNAs) that most likely gave rise to these overall profiles and estimating their background rates of formation; (3) scoring the SCNAs in each region according to their likelihood of occurring by chance; (4) defining the independent genomic regions undergoing statistically significant levels of SCNA; and (5) identifying the likely gene target(s) of each significantly altered region [23].
Immunohistochemistry and assay method
Serial tissue sections fixed in formalin and embedded in paraffin were used to construct TMA and the antibodies used for immunohistochemistry (IHC) staining are summarized in Supplementary Table 5. IHC staining for CD8+ T cells and other molecules were carried out according to previous protocols [14, 24, 25] (representative IHC images, Supplementary Fig. 1B). As before, the infiltration density of immune cells was evaluated as the mean value of cells/HPF under 3 randomized high-power magnification filed (HPF, × 200 magnification) independently by two pathologists who were blind to the clinical information with NanoZoomer-XR (Hamamatsu) and Image Pro plus 6.0 digitally. The cut-off values of CD8+ T cells were determined by the median value in each study cohort. In TCGA cohort, IMvigor210 cohort and NCT03179943 cohort, the cut-off points were 0.910, 1.035 and 0.530, respectively, while the cut-off value of was set at 25 cells/HPF in ZSHS cohort.
Classification of molecular subtype
The molecular subtype of each patient was identified through ‘BLCAsubtyping’ R package from https://github.com/cit-bioinfo/BLCAsubtyping in TCGA, IMvigor210 and NCT03179943 cohorts. In ZSHS cohort, we performed IHC single staining of GATA3 and KRT5/6 to classify patients into either Luminal or Basal subtype according to an IHC subtyping method reported in previous articles [25, 26]. GATA3 is predominantly expressed in the Luminal subtype, whereas KRT5/6 is regarded as a Basal marker. IHC analysis determined whether KRT5/6 and GATA3 expression was positive or negative, with the cut-off value being 20% tumor tissue positivity under a 200 × high power field. KRT5/6+GATA3− tumors were classified as Basal subtype, while the rest were classified as Luminal subtype.
Statistical analysis
Kaplan–Meier analysis and log-rank test were performed to conduct survival analyses. Univariate analysis was conducted using the Cox proportional hazards regression model. Pearson's Chi-square test and Fisher’s exact test were applied to detect categorical variables. Statistical P values were computed using Kruskal–Wallis test, and detailed statistical tests were described in corresponding figure legends. A P value less than 0.05 was considered statistically significant. All statistical analyses were conducted using IBM SPSS Statistics 26.0 and R software 4.1.2.
Results
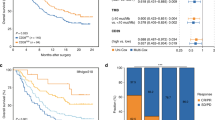
Basal-CD8+Tlow predicts inferior overall survival outcomes in MIBC.
We first explored the association between OS and the four molecular subtype/CD8+ T cells-stratified subgroups. The results demonstrated that patients classified under the Basal subtype with low CD8+ T cells infiltration (median OS: 17.7 months) exhibited the poorest prognosis, in contrast to the more favorable outcomes observed for the Basal-CD8+Thigh (median OS: 32.0 months), Luminal-CD8+Thigh (median OS: 44.9 months), and Luminal-CD8+Tlow (median OS: 44.3 months) subgroups within the TCGA cohort (log-rank P = 0.019, Fig. 1a). Importantly, a coherent pattern emerged from the analysis of the ZSHS cohort, consistently indicating that the Basal-CD8+Thigh subgroup was linked to comparatively inferior OS outcomes (log-rank P = 0.025, Fig. 1c). Moreover, univariate analyses confirmed the independent correlation between the Basal-CD8+Thigh subgroup and decreased mortality when contrasted against the Basal-CD8+Thigh (HR: 0.571, 95% CI: 0.347–0.940, P = 0.027 and HR: 0.348, 95% CI: 0.143–0.848, P = 0.020 in respective cohorts), Luminal-CD8+Tlow (HR: 0.487, 95% CI: 0.297–0.799, P = 0.004 and HR: 0.475, 95% CI: 0.229–0.985, P = 0.046 in respective cohorts) and Luminal-CD8+Thigh (HR: 0.454, 95% CI: 0.258–0.796, P = 0.006 and HR: 0.323, 95% CI: 0.147–0.711, P = 0.005 in respective cohorts) subgroups, consistently across both the TCGA and ZSHS cohorts (Fig. 1b, d).
