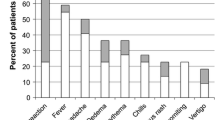
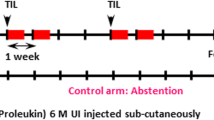
Abstract
Tumor immune escape has recently been shown to be related to the development of an immune tolerance state of the microenvironment. Cytokines activating the immune system such as IFN-γ can be used to reverse the immune escape and thus to potentiate the efficacy of immunotherapy. A clinical study was conducted in 18 stage IIIc/IV melanoma patients treated with tumor-infiltrating lymphocytes (TILs) in combination with intratumoral TG1042 injection (adenovirus expressing IFN-γ). The primary objective was to investigate the safety of treatment. Secondary objectives were to study the clinical response and translational research. The treatment was well tolerated. Among the 13 patients evaluable for tumor response, 38.5 % had an overall objective response (OOR = CR + PR) and disease control rate (DCR = CR + PR + S) of 46 %. The clinical response of the 37 targeted lesions led to an OOR of 51 % and a DCR of 75 %. Translational research on predictive markers did not significantly differ between responder and non-responder patients. However, specifically regarding injected lesions, the clinical response correlated with CD3−/CD56+ NK cells which could be activated by TG1042. Further larger studies of this combined immunotherapy are needed to confirm our findings. Intralesional TG1042 combined with antigen-selected TILs should be discussed.


Similar content being viewed by others
Abbreviations
- ACT:
-
Adoptive cell therapy
- AJCC:
-
American Joint Committee of Cancer
- APC:
-
Allophycocyanin
- CR:
-
Complete regression
- FACS:
-
Fluorescence-activated cell sorting
- FITC:
-
Fluorescein isothiocyanate
- H&E:
-
Hematoxylin and eosin
- IFN-γ:
-
Interferon-gamma
- IL:
-
Interleukin
- MAGE:
-
Melanoma-associated antigen
- NCI-CTC:
-
National Cancer Institute Common Terminology Criteria
- NK:
-
Natural killer
- NY-ESO-1:
-
New York esophageal squamous cell carcinoma
- P:
-
Progression
- PE:
-
Phycoerythrin
- PR:
-
Partial regression
- SD:
-
Stable disease
- TIL:
-
Tumor-infiltrating lymphocytes
References
Restifo NP, Dudley ME, Rosenberg SA (2012) Adoptive immunotherapy for cancer: harnessing the T cell response. Nat Rev Immunol 12:269–281
Kirkwood JM, Ernstoff MS (1990) Role of interferons in the therapy of melanoma. J Invest Dermatol 95:180S–184S
Nemunaitis J, Fong T, Robbins JM, Edelman G, Edwards W, Paulson RS, Bruce J, Ognoskie N, Wynne D, Pike M, Kowal K, Merritt J, Ando D (1999) Phase I trial of interferon-gamma (IFN-gamma) retroviral vector administered intratumorally to patients with metastatic melanoma. Cancer Gene Ther 6:322–330
Khorana AA, Rosenblatt JD, Sahasrabudhe DM, Evans T, Landrigan M, Marquis D, Rosell K, Whiteside T, Phillippe S, Acres B, Slos P, Squiban P, Ross M, Kendra K (2003) A phase I trial of immunotherapy with intratumoral adenovirus-interferon-gamma (TG1041) in patients with malignant melanoma. Cancer Gene Ther 10:251–259
Whittington R, Faulds D (1993) Interleukin-2. A review of its pharmacological properties and therapeutic use in patients with cancer. Drugs 46:446–514
Bachmann MF, Oxenius A (2007) Interleukin 2: from immunostimulation to immunoregulation and back again. EMBO Rep 8:1142–1148
Donia M, Hansen M, Sendrup SL, Iversen TZ, Ellebæk E, Andersen MH, Straten PT, Svane IM (2013) Methods to improve adoptive T-cell therapy for melanoma: IFN-γ enhances anticancer responses of cell products for infusion. J Invest Dermatol 133:545–552
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Jotereau F, Pandolfino MC, Boudart D, Diez E, Dreno B, Douillard JY, Muller JY, LeMevel B (1991) High-fold expansion of human cytotoxic T-lymphocytes specific for autologous melanoma cells for use in immunotherapy. J Immunother 10:405–411
Pandolfino MC, Labarriere N, Tessier MH, Cassidanius A, Bercegeay S, Lemarre P, Dehaut F, Dreno B, Jotereau F (2001) High-scale expansion of melanoma-reactive TIL by a polyclonal stimulus: predictability and relation with disease advancement. Cancer Immunol Immunother 50:134–140
Chebassier N, El Houssein O, Viegas I, Dreno B (2004) In vitro induction of matrix metalloproteinase-2 and matrix metalloproteinase-9 expression in keratinocytes by boron and manganese. Exp Dermatol 13:484–490
Jarry A, Masson D, Cassagnau E, Parois S, Laboisse C, Denis MG (2004) Real-time allele-specific amplification for sensitive detection of the BRAF mutation V600E. Mol Cell Probes 18:349–352
Gervois N, Heuze F, Diez E, Jotereau F (1990) Selective expansion of a specific anti-tumor CD8+ cytotoxic T lymphocyte clone in the bulk culture of tumor-infiltrating lymphocytes from a melanoma patient: cytotoxic activity and T cell receptor gene rearrangements. Eur J Immunol 20:825–831
