Abstract
Purpose
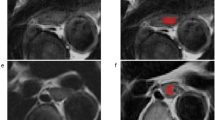
Accurate prediction of prognosis and pathological response to neoadjuvant chemotherapy (NAC) is crucial for optimizing treatment strategies for patients with locally advanced esophageal cancer (LA-EC). This study aimed to investigate the use of radiomics for pretreatment CT in predicting the pathological response of patients with LA-EC to NAC.
Methods
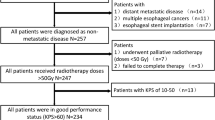
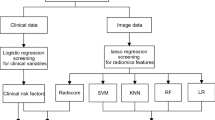
Overall, 144 patients (145 lesions) with LA-EC who underwent pretreatment contrast-enhanced CT and then received NAC followed by surgery with pathological tumor regression grade (TRG) analysis were enrolled. The obtained dataset was randomly divided into training and validation cohorts using fivefold cross-validation. CT-based radiomic features were extracted followed by the feature selection process using the variance threshold, SelectKBest, and least absolute shrinkage and selection operator methods. The radiomic model was constructed using six machine learning classifiers, and predictive performance was evaluated using ROC curve analysis in the training and validation cohorts.
Results
All patients were divided into responders (n = 40, 28%) and non-responders (n = 104, 72%) based on the TRG results and a statistically significant split by overall survival analysis (0.899 [0.754–0.961] vs. 0.630 [0.510–0.729], respectively). There were no significant differences between responders and non-responders in terms of age, sex, tumor size, tumor location, or histopathology. The mean AUC of fivefold in the validation cohort was 0.720 (confidence interval [CI]: 0.594–0.982), and the best AUC of the radiomic model using logistic regression to predict the non-responders was 0.815 (CI: 0.626–1.000, sensitivity 0.620, specificity 0.860).
Conclusion
A radiomic model derived from contrast-enhanced CT may help stratify chemotherapy effect prediction and improve clinical decision-making.
Graphical Abstract






Similar content being viewed by others
References
Sung H, Ferlay J, Siegel R L, Laersanne M, Soerjomataram I, et al. (2021) Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 71:209–249. https://doi.org/https://doi.org/10.3322/caac.21660
Cunningham D, Allum W H, Stenning S P, Thompson J N, Van de Velde C J H, et al. (2006) Perioperative Chemotherapy versus Surgery Alone for Resectable Gastroesophageal Cancer. N Engl J Med 355:11–20. https://doi.org/https://doi.org/10.1056/NEJMoa055531
Matsuda T, Ajiki W, Marugame T, Ioka A, Tsukuma H & Sobue T (2011) Population-based Survival of Cancer Patients Diagnosed Between 1993 and 1999 in Japan: A Chronological and International Comparative Study. Jpn J Clin Oncol 41:40–51. https://doi.org/https://doi.org/10.1093/jjco/hyq167
Siegel R L, Miller K D, Fuchs H E & Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72:7–33. https://doi.org/https://doi.org/10.3322/caac.21708
Watanabe M, Toh Y, Ishihara R, Kono K, Matsubara H, et al. (2022) Comprehensive registry of esophageal cancer in Japan, 2014. Esophagus 19:1–26. https://doi.org/https://doi.org/10.1007/s10388-021-00879-1
Ando N, Kato H, Igaki H, Shinoda M, Ozawa S, et al. (2012) A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol 19:68–74. https://doi.org/https://doi.org/10.1245/s10434-011-2049-9
Al-Batran S-E, Hofheinz R D, Pauligk C, Kopp H-G, Haag G M, et al. (2016) Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO): results from the phase 2 part of a multicentre, open-label, randomised phase 2/3 trial. Lancet Oncol 17:1697–1708. https://doi.org/https://doi.org/10.1016/S1470-2045(16)30531-9
Allum W H, Stenning S P, Bancewicz J, Clark P I & Langley R E (2009) Long-term results of a randomized trial of surgery with or without preoperative chemotherapy in esophageal cancer. J Clin Oncol 27:5062–5067. https://doi.org/https://doi.org/10.1200/JCO.2009.22.2083
Ajani J A, Winter K, Okawara G S, Donohue J H, Pisters P W T, et al. (2006) Phase II Trial of Preoperative Chemoradiation in Patients With Localized Gastric Adenocarcinoma (RTOG 9904): Quality of Combined Modality Therapy and Pathologic Response. J Clin Oncol 24:3953–3958. https://doi.org/https://doi.org/10.1200/JCO.2006.06.4840
Mayanagi S, Irino T, Kawakubo H & Kitagawa Y (2019) Neoadjuvant treatment strategy for locally advanced thoracic esophageal cancer. Ann Gastroenterol Surg 3:269–275. https://doi.org/https://doi.org/10.1002/ags3.12243
Ajani J A, Barthel J S, Bentrem D J, D’Amico T A, Das P, et al. (2011) Esophageal and Esophagogastric Junction Cancers. J Natl Compr Canc Netw 830–887. https://doi.org/10.6004/jnccn.2011.0072
Lordick F, Mariette C, Haustermans K, Obermannová R & Arnold D (2016) Oesophageal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 27:v50–v57. https://doi.org/https://doi.org/10.1093/annonc/mdw329
Shapiro J, Van Lanschot J J B, Hulshof M C, van Hagen P, van Berge Henegouwen M I, et al. (2015) Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol 16:1090–1098.