Basal-CD8+Tlowpredicts inferior overall survival outcomes in MIBC. (A) Kaplan–Meier analyses for OS according to four molecular subtype/CD8+T cells-stratified subgroups in TCGA cohort. (B) Cox regression analyses of OS according to CD8+ T cells, molecular subtype and four stratified subgroups in TCGA cohort. (C) Kaplan–Meier analyses for OS according to four molecular subtype/CD8+T cells-stratified subgroups in ZSHS cohort. (D) Cox regression analyses of OS according to CD8+ T cells, molecular subtype and four stratified subgroups in ZSHS cohort. Log-rank test was conducted for Kaplan–Meier curves. P ≤ 0.05 was considered statistical significance. OS, overall survival
Basal-CD8+Thigh predicts chemotherapy sensitivity in MIBC.
The potential of adjuvant chemotherapy (ACT) to extend OS among patients with the Basal subtype was observed (Supplementary Fig. 2A). To delve deeper into the interplay between molecular subtype, CD8+ T cells infiltration, and the effectiveness of ACT in MIBC, we conducted further exploration. Our analysis unveiled a noteworthy pattern that only patients characterized by the Basal subtype and high levels of CD8+ T cells infiltration experienced discernible clinical advantages from platinum-based chemotherapy. This pattern was consistent across both the TCGA cohort (HR: 0.318, 95% CI: 0.170–0.594, log-rank P < 0.001, Fig. 2a), as well as the ZSHS cohort (HR: 0.139, 95% CI: 0.018–1.105, log-rank P = 0.029, Fig. 2b).
Basal-CD8+Thigh predicts chemotherapy sensitivity in MIBC. (A) Kaplan–Meier analyses of OS between patients treated with ACT or not in four subgroups stratified by molecular subtype and CD8+ T cells in TCGA cohort. (B) Kaplan–Meier analyses of OS between patients treated with ACT or not in four subgroups stratified by molecular subtype and CD8+T cells in ZSHS cohort. Log-rank test was conducted for Kaplan–Meier curves. P ≤ 0.05 was considered statistical significance. OS, overall survival; ACT, adjuvant chemotherapy
Luminal-CD8+Thigh predicts clinical benefits to PD-L1 blockade in MIBC.
We further explored the therapeutic implications for immune checkpoint blockade (ICB) within this paradigm. The Luminal subtype indicated a pronounced survival advantage derived from PD-L1 blockade therapy (log-rank P = 0.028), while the Basal subtype did not show similar benefits (log-rank P = 0.200, Fig. 2a and Supplementary Fig. 2B). Our exploration further delved into the intricate connection between the four molecular subtype/CD8+ T cells-stratified subgroups and their responsiveness to immunotherapy. Utilizing data from the IMvigor210 cohort, our findings highlighted a distinct pattern that solely patients featured by Luminal-CD8+Thigh exhibited significantly enhanced OS (log-rank P = 0.009, Fig. 2b) and a heightened objective response rate (ORR, defined as the proportion of patients with complete response and partial response; P = 0.002, Fig. 2c) subsequent to treatment with atezolizumab.
We next investigated data from the NCT03179943 phase II clinical trial, encompassing 15 patients subjected to guadecitabine in combination with atezolizumab. Specifically, among these patients, those falling under the Basal-CD8+Thigh subgroup (median OS: 20.1 months) exhibited a noteworthy extension in OS through immuno-chemotherapy in comparison with counterparts in the Basal-CD8+Tlow (median OS: 7.1 months), Luminal-CD8+Thigh (median OS: 2.9 months), and Luminal-CD8+Tlow (median OS: 3.7 months, Fig. 3e) subgroups. Importantly, it is worth noting that all individuals who achieved stable disease (SD) belonged to the Basal-CD8+Thigh subgroup (Fig. 3d).
Luminal-CD8+Thigh predicts clinical benefits to PD-L1 blockade in MIBC. (A) Kaplan–Meier analyses for OS of Luminal patients (left panel) and Basal patients (right panel) between high/low CD8+ T cells groups after treatment by atezolizumab in IMvigor210 cohort. (B) Kaplan–Meier analyses for OS according to four molecular subtype/CD8+T cells-stratified subgroups in IMvigor210 cohort. (C) Fractions of objective response to atezolizumab between four molecular subtype/CD8+T cells-stratified subgroups. (D) Characteristics of the response to guadecitabine plus atezolizumab according to four molecular subtype/CD8+T cells-stratified subgroups in NCT03179943 cohort. (E) Kaplan–Meier analyses of OS on the basis of four molecular subtype/CD8+T cells-stratified subgroups patients treated with guadecitabine plus atezolizumab from NCT03179943. Log-rank test was conducted for Kaplan–Meier curves. Kruskal–Wallis test and Chi-square test were applied. P ≤ 0.05 was considered statistical significance. OS, overall survival
Immunogenomic features stratified by molecular subtype and CD8+ T cells Infiltration.
