Abstract
Fibroblast activation protein (FAP) has emerged as a highly promising target for cancer diagnostic imaging and targeted radionuclide therapy. To exploit the therapeutic potential of suitably radiolabeled FAP inhibitors (FAPIs), this study presents the design and synthesis of a series of FAPI dimers to increase tumor uptake and retention. Preclinical evaluation and a pilot clinical PET imaging study were conducted to screen the lead compound with the potential for radionuclide therapy.
Methods
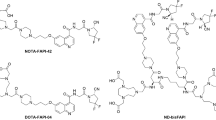
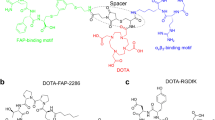
Three new FAPI dimers were synthesized by linking two quinoline-based FAPIs with different spacers. The in vitro binding affinity and preclinical small animal PET imaging of the compounds were compared with their monomeric counterparts, FAPI-04 and FAPI-46. The lead compound, [68Ga]Ga -LNC1013, was then evaluated in a pilot clinical PET imaging study involving seven patients with gastrointestinal cancer.
Results
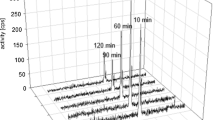
The three newly synthesized FAPI homodimers had high binding affinity and specificity in vitro and in vivo. Small animal PET imaging and biodistribution studies showed that [68Ga]Ga-LNC1013 had persistent tumor retention for at least 4 h, also higher uptake than the other two dimers and the monomer counterparts, making it the lead compound to enter clinical investigation. In the pilot clinical PET imaging study, seven patients were enrolled. The effective dose of [68Ga]Ga-LNC1013 was 8.24E-03 mSv/MBq. The human biodistribution of [68Ga]Ga-LNC1013 demonstrated prominent tumor uptake and good tumor-to-background contrast. [68Ga]Ga-LNC1013 PET imaging showed potential in capturing primary and metastatic lesions and outperforming 18F-FDG PET in detecting pancreatic and esophageal cancers. The SUVmax for lesions with [68Ga]Ga-FAPI-46 decreased over time, whereas [68Ga]Ga-LNC1013 exhibited persistently high tumor uptake from 1 to 4 h post-injection.
Conclusion
Dimerization is an effective strategy to produce FAPI derivatives with favorable tumor uptake, long tumor retention, and imaging contrast over its monomeric counterpart. We demonstrated that [68Ga]Ga-LNC1013, the lead compound without any piperazine moiety, had superior diagnostic potential over [68Ga]Ga-FAPI-46 and 18F-FDG, suggesting the future potential of LNC1013 for radioligand therapy of FAP-positive cancers.







Similar content being viewed by others
Data availability
Not applicable.
References
Hamson EJ, Keane FM, Tholen S, Schilling O, Gorrell MD. Understanding fibroblast activation protein (FAP): substrates, activities, expression and targeting for cancer therapy. Proteomics Clin Appl. 2014;8:454–63. https://doi.org/10.1002/prca.201300095.
Altmann A, Haberkorn U, Siveke J. The latest developments in imaging of fibroblast activation protein. J Nucl Med. 2021;62:160–7. https://doi.org/10.2967/jnumed.120.244806.
Moon ES, Elvas F, Vliegen G, De Lombaerde S, Vangestel C, De Bruycker S, et al. Targeting fibroblast activation protein (FAP): next generation PET radiotracers using squaramide coupled bifunctional DOTA and DATA(5m) chelators. EJNMMI Radiopharm Chem. 2020;5:19. https://doi.org/10.1186/s41181-020-00102-z.
Watabe T, Liu Y, Kaneda-Nakashima K, Shirakami Y, Lindner T, Ooe K, et al. Theranostics targeting fibroblast activation protein in the tumor stroma: (64)Cu- and (225)Ac-labeled FAPI-04 in pancreatic cancer xenograft mouse models. J Nucl Med. 2020;61:563–9. https://doi.org/10.2967/jnumed.119.233122.
Kosmala A, Serfling SE, Dreher N, Lindner T, Schirbel A, Lapa C, et al. Associations between normal organs and tumor burden in patients imaged with fibroblast activation protein inhibitor-directed positron emission tomography. Cancers (Basel). 2022;14. https://doi.org/10.3390/cancers14112609.