Pandolfino MC, Saiagh S, Knol AC, Dreno B (2010) Comparison of three culture media for the establishment of melanoma cell lines. Cytotechnology 62:403–412
Jung T, Schauer U, Heusser C, Neumann C, Rieger C (1993) Detection of intracellular cytokines by flow cytometry. J Immunol Methods 159:197–207
Yang JC (2013) The adoptive transfer of cultured T cells for patients with metastatic melanoma. Clin Dermatol 31:209–219
Khammari A, Knol AC, Nguyen JM, Bossard C, Denis MG, Pandolfino MC, Quéreux G, Bercegeay S, Dréno B (2014) Adoptive TIL transfer in the adjuvant setting for melanoma: long-term patient survival. J Immunol Res 2014:186212. doi:10.1155/2014/186212
Pt Straten, Becker JC (2009) Adoptive cell transfer in the treatment of metastatic melanoma. J Invest Dermatol 129:2743–2745
Dudley ME (2011) Adoptive cell therapy for patients with melanoma. J Cancer 2:360–362
Dreno B, Nguyen JM, Khammari A, Pandolfino MC, Tessier MH, Bercegeay S, Cassidanius A, Lemarre P, Billaudel S, Labarriere N, Jotereau F (2002) Randomized trial of adoptive transfer of melanoma tumor-infiltrating lymphocytes as adjuvant therapy for stage III melanoma. Cancer Immunol Immunother 51:539–546
Dummer R, Hassel JC, Fellenberg F, Eichmuller S, Maier T, Slos P, Acres B, Bleuzen P, Bataille V, Squiban P, Burg G, Urosevic M (2004) Adenovirus-mediated intralesional interferon-gamma gene transfer induces tumor regressions in cutaneous lymphomas. Blood 104:1631–1638
Kalinski P, Giermasz A, Nakamura Y, Basse P, Storkus WJ, Kirkwood JM, Mailliard RB (2005) Helper role of NK cells during the induction of anticancer responses by dendritic cells. Mol Immunol 42:535–539
Hayakawa K, Salmeron MA, Parkinson DR, Markowitz AB, von Eschenbach AC, Legha SS, Balch CM, Ross MI, Augustus LB, Itoh K (1991) Study of tumor infiltrating lymphocytes for adoptive therapy of renal cell carcinoma (RCC) and metastatic melanoma: sequential proliferation of cytotoxic natural killer and noncytotoxic T cells in RCC. J Immunother 10:313–325
Kirkwood JM, Bryant J, Schiller JH, Oken MM, Borden EC, Whiteside TL (1997) Immunomodulatory function of interferon-gamma in patients with metastatic melanoma: results of a phase II-B trial in subjects with metastatic melanoma, ECOG study E 4987. Eastern Cooperative Oncology Group. J Immunother 20:146–157
Gritzapis AD, Dimitroulopoulos D, Paraskevas E, Baxevanis CN, Papamichail M (2002) Large-scale expansion of CD3(+)CD56(+) lymphocytes capable of lysing autologous tumor cells with cytokine-rich supernatants. Cancer Immunol Immunother 51:440–448
Ibarrondo FJ, Yang OO, Chodon T, Avramis E, Lee Y, Sazegar H, Jalil J, Chmielowski B, Koya RC, Schmid I, Gomez-Navarro J, Jamieson BD, Ribas A, Comin-Anduix B (2013) Natural killer T cells in advanced melanoma patients treated with tremelimumab. PLoS One 8:e76829
Mastrangelo MJ, Maguire HC, Eisenlohr LC, Laughlin CE, Monken CE, McCue PA, Kovatich AJ, Lattime EC (1999) Intratumoral recombinant GM-CSF-encoding virus as gene therapy in patients with cutaneous melanoma. Cancer Gene Ther 6:409–422
Senzer NN, Kaufman HL, Amatruda T, Nemunaitis M, Reid T, Daniels G, Gonzalez R, Glaspy J, Whitman E, Harrington K, Goldsweig H, Marshall T, Love C, Coffin R, Nemunaitis JJ (2009) Phase II clinical trial of a granulocyte-macrophage colony-stimulating factor-encoding, second-generation oncolytic herpesvirus in patients with unresectable metastatic melanoma. J Clin Oncol 27:5763–5771
Dummer R, Rochlitz C, Velu T, Acres B, Limacher JM, Bleuzen P, Lacoste G, Slos P, Romero P, Urosevic M (2008) Intralesional adenovirus-mediated interleukin-2 gene transfer for advanced solid cancers and melanoma. Mol Ther 16:985–994
Labarriere N, Fortun A, Bellec A, Khammari A, Dreno B, Saïagh S, Lang F (2013) A full GMP process to select and amplify epitope-specific T lymphocytes for adoptive immunotherapy of metastatic melanoma. Clin Dev Immunol 2013:932318. doi:10.1155/2013/932318
Acknowledgments
We thank M. Yviquel, G. Hatton and J. David from the laboratory of immunodermatology and also the laboratory of immunology from Nantes Hospital for their technical support. This study was partially funded by Transgene and Nantes Hospital.
Conflict of interest
V. Bataille and J.M. Limacher are employees of Transgene SA. Other authors have no conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khammari, A., Nguyen, JM., Saint-Jean, M. et al. Adoptive T cell therapy combined with intralesional administrations of TG1042 (adenovirus expressing interferon-γ) in metastatic melanoma patients. Cancer Immunol Immunother 64, 805–815 (2015). https://doi.org/10.1007/s00262-015-1691-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-015-1691-7