In an effort to unravel the underlying rationale behind the predictive significance of integrating molecular subtype and CD8+ T cells infiltration, we next characterized the immune contexture within the context of the four subgroups. As illustrated, patients belonging to the Luminal-CD8+Thigh subgroup exhibited significantly elevated tumor neoantigen burden across both the TCGA and IMvigor210 cohorts (Fig. 4a and Supplementary Fig. 3B). Furthermore, the subgroup characterized by the Luminal subtype and high CD8+ T cells infiltration demonstrated notably heightened infiltration of effector CD8+ T cells relative to exhausted CD8+ T cells (Fig. 4b). To corroborate these outcomes, we further investigated the presence of CD103+CD8+ T cells and CXCR5+CD8+ T cells, which demonstrated heightened levels among patients with Luminal-CD8+Thigh subtype. In contrast, the Basal-CD8+Thigh subgroup within the ZSHS cohort exhibited a notable enrichment of TIGIT+CD8+ T cells (as illustrated in Fig. 4c).
Immunogenomic features stratified by molecular subtype and CD8+T cells Infiltration. (A) Evaluation of tumor neoantigen burden between four molecular subtype/CD8+T cells-stratified subgroups in TCGA cohort. (B) Heatmap illustrating immune functional signatures and immune cells signatures of TCGA cohort to detect the correlation between four molecular subtype/CD8+T cells-stratified subgroups and immune microenvironment. (C) Correlation between CD8+ T cells infiltration, CD103+CD8+T cells infiltration, CXCR5+CD8+T cells infiltration, TIGIT+CD8+T cells infiltration and four molecular subtype/CD8+T cells-stratified subgroups in ZSHS. (D) Correlation between IFNγ+ cells infiltration, GZMB+ cells infiltration, TGF-β+ cells infiltration, IL10+ cells infiltration and four molecular subtype/CD8+T cells-stratified subgroups in ZSHS. cohort. (E) Correlation between PD-1+ cells infiltration, PD-L1+ cells infiltration, TIM-3+ cells infiltration, CTLA-4+ cells infiltration and four molecular subtype/CD8+T cells-stratified subgroups in ZSHS. cohort. (F) Association between tumor mutational signatures and four molecular subtype/CD8+T cells-stratified subgroups in TCGA cohort. Kruskal–Wallis test and Chi-square test were applied. P ≤ 0.05 was considered statistical significance
Additionally, patients featured by Luminal-CD8+Thigh exhibited elevated IFN-γ-related and IFN-γ response signatures, while demonstrating reduced TGF-β response and pan fibroblast TGF-β signatures (Fig. 4b) in the TCGA cohort. These findings were replicated in the ZSHS cohort. Patients categorized as Luminal-CD8+Thigh exhibited the highest infiltration of IFN-γ+ cells and GZMB+ cells. Conversely, TGF-β+ cells and IL-10+ cells were more abundant within the Basal-CD8+Thigh subgroup (Fig. 4d). Immunohistochemistry analysis further validated these trends. Notably, the Luminal-CD8+Thigh subgroup within the ZSHS cohort displayed an increased presence of various immune checkpoints, including PD-1+ cells and PD-L1+ cells. Conversely, patients in the Basal-CD8+Thigh subgroup showcased elevated expression of exhaustion markers such as TIM-3+ cells and CTLA-4+ cells (Fig. 4e).
Through analyses of the TCGA cohort, we found that as the TMB increased, the Luminal-CD8+Thigh subgroup exhibited an augmentation in both APOBEC mutational signatures and aneuploidy scores (Fig. 4f). Furthermore, beyond the prevalent chromosome 9p21.3 deletion and 11q13.3 amplification characteristic of bladder cancer, we observed unique alterations. Specifically, the Luminal-CD8+Thigh subgroup displayed 3p25.2 PPARG amplification and 13q14.2 RB1 deletion, while the Luminal-CD8+Tlow subgroup exhibited 1q23.3 NECTIN4 amplification, 4p16.3 FGFR3 amplification, and 7p11.2 EGFR amplification (Fig. 4f).
Luminal-CD8+Tlow predicts superior response to targeted therapies in MIBC
In pursuit of further therapeutic avenues for MIBC patients, we sought to establish a connection between emerging biomarkers and the four molecular subtype/CD8+T cells-stratified subgroups. Specifically, patients featured by Luminal-CD8+Tlow exhibited a propensity to possess FGFR3 single nucleotide variants (SNVs), which were classified under level 1 of actionability according to the OncoKB database (Fig. 5a). Moreover, FGFR3 hotspot mutations and FGFR3-TACC fusion events were notably enriched in patients characterized by the Luminal subtype and low CD8+ T cells infiltration (Fig. 5b). This finding underscored the potential benefits that patients with Luminal-CD8+Tlow subtype could derive from FGFR3 inhibitors.