Privé BM, Boussihmad MA, Timmermans B, van Gemert WA, Peters SMB, Derks YHW, et al. Fibroblast activation protein-targeted radionuclide therapy: background, opportunities, and challenges of first (pre)clinical studies. Eur J Nucl Med Mol Imaging. 2023;50:1906–18. https://doi.org/10.1007/s00259-023-06144-0.
Ferdinandus J, Costa PF, Kessler L, Weber M, Hirmas N, Kostbade K, et al. Initial clinical experience with (90)Y-FAPI-46 radioligand therapy for advanced-stage solid tumors: a case series of 9 patients. J Nucl Med. 2022;63:727–34. https://doi.org/10.2967/jnumed.121.262468.
Moon ES, Ballal S, Yadav MP, Bal C, Van Rymenant Y, Stephan S, et al. Fibroblast Activation Protein (FAP) targeting homodimeric FAP inhibitor radiotheranostics: a step to improve tumor uptake and retention time. Am J Nucl Med Mol Imaging. 2021;11:476–91.
Millul J, Koepke L, Haridas GR, Sparrer KMJ, Mansi R, Fani M. Head-to-head comparison of different classes of FAP radioligands designed to increase tumor residence time: monomer, dimer, albumin binders, and small molecules vs peptides. Eur J Nucl Med Mol Imaging. 2023. https://doi.org/10.1007/s00259-023-06272-7.
Yadav MP, Ballal S, Martin M, Roesch F, Satapathy S, Moon ES, et al. Therapeutic potential of [177Lu]Lu-DOTAGA-FAPi dimers in metastatic breast cancer patients with limited treatment options: efficacy and safety assessment. Eur J Nucl Med Mol Imaging. 2023. https://doi.org/10.1007/s00259-023-06482-z.
Galbiati A, Zana A, Bocci M, Millul J, Elsayed A, Mock J, et al. A dimeric FAP-targeting small-molecule radioconjugate with high and prolonged tumor uptake. J Nucl Med. 2022;63:1852–8. https://doi.org/10.2967/jnumed.122.264036.
Zhao L, Niu B, Fang J, Pang Y, Li S, Xie C, et al. Synthesis, preclinical evaluation, and a pilot clinical PET imaging study of (68)Ga-labeled FAPI dimer. J Nucl Med. 2022;63:862–8. https://doi.org/10.2967/jnumed.121.263016.
Zhao L, Chen J, Pang Y, Fang J, Fu K, Meng L, et al. Development of fibroblast activation protein inhibitor-based dimeric radiotracers with improved tumor retention and antitumor efficacy. Mol Pharm. 2022;19:3640–51. https://doi.org/10.1021/acs.molpharmaceut.2c00424.
Loktev A, Lindner T, Burger EM, Altmann A, Giesel F, Kratochwil C, et al. Development of fibroblast activation protein-targeted radiotracers with improved tumor retention. J Nucl Med. 2019;60:1421–9. https://doi.org/10.2967/jnumed.118.224469.
Zhong X, Guo J, Han X, Wu W, Yang R, Zhang J, et al. Synthesis and preclinical evaluation of a novel FAPI-04 dimer for cancer theranostics. Mol Pharm. 2023;20:2402–14. https://doi.org/10.1021/acs.molpharmaceut.2c00965.
Li H, Ye S, Li L, Zhong J, Yan Q, Zhong Y, et al. (18)F- or (177)Lu-labeled bivalent ligand of fibroblast activation protein with high tumor uptake and retention. Eur J Nucl Med Mol Imaging. 2022;49:2705–15. https://doi.org/10.1007/s00259-022-05757-1.
Zhao L, Chen J, Pang Y, Fu K, Shang Q, Wu H, et al. Fibroblast activation protein-based theranostics in cancer research: a state-of-the-art review. Theranostics. 2022;12:1557–69. https://doi.org/10.7150/thno.69475.
Giesel FL, Kratochwil C, Lindner T, Marschalek MM, Loktev A, Lehnert W, et al. (68)Ga-FAPI PET/CT: biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J Nucl Med. 2019;60:386–92. https://doi.org/10.2967/jnumed.118.215913.