Burmeister B H, Smithers B M, Gebski V, Fitzgerald L, Simes R J, et al. (2005) Surgery alone versus chemoradiotherapy followed by surgery for resectable cancer of the oesophagus: a randomised controlled phase III trial. Lancet Oncol 6:659–668. https://doi.org/https://doi.org/10.1016/S1470-2045(05)70288-6
Kato K, Ito Y, Daiko H, Ozawa S, Ogata T, et al. (2022) A randomized controlled phase III trial comparing two chemotherapy regimen and chemoradiotherapy regimen as neoadjuvant treatment for locally advanced esophageal cancer, JCOG1109 NExT study. J Clin Oncol 40:238–238. https://doi.org/https://doi.org/10.1200/JCO.2022.40.4_suppl.238
Meredith K L, Weber J M, Turaga K K, Siegel E M, McLoughlin J, et al. (2010) Pathologic Response after Neoadjuvant Therapy is the Major Determinant of Survival in Patients with Esophageal Cancer. Ann Surg Oncol 17:1159–1167. https://doi.org/https://doi.org/10.1245/s10434-009-0862-1
Ancona E, Ruol A, Santi S, Merigliano S, Chiarion Sileni V, et al. (2001) Only pathologic complete response to neoadjuvant chemotherapy improves significantly the long term survival of patients with resectable esophageal squamous cell carcinoma. Cancer 91:2165–2174. https://acsjournals.onlinelibrary.wiley.com/doi/full/10.1002/1097-0142%2820010601%2991%3A11%3C2165%3A%3AAID-CNCR1245%3E3.0.CO%3B2-H
Li Y, Huang H-C, Chen L-Q, Xu L-Y, Li E-M & Zhang J-J (2017) Predictive biomarkers for response of esophageal cancer to chemo(radio)therapy: A systematic review and meta-analysis. Surg Oncol 26:460–472. https://doi.org/https://doi.org/10.1016/j.suronc.2017.09.003
Tomasello G, Petrelli F, Ghidini M, Pezzica E, Passalacqua R, et al. (2017) Tumor regression grade and survival after neoadjuvant treatment in gastro-esophageal cancer: A meta-analysis of 17 published studies. Eur J Surg Oncol 43:1607–1616. https://doi.org/https://doi.org/10.1016/j.ejso.2017.03.001
Lin J-W, Hsu C-P, Yeh H-L, Chuang C-Y & Lin C-H (2018) The impact of pathological complete response after neoadjuvant chemoradiotherapy in locally advanced squamous cell carcinoma of esophagus. J Chin Med Assoc 81:18–24. https://doi.org/https://doi.org/10.1016/j.jcma.2017.08.007
Hatogai K, Fujii S, Kojima T, Daiko H, Kadota T, et al. (2016) Prognostic significance of tumor regression grade for patients with esophageal squamous cell carcinoma after neoadjuvant chemotherapy followed by surgery: Impact of Tumor Regression on Survival. J Surg Oncol 113:390–396. https://doi.org/https://doi.org/10.1002/jso.24151
Kadota T, Hatogai K, Yano T, Fujita T, Kojima T, et al. (2018) Pathological tumor regression grade of metastatic tumors in lymph node predicts prognosis in esophageal cancer patients. Cancer Sci 109:2046–2055. https://doi.org/https://doi.org/10.1111/cas.13596
Donahue J M, Nichols F C, Li Z, Schomas D A, Allen M S, et al. (2009) Complete pathologic response after neoadjuvant chemoradiotherapy for esophageal cancer is associated with enhanced survival. Ann Thorac Surg 87:392–398; discussion 398–399. https://doi.org/10.1016/j.athoracsur.2008.11.001