Luminal-CD8+Tlow predicts superior response to novel targets treatment in MIBC. (A) Highest levels of therapeutic actionability in MIBC stratified by four molecular subtype/CD8+T cells-stratified subgroups. (B-E) Distribution of FGFR3 hotspot mutation, FGFR3-TACC3 fusion(B) NECTIN4 mRNA expression, NECTIN4 amplification(C), TACSTD2 (Trop-2) mRNA expression(D) and HER2 protein expression among four molecular subtype/CD8+T cells-stratified subgroups in TCGA cohort. Kruskal–Wallis test and Chi-square test were applied. P ≤ 0.05 was considered statistical significance
In terms of the potential benefit of antibody–drug conjugate (ADC) drugs, we identified that patients featured by Luminal-CD8+Tlow displayed elevated NECTIN4 mRNA expression and a greater proportion of NECTIN4 amplifications (Fig. 5c). These observations proposed a potential clinical advantage for patients with Luminal-CD8+Tlow subtype through the utilization of NECTIN4-ADC drugs. Moreover, we ascertained that TACSTD2 mRNA expression was prominently enriched in the Luminal-CD8+Tlow subgroup (Fig. 5d). This finding indicated that patients within this subgroup might benefit from TROP2-ADC drug treatments. Furthermore, the high expression of HER2 protein within the Luminal-CD8+Tlow subgroup (Fig. 5e) indicated the sensitivity of these patients to HER2-targeted drug interventions as well.
Discussion
Using independent discovery in distinct datasets, we defined four subtypes of MIBC with differ molecular characteristics, immune infiltration, clinical outcomes and therapeutic strategies based on integrating CD8+ T cells and molecular subtype (Supplementary Figs. 3 and 4).
Patients belonging to the Luminal-CD8+Thigh subgroup exhibited the most notable levels of TMB and tumor neoantigen burden (TNB). It is well-established that MIBC patients with elevated TMB tend to experience more favorable clinical outcomes when treated with ICB [8, 27]. Remarkably, patients featured by Luminal-CD8+Thigh displayed a large amount of effector CD8+T cells infiltration, particularly CD103+CD8+T cells and CXCR5+CD8+T cells. Previous studies have consistently demonstrated that the enrichment of CD103+CD8+T cells and CXCR5+CD8+T cells is associated with robust cytotoxic activity [14, 16]. This highlights the potential of these infiltrating immune cells to mount an effective defense against tumor cells. Furthermore, research has indicated that the accumulation of APOBEC-mediated mutagenesis signatures serves as a positive prognostic indicator and predictor of favorable response to immunotherapy in bladder cancer patients [28]. In our study, we confirmed that Luminal-CD8+Thigh subgroup exhibited increased levels of both IFN-γ-related signature and APOBEC signature. These findings strongly suggest that patients within this subgroup are highly likely to benefit significantly from immunotherapy interventions. Moreover, patients with Luminal-CD8+Thigh subtype were characterized by the amplification of PPARG. Notably, a pre-clinical study demonstrated that the downregulation of PPARG expression levels using a PPARγ-inhibitor led to an anti-proliferative effect on tumor cells [29]. Therefore, combining PPARγ-inhibitor therapy with other treatment modalities could potentially provide added value and improved outcomes for patients featured by Luminal-CD8+Thigh.
In Luminal-CD8+Tlow subtype of MIBC patients, we observed an enrichment of FGFR3 mutations and FGFR3-TACC3 fusion. These findings suggest that this particular subtype may be responsive to treatment with FGFR inhibitors. In addition, a promising treatment option is the NECTIN4-ADC enfortumab vedotin (EV), which induced objective clinical responses in 44% of mUC patients with failed treatment for chemotherapy and immunotherapy [30]. We also discovered significantly increased in the mRNA expression of NECTIN4 and the NECTIN4 amplification faction within the Luminal-CD8+Tlow subgroup. This suggests that this subgroup is likely to experience clinical benefits from EV treatment. Another ADC, sacituzumab govitecan (SG), targeting TROP2, demonstrated a 27% ORR in mUC patients [31]. In our study, we validated that TACSTD2 mRNA expression is expanded in the Luminal-CD8+Tlow subgroup, suggesting that these patients may also be sensitive to TROP2-ADC. Furthermore, the expression levels of HER2 and EGFR were also elevated in this subgroup, indicating that HER2-ADC and EGFR-targeted drugs might bring clinical benefits to these patients. Currently, bispecific ADCs are undergoing clinical trials and are expected to provide more treatment options for patients in Luminal-CD8+Tlow subgroup.
