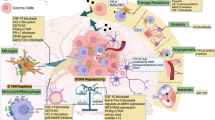
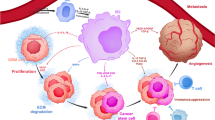
Abstract
Anti-PD-1-based therapy has resulted in a minimal clinical response in malignant gliomas. Gliomas contain numerous glioma-associated microglia/macrophages (GAMs), reported to contribute to an immunosuppressive microenvironment and promote glioma progression. However, whether and how GAMs affect anti-PD-1 immunotherapy in glioma remains unclear. Here, we demonstrated that M1-like GAMs contribute to the anti-PD-1 therapeutic response, while the accumulation of M2-like GAMs is associated with therapeutic resistance. Furthermore, we found that PD-L1 ablation reverses GAMs M2-like phenotype and is beneficial to anti-PD-1 therapy. We also demonstrated that tumor-induced impairment of the antigen-presenting function of GAMs could limit the antitumor immunity of CD4+ T cells in anti-PD-1 therapy. Our study highlights the impact of GAMs activation on anti-PD-1 treatment and provides new insights into the role of GAMs in regulating anti-PD-1 therapy in gliomas.







Similar content being viewed by others
Data availability
All data generated or analyzed in this study are included in this published article. Further details are available upon request.
Abbreviations
- APCs:
-
Antigen-presenting cells
- Arg1:
-
Arginase 1
- DAPI:
-
4′, 6-Diamidino-2-phenylindole
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- FACS:
-
Fluorescence-activated cell sorting
- GAMs:
-
Glioma-associated microglia/macrophages
- GBM:
-
Glioblastoma
- IFN-γ :
-
Interferon-γ
- iNOS:
-
Inducible nitric oxide synthase
- MCP-1:
-
Monocyte chemoattractant protein-1
- MHC-II:
-
Major histocompatibility complex class II
- OVA:
-
Ovalbumin
- PD-1:
-
Programmed cell death protein 1
- PD-L1:
-
Programmed death-ligand 1
- TME:
-
Tumor microenvironment
- TGF-β :
-
Transforming growth factor-beta
- TNF-α :
-
Tumor necrosis factor α
References
Miller KD, Ostrom QT, Kruchko C, Patil N, Tihan T, Cioffi G, Fuchs HE, Waite KA, Jemal A, Siegel RL, Barnholtz-Sloan JS (2021) Brain and other central nervous system tumor statistics, 2021. CA Cancer J Clin 71(5):381–406. https://doi.org/10.3322/caac.21693
Zou W, Wolchok JD, Chen L (2016) PD-L1 (B7–H1) and PD-1 pathway blockade for cancer therapy: mechanisms, response biomarkers, and combinations. Sci Transl Med 8(328):328rv324-328rv324. https://doi.org/10.1126/scitranslmed.aad7118
Ribas A, Wolchok JD (2018) Cancer immunotherapy using checkpoint blockade. Science (New York, N.Y.) 359(6382):1350–1355. https://doi.org/10.1126/science.aar4060
Reardon DA, Brandes AA, Omuro A, Mulholland P, Lim M, Wick A, Baehring J, Ahluwalia MS, Roth P, Bahr O, Phuphanich S, Sepulveda JM, De Souza P, Sahebjam S, Carleton M, Tatsuoka K, Taitt C, Zwirtes R, Sampson J, Weller M (2020) Effect of Nivolumab versus Bevacizumab in patients with recurrent glioblastoma: the checkmate 143 phase 3 randomized clinical trial. JAMA Oncol 6(7):1003–1010. https://doi.org/10.1001/jamaoncol.2020.1024
Wang X, Guo G, Guan H, Yu Y, Lu J, Yu J (2019) Challenges and potential of PD-1/PD-L1 checkpoint blockade immunotherapy for glioblastoma. J Exp Clin Cancer Res CR 38(1):87. https://doi.org/10.1186/s13046-019-1085-3
Lim M, Xia Y, Bettegowda C, Weller M (2018) Current state of immunotherapy for glioblastoma. Nat Rev Clin Oncol 15(7):422–442. https://doi.org/10.1038/s41571-018-0003-5