Alfieri R, Pintacuda G, Cagol M, Occhipinti T, Capraro I, et al. (2015) Oesophageal cancer: assessment of tumour response to chemoradiotherapy with tridimensional CT. Radiol med 120:430–439. https://doi.org/https://doi.org/10.1007/s11547-014-0466-0
Kukar M, Alnaji R M, Jabi F, Platz T A, Attwood K, et al. (2015) Role of Repeat 18F-Fluorodeoxyglucose Positron Emission Tomography Examination in Predicting Pathologic Response Following Neoadjuvant Chemoradiotherapy for Esophageal Adenocarcinoma. JAMA Sur 150:555–562. https://doi.org/https://doi.org/10.1001/jamasurg.2014.3867
Rizk N P, Tang L, Adusumilli P S, Bains M S, Akhurst T J, et al. (2009) Predictive value of initial PET-SUVmax in patients with locally advanced esophageal and gastroesophageal junction adenocarcinoma. J Thorac Oncol 4:875–879. https://doi.org/https://doi.org/10.1097/JTO.0b013e3181a8cebf
Schmidt M, Bollschweiler E, Dietlein M, Mönig S P, Kobe C, et al. (2009) Mean and maximum standardized uptake values in [18F]FDG-PET for assessment of histopathological response in oesophageal squamous cell carcinoma or adenocarcinoma after radiochemotherapy. Eur J Nucl Med Mol Imaging 36:735–744. https://doi.org/https://doi.org/10.1007/s00259-008-1011-y
Piessen G, Petyt G, Duhamel A, Mirabel X, Huglo D & Mariette C (2013) Ineffectiveness of 18F-fluorodeoxyglucose positron emission tomography in the evaluation of tumor response after completion of neoadjuvant chemoradiation in esophageal cancer. Ann Surg 258:66–76. https://doi.org/https://doi.org/10.1097/SLA.0b013e31828676c4
Sah B-R, Owczarczyk K, Siddique M, Cook G J R & Goh V (2019) Radiomics in esophageal and gastric cancer. Abdom Radiol (NY) 44:2048–2058. https://doi.org/https://doi.org/10.1007/s00261-018-1724-8
van Rossum P S N, Xu C, Fried D V, Goense L, Court L E & Lin S H (2016) The emerging field of radiomics in esophageal cancer: current evidence and future potential. Transl Cancer Res 5:410–423. https://doi.org/https://doi.org/10.21037/tcr.2016.06.19
Erickson B J, Korfiatis P, Akkus Z & Kline T L (2017) Machine Learning for Medical Imaging. RadioGraphics 37:505–515. https://doi.org/https://doi.org/10.1148/rg.2017160130
Liu Z, Wang S, Dong D, Wei J, Fang C, et al. (2019) The Applications of Radiomics in Precision Diagnosis and Treatment of Oncology: Opportunities and Challenges. Theranostics 9:1303–1322. https://doi.org/https://doi.org/10.7150/thno.30309
Hatt M, Majdoub M, Vallières M, Tixier F, Le Rest C C, et al. (2015) 18F-FDG PET uptake characterization through texture analysis: investigating the complementary nature of heterogeneity and functional tumor volume in a multi-cancer site patient cohort. J Nucl Med 56:38–44. https://doi.org/https://doi.org/10.2967/jnumed.114.144055
Japan Esophageal Society (2017) Japanese Classification of Esophageal Cancer, 11th Edition: part I. Esophagus 14:1–36. https://doi.org/https://doi.org/10.1007/s10388-016-0551-7
Hastie T, Tibshirani R & Friedman J (Springer, 2009.) The Elements of Statistical Learning: Data Mining, Inference, and Prediction, Second Edition.