In Basal-CD8+Thigh subtype of MIBC patients, patients showed a satisfactory median OS after receiving chemotherapy or chemotherapy plus immunotherapy. Although these patients exhibited a remarkable infiltration of CD8+ T cells, it is important to note that a significant proportion of these CD8+ T cells are characterized dysfunctional TIGIT+CD8+T cells, which could promote MIBC immune escape [15]. Furthermore, the Basal-CD8+Thigh MIBC subgroup showed an enrichment epithelial-to-mesenchymal transition (EMT) signature, suggesting that Basal-CD8+Thigh tumor cells are more invasive and associated with worse clinical outcomes [32]. Previous studies have shown that TGF-β expression and angiogenesis are associated with resistance to immune checkpoint inhibitors (ICIs) in UC [33], [34]. In our study, we observed that the Basal-CD8+Thigh subgroup displayed the highest TGF-β pathway activity and angiogenesis signature, which may help explain the different clinical outcomes observed in these patients compared to those in the Luminal-CD8+Thigh subgroup [35]. Furthermore, combination therapy with TGF-β inhibitors or anti-angiogenic drugs could provide added value in treating these patients.
Patients with the Basal-CD8+Tlow subtype experienced the worst OS compared to other subtypes. Numerous researches have demonstrated the detrimental role of WNT5A, a protein involved in cell signaling, in driving chemotherapy resistance and promoting the expression of stemness markers in bladder cancer cells [36]. This effect is primarily due to the activation of the WNT/β-catenin signaling pathway, which has been extensively linked to poor prognosis and drug resistance across various cancer types [37,38,39,40]. We also found that the activity of the WNT pathway significantly increases in patients with the Basal-CD8+Tlow subtype. CDKs play a crucial role in regulating the cell cycle and their inhibitors have shown promising results in targeting cancer cells, repressing their uncontrolled growth, and potentially sensitizing them to chemotherapy [41]. By specifically targeting the dysregulated cell cycle pathway in patients with Basal-CD8+Tlow tumor, CDK inhibitors have the potential to improve treatment outcomes and enhance OS rates in this challenging patient population [42].
Our research has several limitations. Firstly, given the retrospective and exploratory design of our study, the results still require further validation within the framework of more extensive, multi-centered clinical cohorts. Secondly, while we do present compelling evidence of the correlation between four molecular subtype/CD8+T cells-stratified subgroups and the survival benefit from ACT in our local cohort, validating our immunotherapy-related findings still requires further investigation. Thirdly, the patient number of some cohorts included is too small to achieve statistical significance for overall survival which needs further validation. Fourthly, the four subgroups are different in patients’ numbers and heterogeneity, and more validation is needed to enhance the credibility of the study. Finally, we recognize the need for functional research and prospective experiments to validate our hypotheses.
In summary, we integrated findings from our study analyses of patients with MIBC (Fig. 6). Our study highlighted the potential of the immunomolecular subtype based on molecular subtype (Luminal and Basal) and CD8+T cells infiltration. For patients featured by Luminal-CD8+Thigh, along with the highest level of TMB and PD-L1 expression, these patients were likely to benefit from immunotherapy. Patients with Luminal-CD8+Tlow tumors are more likely to benefit from FGFR3 inhibitors and ADC drugs including NECTIN4-ADC, TROP2-ADC and HER2-ADC. Patients with Basal-CD8+Thigh subtype were sensitive to chemotherapy or chemotherapy plus immunotherapy. Patients featured by Basal-CD8+Tlow had the worst three-year OS.
Clinical outcomes summary for patients with MIBC organized by four molecular subtype/CD8+T cells-stratified subgroups. Three-year overall survival was calculated using Kaplan–Meier analysis for MIBC patients. The average PD-L1 expression (scaled dot size) and effective treatment strategies (dot color) are represented from multi-cohort analyses
Conclusion
Our study defined an immune-molecular subtyping classification based on CD8+T cells and molecular subtypes. This classification is hoped to serve as a foundation for developing tailored therapeutic approaches for MIBC patients.
Data availability
Data and materials generated that are relevant to the results are included in this article. Other data are available from the corresponding author Prof. Zhang upon reasonable request.