Woroniecka K, Chongsathidkiet P, Rhodin K, Kemeny H, Dechant C, Farber SH, Elsamadicy AA, Cui X, Koyama S, Jackson C, Hansen LJ, Johanns TM, Sanchez-Perez L, Chandramohan V, Yu YA, Bigner DD, Giles A, Healy P, Dranoff G, Weinhold KJ, Dunn GP, Fecci PE (2018) T-Cell exhaustion signatures vary with tumor type and are severe in Glioblastoma. Clin Cancer Res Off J Am Assoc Cancer Res 24(17):4175–4186. https://doi.org/10.1158/1078-0432.CCR-17-1846
Antonios JP, Soto H, Everson RG, Moughon D, Orpilla JR, Shin NP, Sedighim S, Treger J, Odesa S, Tucker A, Yong WH, Li G, Cloughesy TF, Liau LM, Prins RM (2017) Immunosuppressive tumor-infiltrating myeloid cells mediate adaptive immune resistance via a PD-1/PD-L1 mechanism in glioblastoma. Neuro Oncol 19(6):796–807. https://doi.org/10.1093/neuonc/now287
Nduom EK, Weller M, Heimberger AB (2015) Immunosuppressive mechanisms in glioblastoma. Neuro Oncol 17(7):9–14. https://doi.org/10.1093/neuonc/nov151
Charles NA, Holland EC, Gilbertson R, Glass R, Kettenmann H (2012) The brain tumor microenvironment. Glia 60(3):502–514. https://doi.org/10.1002/glia.21264
Hambardzumyan D, Gutmann DH, Kettenmann H (2016) The role of microglia and macrophages in glioma maintenance and progression. Nat Neurosci 19(1):20–27. https://doi.org/10.1038/nn.4185
Szulzewsky F, Pelz A, Feng X, Synowitz M, Markovic D, Langmann T, Holtman IR, Wang X, Eggen BJ, Boddeke HW, Hambardzumyan D, Wolf SA, Kettenmann H (2015) Glioma-associated microglia/macrophages display an expression profile different from M1 and M2 polarization and highly express Gpnmb and Spp1. PLoS ONE 10(2):e0116644. https://doi.org/10.1371/journal.pone.0116644
Li W, Graeber MB (2012) The molecular profile of microglia under the influence of glioma. Neuro Oncol 14(8):958–978. https://doi.org/10.1093/neuonc/nos116
Orihuela R, McPherson CA, Harry GJ (2016) Microglial M1/M2 polarization and metabolic states. Br J Pharmacol 173(4):649–665. https://doi.org/10.1111/bph.2016.173.issue-4
Yao A, Liu F, Chen K, Tang L, Liu L, Zhang K, Yu C, Bian G, Guo H, Zheng J, Cheng P, Ju G, Wang J (2014) Programmed death 1 deficiency induces the polarization of macrophages/microglia to the M1 phenotype after spinal cord injury in mice. Neurotherapeutics 11(3):636–650. https://doi.org/10.1007/s13311-013-0254-x
Roesch S, Rapp C, Dettling S, Herold-Mende C (2018) When immune cells turn bad-tumor-associated microglia/macrophages in Glioma. Int J Mol Sci 19(2):436. https://doi.org/10.3390/ijms19020436
Zhu Z, Zhang H, Chen B, Liu X, Zhang S, Zong Z, Gao M (2020) PD-L1-mediated immunosuppression in Glioblastoma is associated with the infiltration and M2-polarization of tumor-associated macrophages. Front Immunol 11:588552. https://doi.org/10.3389/fimmu.2020.588552
Miyauchi JT, Caponegro MD, Chen D, Choi MK, Li M, Tsirka SE (2018) Deletion of neuropilin 1 from microglia or bone marrow-derived macrophages slows glioma progression. Cancer Res 78(3):685–694. https://doi.org/10.1158/0008-5472.CAN-17-1435
Locarno CV, Simonelli M, Carenza C, Capucetti A, Stanzani E, Lorenzi E, Persico P, Della Bella S, Passoni L, Mavilio D, Bonecchi R, Locati M, Savino B (2019) Role of myeloid cells in the immunosuppressive microenvironment in gliomas. Immunobiology 225(1):151853. https://doi.org/10.1016/j.imbio.2019.10.002
