Abstract
Purpose
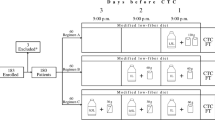
To determine whether the quality of a low-volume reduced bowel preparation (LV-RBP) for CT Colonography (CTC) is noninferior to full-volume reduced bowel preparation (FV-RBP) regimen.
Methods
In this randomized controlled trial, consecutive participants referred for CTC were randomly assigned to receive LV-RBP (52.5 g of PMF104 in 500 mL of water) or FV-RBP (105 g of PMF104 in 1000 mL of water). Images were independently reviewed by five blinded readers who rated the quality of bowel preparation from 0 (best score) to 3 (worst score). The primary outcome was the noninferiority of LV-RBP to FV-RBP in the proportion of colonic segments scored 0 for cleansing quality, with noninferiority margin of 10%. Volume of residual fluids, colonic distension, lesions and polyps detection rates and patient tolerability were secondary outcomes.
Results
From March 2019 to January 2020, 110 participants (mean age 65 years ± 14 [standard deviation]; 74 women) were allocated to LV-RBP (n = 55) or FV-RBP (n = 55) arms. There were 92% segment scored 0 in colon cleansing quality in LV-RBP and 94% in FV-RBP for prone scans, and 94% vs 92% for supine scans. Risk difference was – 2.1 (95% CI −5.9 to 1.7) and 1.5 (95% CI −2.4 to 5.4) for prone and supine positions, respectively. Residual fluids and colonic distension were also noninferior in LV-RBP. LV-RBP was associated with a lower number of evacuations during preparation (7 ± 5 vs 10 ± 6, p = 0.002).
Conclusion
The LV-RBP for CTC demonstrated noninferior quality of colon cleansing with improved gastrointestinal tolerability compared to FV-RBP regimen.
Graphical abstract






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
1. Spada C, Hassan C, Bellini D, et al (2020) Imaging alternatives to colonoscopy: CT colonography and colon capsule. European Society of Gastrointestinal Endoscopy (ESGE) and European Society of Gastrointestinal and Abdominal Radiology (ESGAR) Guideline – Update 2020. Eur Radiol. https://doi.org/https://doi.org/10.1007/s00330-020-07413-4
2. Laghi A, Rengo M, Graser A, Iafrate F (2013) Current status on performance of CT colonography and clinical indications. Eur J Radiol 82:1192–1200. https://doi.org/https://doi.org/10.1016/j.ejrad.2012.05.026
3. Laghi A, Bellini D, Petrozza V, et al (2015) Imaging of colorectal polyps and early rectal cancer. Color Dis 17 Suppl 1:36–43. https://doi.org/https://doi.org/10.1111/codi.12820
4. Utano K, Nagata K, Honda T, et al (2017) Diagnostic performance and patient acceptance of reducedlaxative CT colonography for the detection of polypoid and non-polypoid neoplasms: A multicenter prospective trial. Radiology 282:399–407. https://doi.org/https://doi.org/10.1148/radiol.2016160320
5. Liedenbaum MH, De Vries AH, Gouw CIBF, et al (2010) CT colonography with minimal bowel preparation: Evaluation of tagging quality, patient acceptance and diagnostic accuracy in two iodine-based preparation schemes. Eur Radiol 20:367–376. https://doi.org/https://doi.org/10.1007/s00330-009-1570-8
6. Senore C, Correale L, Regge D, et al (2018) Flexible sigmoidoscopy and CT colonography screening: Patients’ experience with and factors for undergoing screening-insight from the proteus colon trial. Radiology 286:873–883. https://doi.org/https://doi.org/10.1148/radiol.2017170228
The American College of Radiology (2019) ACR–SAR–SCBT-MR practice parameter for the performance of computed tomography (CT) colonography in adults. The American College of Radiology, Virginia, United States. Available via https://www.acr.org/-/media/ACR/Files/Practice-Parameters/ct-colonog.pdf. Accessed 15 Jan 2021.