Abbreviations
- ACT:
-
Adjuvant chemotherapy
- ADC:
-
Antibody–drug conjugate
- CNV:
-
Copy number variants
- CTLA-4:
-
Cytotoxic T lymphocyte-associated antigen-4
- EMT:
-
Epithelial-mesenchymal transition
- FGFR3:
-
Fibroblast growth factor receptor 3
- FPKM:
-
Fragments per kilobase of transcript per million mapped reads
- GISTIC:
-
Genomic Identification of significant targets in cancer
- GZMB:
-
Granzyme B
- HER2:
-
Human epithelial growth factor receptor-2
- ICB:
-
Immune checkpoint blockade
- ICIs:
-
Immune checkpoint inhibitors
- IFN-γ:
-
Interferon-γ
- IHC:
-
Immunohistochemistry
- IL-10:
-
Interleukin 10
- MIBC:
-
Muscle-invasive bladder cancer
- mUC:
-
Metastatic urothelial carcinoma
- NMIBC:
-
Non-muscle-invasive bladder cancer
- ORR:
-
Objective response rate
- OS:
-
Overall survival
- PD-1:
-
Programmed cell death 1
- PD-L1:
-
Programmed cell death-ligand 1
- SD:
-
Stable disease
- ssGSEA:
-
Single sample gene set enrichment analysis
- TCGA:
-
The Cancer Genome Atlas
- TGF-β:
-
Transforming growth factor-β
- TIM-3:
-
T cell immunoglobulin domain and mucin domain-3
- TMA:
-
Tissue microarray
- TMB:
-
Tumor mutation burden
- TNB:
-
Tumor neoantigen burden
- ZSHS:
-
Zhongshan hospital
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Sanli O, Dobruch J, Knowles MA, Burger M, Alemozaffar M, Nielsen ME, Lotan Y (2017) Bladder cancer. Nat Rev Dis Primers 3:17022. https://doi.org/10.1038/nrdp.2017.22
Cathomas R, Lorch A, Bruins HM et al (2022) The 2021 updated european association of urology guidelines on metastatic urothelial carcinoma. Eur Urol 81:95–103. https://doi.org/10.1016/j.eururo.2021.09.026
Rosenberg JE, Hoffman-Censits J, Powles T et al (2016) Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet (London, England) 387:1909–1920. https://doi.org/10.1016/s0140-6736(16)00561-4
Damrauer JS, Hoadley KA, Chism DD et al (2014) Intrinsic subtypes of high-grade bladder cancer reflect the hallmarks of breast cancer biology. Proc Natl Acad Sci USA 111:3110–3115. https://doi.org/10.1073/pnas.1318376111
Choi W, Porten S, Kim S et al (2014) Identification of distinct basal and luminal subtypes of muscle-invasive bladder cancer with different sensitivities to frontline chemotherapy. Cancer Cell 25:152–165. https://doi.org/10.1016/j.ccr.2014.01.009
Cancer Genome Atlas Research Network (2014) Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 507:315–322. https://doi.org/10.1038/nature12965
Balar AV, Galsky MD, Rosenberg JE et al (2017) Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma: a single-arm, multicentre, phase 2 trial. Lancet (London, England) 389:67–76. https://doi.org/10.1016/s0140-6736(16)32455-2
Zhang L, Conejo-Garcia JR, Katsaros D et al (2003) Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med 348:203–213. https://doi.org/10.1056/NEJMoa020177
Cho Y, Miyamoto M, Kato K et al (2003) CD4+ and CD8+ T cells cooperate to improve prognosis of patients with esophageal squamous cell carcinoma. Can Res 63:1555–1559
Naito Y, Saito K, Shiiba K, Ohuchi A, Saigenji K, Nagura H, Ohtani H (1998) CD8+ T cells infiltrated within cancer cell nests as a prognostic factor in human colorectal cancer. Can Res 58:3491–3494
Zitvogel L, Kepp O, Kroemer G (2011) Immune parameters affecting the efficacy of chemotherapeutic regimens. Nat Rev Clin Oncol 8:151–160. https://doi.org/10.1038/nrclinonc.2010.223
Rahim MK, Okholm TLH, Jones KB et al (2023) Dynamic CD8(+) T cell responses to cancer immunotherapy in human regional lymph nodes are disrupted in metastatic lymph nodes. Cell 186:1127–43.e18. https://doi.org/10.1016/j.cell.2023.02.021
Jin K, Yu Y, Zeng H et al (2022) CD103(+)CD8(+) tissue-resident memory T cell infiltration predicts clinical outcome and adjuvant therapeutic benefit in muscle-invasive bladder cancer. Br J Cancer 126:1581–1588. https://doi.org/10.1038/s41416-022-01725-6
Liu Z, Zhou Q, Wang Z et al (2020) Intratumoral TIGIT(+) CD8(+) T-cell infiltration determines poor prognosis and immune evasion in patients with muscle-invasive bladder cancer. J Immunother Cancer. https://doi.org/10.1136/jitc-2020-000978