Pinton L, Masetto E, Vettore M, Solito S, Magri S, D’Andolfi M, Del Bianco P, Lollo G, Benoit JP, Okada H, Diaz A, Della Puppa A, Mandruzzato S (2019) The immune suppressive microenvironment of human gliomas depends on the accumulation of bone marrow-derived macrophages in the center of the lesion. J Immunother Cancer 7(1):58. https://doi.org/10.1186/s40425-019-0536-x
Matias D, Balca-Silva J, da Graca GC, Wanjiru CM, Macharia LW, Nascimento CP, Roque NR, Coelho-Aguiar JM, Pereira CM, Dos Santos MF, Pessoa LS, Lima FRS, Schanaider A, Ferrer VP, Tania Cristina Leite de Sampaio e S, Moura-Neto V (2018) Microglia/astrocytes-glioblastoma crosstalk: crucial molecular mechanisms and microenvironmental factors. Front Cell Neurosci 12:235. https://doi.org/10.3389/fncel.2018.00235
Zhao J, Chen AX, Gartrell RD, Silverman AM, Aparicio L, Chu T, Bordbar D, Shan D, Samanamud J, Mahajan A, Filip I, Orenbuch R, Goetz M, Yamaguchi JT, Cloney M, Horbinski C, Lukas RV, Raizer J, Rae AI, Yuan J, Canoll P, Bruce JN, Saenger YM, Sims P, Iwamoto FM, Sonabend AM, Rabadan R (2019) Immune and genomic correlates of response to anti-PD-1 immunotherapy in glioblastoma. Nat Med 25(3):462–469. https://doi.org/10.1038/s41591-019-0349-y
Hutter G, Theruvath J, Graef CM, Zhang M, Schoen MK, Manz EM, Bennett ML, Olson A, Azad TD, Sinha R, Chan C, Assad Kahn S, Gholamin S, Wilson C, Grant G, He J, Weissman IL, Mitra SS, Cheshier SH (2019) Microglia are effector cells of CD47-SIRPalpha antiphagocytic axis disruption against glioblastoma. Proc Natl Acad Sci USA 116(3):997–1006. https://doi.org/10.1073/pnas.1721434116
Kennedy BC, Showers CR, Anderson DE, Anderson L, Canoll P, Bruce JN, Anderson RC (2013) Tumor-associated macrophages in glioma: friend or foe? J Oncol 2013:486912. https://doi.org/10.1155/2013/486912
Zuazo M, Arasanz H, Fernandez-Hinojal G, Garcia-Granda MJ, Gato M, Bocanegra A, Martinez M, Hernandez B, Teijeira L, Morilla I, Lecumberri MJ, Fernandez de Lascoiti A, Vera R, Kochan G, Escors D (2019) Functional systemic CD4 immunity is required for clinical responses to PD-L1/PD-1 blockade therapy. EMBO Mol Med 11(7):e10293. https://doi.org/10.15252/emmm.201910293
Alspach E, Lussier DM, Miceli AP, Kizhvatov I, DuPage M, Luoma AM, Meng W, Lichti CF, Esaulova E, Vomund AN, Runci D, Ward JP, Gubin MM, Medrano RFV, Arthur CD, White JM, Sheehan KCF, Chen A, Wucherpfennig KW, Jacks T, Unanue ER, Artyomov MN, Schreiber RD (2019) MHC-II neoantigens shape tumour immunity and response to immunotherapy. Nature 574(7780):696–701. https://doi.org/10.1038/s41586-019-1671-8
Dong H, Zhu G, Tamada K, Flies D, van Deursen J, Chen L (2004) B7–H1 determines accumulation and deletion of intrahepatic CD8(+) T lymphocytes. Immunity 20(3):327–336. https://doi.org/10.1016/s1074-7613(04)00050-0
Lu S, Stein JE, Rimm DL, Wang DW, Bell JM, Johnson DB, Sosman JA, Schalper KA, Anders RA, Wang H, Hoyt C, Pardoll DM, Danilova L, Taube JM (2019) Comparison of biomarker modalities for predicting response to PD-1/PD-L1 checkpoint blockade: a systematic review and meta-analysis. JAMA Oncol 5(8):1195–1204. https://doi.org/10.1001/jamaoncol.2019.1549
Haabeth OAW, Fauskanger M, Manzke M, Lundin KU, Corthay A, Bogen B, Tveita AA (2018) CD4(+) T-cell-mediated rejection of MHC class II-positive tumor cells is dependent on antigen secretion and indirect presentation on host APCs. Cancer Res 78(16):4573–4585. https://doi.org/10.1158/0008-5472.CAN-17-2426