8. Neri E, Halligan S, Hellström M, et al (2013) The second ESGAR consensus statement on CT colonography. Eur Radiol 23:720–729. https://doi.org/https://doi.org/10.1007/s00330-012-2632-x
9. Scalise P, Mantarro A, Pancrazi F, Neri E (2016) Computed tomography colonography for the practicing radiologist: A review of current recommendations on methodology and clinical indications. World J Radiol 8:472. https://doi.org/https://doi.org/10.4329/wjr.v8.i5.472
10. Neri E, Lefere P, Gryspeerdt S, et al (2013) Bowel preparation for CT colonography. Eur J Radiol 82:1137–1143. https://doi.org/https://doi.org/10.1016/j.ejrad.2012.11.006
11. Spada C, Cesaro P, Bazzoli F, et al (2017) Evaluation of Clensia®, a new low-volume PEG bowel preparation in colonoscopy: Multicentre randomized controlled trial versus 4L PEG. Dig Liver Dis 49:651–656. https://doi.org/https://doi.org/10.1016/j.dld.2017.01.167
12. Lefere PA, Gryspeerdt SS, Dewyspelaere J, et al (2002) Dietary fecal tagging as a cleansing method before CT colonography: Initial results - Polyp detection and patient acceptance. Radiology 224:393–403. https://doi.org/https://doi.org/10.1148/radiol.2241011222
13. Iannaccone R, Laghi A, Catalano C, et al (2004) Computed tomographic colonography without cathartic preparation for the detection of colorectal polyps. Gastroenterology 127:1300–1311. https://doi.org/https://doi.org/10.1053/j.gastro.2004.08.025
14. Utano K, Takayanagi D, Nagata K, et al (2019) A novel volume-reduced CT colonography regimen using hypertonic laxative (polyethylene glycol with ascorbic acid): randomized controlled trial. Eur Radiol 29:5236–5246. https://doi.org/https://doi.org/10.1007/s00330-019-06127-6
15. Moher D, Hopewell S, Schulz KF, et al (2010) CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. BMJ 340:. https://doi.org/https://doi.org/10.1136/bmj.c869
16. Piaggio G, Elbourne DR, Pocock SJ, et al (2012) Reporting of noninferiority and equivalence randomized trials: Extension of the CONSORT 2010 statement. JAMA - J. Am. Med. Assoc. 308:2594–2604
17. Bellini D, De Santis D, Caruso D, et al (2018) Bowel preparation in CT colonography: Is diet restriction necessary? A randomised trial (DIETSAN). Eur Radiol 28:382–389. https://doi.org/https://doi.org/10.1007/s00330-017-4997-3
18. Pollentine A, Mortimer A, Mccoubrie P, Archer L (2012) Evaluation of two minimal-preparation regimes for CT colonography: Optimising image quality and patient acceptability. Br J Radiol 85:1085–1092. https://doi.org/https://doi.org/10.1259/bjr/22421731
19. Ahn S, Park SH, Lee KH (2013) How to demonstrate similarity by using noninferiority and equivalence statistical testing in radiology research. Radiology 267:328–338
20. Hallgren KA (2012) Computing Inter-Rater Reliability for Observational Data: An Overview and Tutorial. Tutor Quant Methods Psychol 8:23–34. https://doi.org/https://doi.org/10.20982/tqmp.08.1.p023
21. Landis JR, Koch GG (1977) A One-Way Components of Variance Model for Categorical Data. Biometrics 33:671. https://doi.org/https://doi.org/10.2307/2529465
Sali L, Regge D (2016) CT colonography for population screening of colorectal cancer: Hints from European trials. Br. J. Radiol. 89
23. Johnson CD, Chen M-H, Toledano AY, et al (2008) Accuracy of CT Colonography for Detection of Large Adenomas and Cancers. N Engl J Med 359:1207–1217. https://doi.org/https://doi.org/10.1056/nejmoa0800996
24. Bibbins-Domingo K, Grossman DC, Curry SJ, et al (2016) Screening for colorectal cancer: US preventive services task force recommendation statement. JAMA - J Am Med Assoc 315:2564–2575. https://doi.org/https://doi.org/10.1001/jama.2016.5989
25. Laghi A, Iafrate F, Rengo M, Hassan C (2010) Colorectal cancer screening: the role of CT colonography. World J Gastroenterol 16:3987–3994
26. Hassan C, Laghi A, Pickhardt PJ, et al (2008) Projected impact of colorectal cancer screening with computerized tomographic colonography on current radiological capacity in Europe. Aliment Pharmacol Ther 27:366–374. https://doi.org/https://doi.org/10.1111/j.1365-2036.2007.03575.x
27. Iafrate F, Hassan C, Ciolina. M, et al (2011) High positive predictive value of CT colonography in a referral centre. Eur J Radiol 80:. https://doi.org/https://doi.org/10.1016/j.ejrad.2010.12.080
28. Sali L, Mascalchi M, Falchini M, et al (2016) Reduced and Full-Preparation CT Colonography, Fecal Immunochemical Test, and Colonoscopy for Population Screening of Colorectal Cancer: A Randomized Trial. J Natl Cancer Inst 108:. https://doi.org/https://doi.org/10.1093/jnci/djv319
Zhu H, Li F, Tao K, et al (2020) Comparison of the participation rate between CT colonography and colonoscopy in screening population: A systematic review and meta-analysis of randomized controlled trials. Br. J. Radiol. 93
30. Patrick JL, Bakke JR, Bannas P, et al (2015) Objective volumetric comparison of room air versus carbon dioxide for colonic distention at screening CT colonography. Abdom Imaging 40:. https://doi.org/https://doi.org/10.1007/s00261-014-0206-x
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Conceptualization: DB, NP, AL; Methodology: DB, NP; Formal analysis and investigation: DB, NP, SV, MR, PL; Writing—original draft preparation: NP, SV; Writing—review and editing: DB, NP, MR, DC, IC, AL; Supervision: DB, IC, AL.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical approval
Approval was obtained from the ethics committee of Sapienza University of Rome. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bellini, D., Panvini, N., Vicini, S. et al. Low-volume reduced bowel preparation regimen for CT colonography: a randomized noninferiority trial. Abdom Radiol 46, 4556–4566 (2021). https://doi.org/10.1007/s00261-021-03176-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03176-8