Lambin P, Rios-Velazquez E, Leijenaar R, Carvalho S, Stiphout R G P M van, et al. (2012) Radiomics: Extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446. https://doi.org/https://doi.org/10.1016/j.ejca.2011.11.036
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 48:452–458. https://doi.org/https://doi.org/10.1038/bmt.2012.244
Kao Y-S & Hsu Y (2021) A Meta-Analysis for Using Radiomics to Predict Complete Pathological Response in Esophageal Cancer Patients Receiving Neoadjuvant Chemoradiation. In Vivo 35:1857–1863. https://doi.org/https://doi.org/10.21873/invivo.12448
Hu Y, Xie C, Yang H, Ho J W K, Wen J, et al. (2021) Computed tomography-based deep-learning prediction of neoadjuvant chemoradiotherapy treatment response in esophageal squamous cell carcinoma. Radiother Oncol 154:6–13. https://doi.org/https://doi.org/10.1016/j.radonc.2020.09.014
Hu Y, Xie C, Yang H, Ho J W K, Wen J, et al. (2020) Assessment of Intratumoral and Peritumoral Computed Tomography Radiomics for Predicting Pathological Complete Response to Neoadjuvant Chemoradiation in Patients With Esophageal Squamous Cell Carcinoma. JAMA Netw Open 3:e2015927. https://doi.org/https://doi.org/10.1001/jamanetworkopen.2020.15927
Yang Z, He B, Zhuang X, Gao X, Wang D, et al. (2019) CT-based radiomic signatures for prediction of pathologic complete response in esophageal squamous cell carcinoma after neoadjuvant chemoradiotherapy. J Radiat Res https://doi.org/https://doi.org/10.1093/jrr/rrz027
Tixier F, Rest C C L, Hatt M, Albarghach N, Pradier O, et al. (2011) Intratumor Heterogeneity Characterized by Textural Features on Baseline 18F-FDG PET Images Predicts Response to Concomitant Radiochemotherapy in Esophageal Cancer. J Nucl Med 52:369–378. https://doi.org/https://doi.org/10.2967/jnumed.110.082404
Hirata A, Hayano K, Ohira G, Imanishi S, Hanaoka T, et al. (2020) Volumetric histogram analysis of apparent diffusion coefficient for predicting pathological complete response and survival in esophageal cancer patients treated with chemoradiotherapy. Am J Surg 219:1024–1029. https://doi.org/https://doi.org/10.1016/j.amjsurg.2019.07.040
Jin X, Zheng X, Chen D, Jin J, Zhu G, et al. (2019) Prediction of response after chemoradiation for esophageal cancer using a combination of dosimetry and CT radiomics. Eur Radiol 29:6080–6088. https://doi.org/https://doi.org/10.1007/s00330-019-06193-w
Hou Z, Ren W, Li S, Liu J, Sun Y, et al. (2017) Radiomic analysis in contrast-enhanced CT: predict treatment response to chemoradiotherapy in esophageal carcinoma. Oncotarget 8:104444-104454. https://doi.org/https://doi.org/10.18632/oncotarget.22304
Yip C, Davnall F, Kozarski R, Landau D B, Cook G J R, et al. (2015) Assessment of changes in tumor heterogeneity following neoadjuvant chemotherapy in primary esophageal cancer. Dis Esophagus 28:172–179.
Qu J, Ma L, Lu Y, Wang Z, Guo J, et al. (2022) DCE-MRI radiomics nomogram can predict response to neoadjuvant chemotherapy in esophageal cancer. Discov Onc 13:3. https://doi.org/https://doi.org/10.1007/s12672-022-00464-7
Zhang D, Zhang W, Liu W, Mao Y, Fu Z, et al. (2017) Human papillomavirus infection increases the chemoradiation response of esophageal squamous cell carcinoma based on P53 mutation. Radiother Oncol 124:155–160. https://doi.org/https://doi.org/10.1016/j.radonc.2017.06.008
Kumar R, Ghosh S K, Verma A K, Talukdar A, Deka M K, et al. (2015) p16 Expression as a Surrogate Marker for HPV Infection in Esophageal Squamous Cell Carcinoma can Predict Response to Neo-Adjuvant Chemotherapy. Asian Pac J Cancer Prev 16:7161–7165. https://doi.org/https://doi.org/10.7314/APJCP.2015.16.16.7161
Beukinga R J, Hulshoff J B, Mul V E M, Noordzij W, Kats-Ugurlu G, et al. (2018) Prediction of Response to Neoadjuvant Chemotherapy and Radiation Therapy with Baseline and Restaging 18F-FDG PET Imaging Biomarkers in Patients with Esophageal Cancer. Radiology 287:983–992. https://doi.org/https://doi.org/10.1148/radiol.2018172229
Elliott J A, O’Farrell N J, King S, Halpenny D, Malik V, et al. (2014) Value of CT–PET after neoadjuvant chemoradiation in the prediction of histological tumour regression, nodal status and survival in oesophageal adenocarcinoma. Br J Surg 101:1702–1711. https://doi.org/https://doi.org/10.1002/bjs.9670
Acknowledgements
None
Funding
This study was supported by a grant from the Japanese Ministry of Education, Culture, Sports, Science, and Technology (Grant-in-Aid for Young Scientists KAKEN; no. 18K15573) and Canon Medical Systems.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by SO, SS, and HK. The first draft of the manuscript was written by SO and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
Tatsushi Kobayashi has funding from Canon Medical Systems. The other authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Ethical approval
This retrospective study was approved by the institutional review board in our center, and the requirement for written informed consent was waived.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oda, S., Kuno, H., Hiyama, T. et al. Computed tomography-based radiomic analysis for predicting pathological response and prognosis after neoadjuvant chemotherapy in patients with locally advanced esophageal cancer. Abdom Radiol 48, 2503–2513 (2023). https://doi.org/10.1007/s00261-023-03938-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-023-03938-6