de Groot J, Penas-Prado M, Alfaro-Munoz KD, Hunter K, Pei B, O’Brien B, Weathers SP, Loghin M, Kamiya Matsouka C, Yung WKA, Mandel J, Wu J, Yuan Y, Zhou S, Fuller GN, Huse J, Rao G, Weinberg JS, Prabhu SS, McCutcheon IE, Lang FF, Ferguson SD, Sawaya R, Colen R, Yadav SS, Blando J, Vence L, Allison J, Sharma P, Heimberger AB (2019) Window-of-opportunity clinical trial of pembrolizumab in patients with recurrent glioblastoma reveals predominance of immune-suppressive macrophages. Neuro Oncol 22(4):539–549. https://doi.org/10.1093/neuonc/noz185
Chocarro de Erauso L, Zuazo M, Arasanz H, Bocanegra A, Hernandez C, Fernandez G, Garcia-Granda MJ, Blanco E, Vera R, Kochan G, Escors D (2020) Resistance to PD-L1/PD-1 Blockade Immunotherapy. A tumor-intrinsic or tumor-extrinsic phenomenon? Front Pharmacol 11:441. https://doi.org/10.3389/fphar.2020.00441
Sarkar S, Döring A, Zemp FJ, Silva C, Lun X, Wang X, Kelly J, Hader W, Hamilton M, Mercier P, Dunn JF, Kinniburgh D, van Rooijen N, Robbins S, Forsyth P, Cairncross G, Weiss S, Yong VW (2013) Therapeutic activation of macrophages and microglia to suppress brain tumor-initiating cells. Nat Neurosci 17(1):46–55. https://doi.org/10.1038/nn.3597
Rao G, Latha K, Ott M, Sabbagh A, Marisetty A, Ling X, Zamler D, Doucette TA, Yang Y, Kong LY, Wei J, Fuller GN, Benavides F, Sonabend AM, Long J, Li S, Curran M, Heimberger AB (2020) Anti-PD-1 induces M1 polarization in the glioma microenvironment and exerts therapeutic efficacy in the absence of CD8 cytotoxic T cells. Clin Cancer Res Off J Am Assoc Cancer Res 26(17):4699–4712. https://doi.org/10.1158/1078-0432.CCR-19-4110
Lin H, Wei S, Hurt EM, Green MD, Zhao L, Vatan L, Szeliga W, Herbst R, Harms PW, Fecher LA, Vats P, Chinnaiyan AM, Lao CD, Lawrence TS, Wicha M, Hamanishi J, Mandai M, Kryczek I, Zou W (2018) Host expression of PD-L1 determines efficacy of PD-L1 pathway blockade-mediated tumor regression. J Clin Investig 128(2):805–815. https://doi.org/10.1172/JCI96113
Bloch O, Crane CA, Kaur R, Safaee M, Rutkowski MJ, Parsa AT (2013) Gliomas promote immunosuppression through Induction of B7–H1 expression in tumor-associated macrophages. Clin Cancer Res 19(12):3165–3175. https://doi.org/10.1158/1078-0432.ccr-12-3314
Curiel TJ, Wei S, Dong H, Alvarez X, Cheng P, Mottram P, Krzysiek R, Knutson KL, Daniel B, Zimmermann MC, David O, Burow M, Gordon A, Dhurandhar N, Myers L, Berggren R, Hemminki A, Alvarez RD, Emilie D, Curiel DT, Chen L, Zou W (2003) Blockade of B7–H1 improves myeloid dendritic cell-mediated antitumor immunity. Nat Med 9(5):562–567. https://doi.org/10.1038/nm863
Diskin B, Adam S, Cassini MF, Sanchez G, Liria M, Aykut B, Buttar C, Li E, Sundberg B, Salas RD, Chen R, Wang J, Kim M, Farooq MS, Nguy S, Fedele C, Tang KH, Chen T, Wang W, Hundeyin M, Rossi JAK, Kurz E, Haq MIU, Karlen J, Kruger E, Sekendiz Z, Wu D, Shadaloey SAA, Baptiste G, Werba G, Selvaraj S, Loomis C, Wong KK, Leinwand J, Miller G (2020) PD-L1 engagement on T cells promotes self-tolerance and suppression of neighboring macrophages and effector T cells in cancer. Nat Immunol 21(4):442–454. https://doi.org/10.1038/s41590-020-0620-x