Huang Q, Zhou Q, Zhang H et al (2020) Identification and validation of an excellent prognosis subtype of muscle-invasive bladder cancer patients with intratumoral CXCR5(+) CD8(+) T cell abundance. Oncoimmunology 9:1810489. https://doi.org/10.1080/2162402x.2020.1810489
Witjes JA, Bruins HM, Cathomas R et al (2021) European association of urology guidelines on muscle-invasive and metastatic bladder cancer: summary of the 2020 guidelines. Eur Urol 79:82–104. https://doi.org/10.1016/j.eururo.2020.03.055
Bagaev A, Kotlov N, Nomie K et al (2021) Conserved pan-cancer microenvironment subtypes predict response to immunotherapy. Cancer Cell 39:845–65.e7. https://doi.org/10.1016/j.ccell.2021.04.014
Wherry EJ, Ha SJ, Kaech SM et al (2007) Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 27:670–684. https://doi.org/10.1016/j.immuni.2007.09.006
Ayers M, Lunceford J, Nebozhyn M et al (2017) IFN-γ-related mRNA profile predicts clinical response to PD-1 blockade. J Clin Invest 127:2930–2940. https://doi.org/10.1172/jci91190
Mariathasan S, Turley SJ, Nickles D et al (2018) TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 554:544–548. https://doi.org/10.1038/nature25501
Barbie DA, Tamayo P, Boehm JS et al (2009) Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature 462:108–112. https://doi.org/10.1038/nature08460
Mermel CH, Schumacher SE, Hill B, Meyerson ML, Beroukhim R, Getz G (2011) GISTIC2.0 facilitates sensitive and confident localization of the targets of focal somatic copy-number alteration in human cancers. Genome Biol 12:41. https://doi.org/10.1186/gb-2011-12-4-r41
Zeng H, Liu Z, Wang Z et al (2020) Intratumoral IL22-producing cells define immunoevasive subtype muscle-invasive bladder cancer with poor prognosis and superior nivolumab responses. Int J Cancer 146:542–552. https://doi.org/10.1002/ijc.32715
Dadhania V, Zhang M, Zhang L et al (2016) Meta-analysis of the luminal and basal subtypes of bladder cancer and the identification of signature immunohistochemical markers for clinical use. EBioMedicine 12:105–117. https://doi.org/10.1016/j.ebiom.2016.08.036
Xu Y, Zeng H, Jin K, Liu Z, Zhu Y, Xu L, Wang Z, Chang Y, Xu J (2022) Immunosuppressive tumor-associated macrophages expressing interlukin-10 conferred poor prognosis and therapeutic vulnerability in patients with muscle-invasive bladder cancer. J Immunother Cancer. https://doi.org/10.1136/jitc-2021-003416
Powles T, Durán I, van der Heijden MS et al (2018) Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet (London, England) 391:748–757. https://doi.org/10.1016/s0140-6736(17)33297-x
Shi R, Wang X, Wu Y et al (2022) APOBEC-mediated mutagenesis is a favorable predictor of prognosis and immunotherapy for bladder cancer patients: evidence from pan-cancer analysis and multiple databases. Theranostics 12:4181–4199. https://doi.org/10.7150/thno.73235
Goldstein JT, Berger AC, Shih J, Duke FF, Furst L, Kwiatkowski DJ, Cherniack AD, Meyerson M, Strathdee CA (2017) Genomic activation of PPARG reveals a candidate therapeutic axis in bladder cancer. Can Res 77:6987–6998. https://doi.org/10.1158/0008-5472.Can-17-1701
Rosenberg JE, O’Donnell PH, Balar AV et al (2019) Pivotal trial of enfortumab vedotin in urothelial carcinoma after platinum and anti-programmed death 1/programmed death ligand 1 therapy. J Clin Oncol 37:2592–2600. https://doi.org/10.1200/jco.19.01140
Tagawa ST, Balar AV, Petrylak DP et al (2021) TROPHY-U-01: a phase II open-label study of sacituzumab govitecan in patients with metastatic urothelial carcinoma progressing after platinum-based chemotherapy and checkpoint inhibitors. J Clin Oncol 39:2474–2485. https://doi.org/10.1200/jco.20.03489
Tsai JH, Donaher JL, Murphy DA, Chau S, Yang J (2012) Spatiotemporal regulation of epithelial-mesenchymal transition is essential for squamous cell carcinoma metastasis. Cancer Cell 22:725–736. https://doi.org/10.1016/j.ccr.2012.09.022