Ballal S, Yadav MP, Moon ES, Kramer VS, Roesch F, Kumari S, et al. Biodistribution, pharmacokinetics, dosimetry of [(68)Ga]Ga-DOTA.SA.FAPi, and the head-to-head comparison with [(18)F]F-FDG PET/CT in patients with various cancers. Eur J Nucl Med Mol Imaging. 2021;48:1915–31. https://doi.org/10.1007/s00259-020-05132-y.
Loktev A, Lindner T, Mier W, Debus J, Altmann A, Jager D, et al. A tumor-imaging method targeting cancer-associated fibroblasts. J Nucl Med. 2018;59:1423–9. https://doi.org/10.2967/jnumed.118.210435.
Assadi M, Rekabpour SJ, Jafari E, Divband G, Nikkholgh B, Amini H, et al. Feasibility and therapeutic potential of 177Lu-fibroblast activation protein inhibitor-46 for patients with relapsed or refractory cancers: a preliminary study. Clin Nucl Med. 2021;46:e523–30. https://doi.org/10.1097/RLU.0000000000003810.
Li ZB, Chen K, Chen X. (68)Ga-labeled multimeric RGD peptides for microPET imaging of integrin alpha(v)beta (3) expression. Eur J Nucl Med Mol Imaging. 2008;35:1100–8. https://doi.org/10.1007/s00259-007-0692-y.
Zang J, Wen X, Lin R, Zeng X, Wang C, Shi M, et al. Synthesis, preclinical evaluation and radiation dosimetry of a dual targeting PET tracer [(68)Ga]Ga-FAPI-RGD. Theranostics. 2022;12:7180–90. https://doi.org/10.7150/thno.79144.
Pang Y, Zhao L, Fang J, Chen J, Meng L, Sun L, et al. Development of FAPI tetramers to improve tumor uptake and efficacy of FAPI radioligand therapy. J Nucl Med. 2023:jnumed.123.265599. https://doi.org/10.2967/jnumed.123.265599.
Lang L, Li W, Guo N, Ma Y, Zhu L, Kiesewetter DO, et al. Comparison study of [18F]FAl-NOTA-PRGD2, [18F]FPPRGD2, and [68Ga]Ga-NOTA-PRGD2 for PET imaging of U87MG tumors in mice. Bioconjug Chem. 2011;22:2415–22. https://doi.org/10.1021/bc200197h.
Zhang RH, Guo HY, Deng H, Li J, Quan ZS. Piperazine skeleton in the structural modification of natural products: a review. J Enzyme Inhib Med Chem. 2021;36:1165–97. https://doi.org/10.1080/14756366.2021.1931861.
Meyer C, Dahlbom M, Lindner T, Vauclin S, Mona C, Slavik R, et al. Radiation dosimetry and biodistribution of (68)Ga-FAPI-46 PET imaging in cancer patients. J Nucl Med. 2020;61:1171–7. https://doi.org/10.2967/jnumed.119.236786.
Acknowledgements
The authors acknowledge the staff of the Department of Nuclear Medicine, Xiangya Hospital, for their help in the first-in-human study. The authors thank Professor Haojun Chen (Xiamen University) for providing HT-1080-FAP and HT-1080 cell lines.
Funding
This study was supported by the Ministry of Education, Singapore, under the Academic Research Fund Tier 1 FY2022 (NUHSRO/2022/093/T1/Seed-Sep/06), the National University of Singapore Start-up Grant (NUHSRO/2021/097/Startup/13; NUHSRO/2020/133/Startup/08, NUHSRO/2023/008/NUSMed/TCE/LOA, NUHSRO/2021/034/TRP/09/Nanomedicine), Shandong Provincial Natural Science Foundation (ZR2022MH099), National Medical Research Council (MOH-001334-00, MOH-001254-00, MOH-001388–00, MOH-001041, CG21APR1005), Singapore Ministry of Education (MOE-000387–00), and National Research Foundation (NRF-000352–00).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
Ethical approval was obtained from the Medical Ethics Committee of Xiangya Hospital, Central South University (Ethics Approval No. 202106053), and this study was conducted in accordance with the principles of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all participants included in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tan, Y., Li, J., Zhao, T. et al. Clinical translation of a novel FAPI dimer [68Ga]Ga-LNC1013. Eur J Nucl Med Mol Imaging (2024). https://doi.org/10.1007/s00259-024-06703-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00259-024-06703-z