Tran E, Turcotte S, Gros A, Robbins PF, Lu YC, Dudley ME, Wunderlich JR, Somerville RP, Hogan K, Hinrichs CS, Parkhurst MR, Yang JC, Rosenberg SA (2014) Cancer immunotherapy based on mutation-specific CD4+ T cells in a patient with epithelial cancer. Science (New York, N.Y.) 344(6184):641–645. https://doi.org/10.1126/science.1251102
Kreiter S, Vormehr M, van de Roemer N, Diken M, Lower M, Diekmann J, Boegel S, Schrors B, Vascotto F, Castle JC, Tadmor AD, Schoenberger SP, Huber C, Tureci O, Sahin U (2015) Mutant MHC class II epitopes drive therapeutic immune responses to cancer. Nature 520(7549):692–696. https://doi.org/10.1038/nature14426
Haabeth OA, Tveita AA, Fauskanger M, Schjesvold F, Lorvik KB, Hofgaard PO, Omholt H, Munthe LA, Dembic Z, Corthay A, Bogen B (2014) How Do CD4(+) T cells detect and eliminate tumor cells that either lack or express MHC class II molecules? Front Immunol 5:174. https://doi.org/10.3389/fimmu.2014.00174
Corthay A, Skovseth DK, Lundin KU, Rosjo E, Omholt H, Hofgaard PO, Haraldsen G, Bogen B (2005) Primary antitumor immune response mediated by CD4+ T cells. Immunity 22(3):371–383. https://doi.org/10.1016/j.immuni.2005.02.003
Ulvestad E, Williams K, Bjerkvig R, Tiekotter K, Antel J, Matre R (1994) Human microglial cells have phenotypic and functional characteristics in common with both macrophages and dendritic antigen-presenting cells. J Leukoc Biol 56(6):732–740. https://doi.org/10.1002/jlb.56.6.732
Tran C, Wolz P, Egensperger R, Kösel S, Imai Y, Bise K, Kohsaka S, Mehraein P, Graeber M (1998) Differential expression of MHC class II molecules by microglia and neoplastic astroglia: relevance for the escape of astrocytoma cells from immune surveillance. Neuropathol Appl Neurobiol 244(4):293–301. https://doi.org/10.1046/j.1365-2990.1998.00120.x
Tritz ZP, Ayasoufi K, Johnson AJ (2021) Anti-PD-1 checkpoint blockade monotherapy in the orthotopic GL261 glioma model: the devil is in the detail. Neuro-Oncol Adv 3(1):1–9. https://doi.org/10.1093/noajnl/vdab066
Acknowledgements
We thank Cailing Yan at the Public Technology Service Center of Fujian Medical University for technical assistance with the bioluminescence imaging. This work was supported by the Natural Science Foundation of Fujian Province (No. 2019J01464) and the Joint Funds for the Innovation of Science and Technology, Fujian Province (No. 2018Y9053).
Author information
Authors and Affiliations
Contributions
CW and LL conceived and designed the experiments. CW and QC performed the experiments and collected and analyzed the data. MC cooperated with the establishment of tumor models. SG prepared the experimental materials and bred and identified gene knockout mice. PH and YZ isolated and purified the blocking antibodies. JW and BH provided technical guidance. CW wrote the first draft of the manuscript, and LL, QZ, and LC supervised the study and commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving animals were approved by the Fujian Medical University Institutional Animal Care and Use Committee (IACUC, No. 2019-0114) in accordance with ethical standards. All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, C., Chen, Q., Chen, M. et al. Interaction of glioma-associated microglia/macrophages and anti-PD1 immunotherapy. Cancer Immunol Immunother 72, 1685–1698 (2023). https://doi.org/10.1007/s00262-022-03358-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-022-03358-3