Powles T, Kockx M, Rodriguez-Vida A et al (2019) Clinical efficacy and biomarker analysis of neoadjuvant atezolizumab in operable urothelial carcinoma in the ABACUS trial. Nat Med 25:1706–1714. https://doi.org/10.1038/s41591-019-0628-7
van Dijk N, Gil-Jimenez A, Silina K et al (2020) Preoperative ipilimumab plus nivolumab in locoregionally advanced urothelial cancer: the NABUCCO trial. Nat Med 26:1839–1844. https://doi.org/10.1038/s41591-020-1085-z
Rozeman EA, Hoefsmit EP, Reijers ILM et al (2021) Survival and biomarker analyses from the OpACIN-neo and OpACIN neoadjuvant immunotherapy trials in stage III melanoma. Nat Med 27:256–263. https://doi.org/10.1038/s41591-020-01211-7
Ma Z, Li X, Mao Y, Wei C, Huang Z, Li G, Yin J, Liang X, Liu Z (2022) Interferon-dependent SLC14A1(+) cancer-associated fibroblasts promote cancer stemness via WNT5A in bladder cancer. Cancer Cell 40:1550–65.e7. https://doi.org/10.1016/j.ccell.2022.11.005
Spaan I, Raymakers RA, van de Stolpe A, Peperzak V (2018) Wnt signaling in multiple myeloma: a central player in disease with therapeutic potential. J Hematol Oncol 11:67. https://doi.org/10.1186/s13045-018-0615-3
Zhang M, Weng W, Zhang Q et al (2018) The lncRNA NEAT1 activates Wnt/β-catenin signaling and promotes colorectal cancer progression via interacting with DDX5. J Hematol Oncol 11:113. https://doi.org/10.1186/s13045-018-0656-7
Zhang J, Cai H, Sun L, Zhan P, Chen M, Zhang F, Ran Y, Wan J (2018) LGR5, a novel functional glioma stem cell marker, promotes EMT by activating the Wnt/β-catenin pathway and predicts poor survival of glioma patients. J Exp Clin Cancer Res CR 37:225. https://doi.org/10.1186/s13046-018-0864-6
Stewart DJ (2014) Wnt signaling pathway in non-small cell lung cancer. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djt356
Álvarez-Fernández M, Malumbres M (2020) Mechanisms of sensitivity and resistance to CDK4/6 inhibition. Cancer Cell 37:514–529. https://doi.org/10.1016/j.ccell.2020.03.010
Ingham M, Schwartz GK (2017) Cell-cycle therapeutics come of age. J Clin Oncol 35:2949–2959. https://doi.org/10.1200/jco.2016.69.0032
Acknowledgements
We thank Dr. Lingli Chen (Department of Pathology, Zhongshan Hospital, Fudan University, Shanghai, China) and Dr. Yunyi Kong (Department of Pathology, Fudan University Shanghai Cancer Center, Shanghai, China) for their excellent pathological technology help.
Funding
This study was funded by grants from National Natural Science Foundation of China (82002670, 82103408, 82272930, 82372793, 82373276), China Postdoctoral Science Foundation (BX20230091), Shanghai Municipal Natural Science Foundation (22ZR1413400, 23ZR1411700, 23ZR1440300), Shanghai Sailing Program (21YF1407000), Fudan University Shanghai Cancer Center for Outstanding Youth Scholars Foundation (YJYQ201802) and Shanghai Anticancer Association EYAS PROJECT (SACA-CY22B02, ZYJH202309). All these study sponsors have no roles in the study design, in the collection, analysis and interpretation of data.
Author information
Authors and Affiliations
Contributions
BL, KJ and ZL contributed to the acquisition of data, analysis and interpretation of data, statistical analysis and drafting of the manuscript; XS, ZX, GL, JX, HL, YC, YW and YZ provided technical and material support; ZW, LX and WZ assisted in the study concept and design, analysis and interpretation of data, drafting of the manuscript, obtaining funding and study supervision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have declared no conflicts of interest.
Ethics approval
This study was approved by the Clinical Research Ethics Committee of Zhongshan Hospital and Fudan University (No. B2015-030).
Informed consent
Written informed consent was obtained from each patient.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, B., Jin, K., Liu, Z. et al. Integrating molecular subtype and CD8+ T cells infiltration to predict treatment response and survival in muscle-invasive bladder cancer. Cancer Immunol Immunother 73, 66 (2024). https://doi.org/10.1007/s00262-024-03651-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00262-024-03651-3