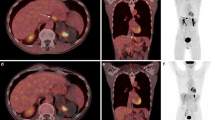
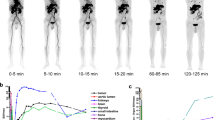
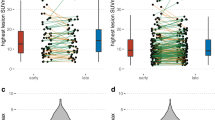
Abstract
Purpose
Positron emission tomography/computed tomography (PET/CT) based on fibroblast activation protein inhibitors (FAPI) has shown complementary values to 2-[18F]-fluoro-2-deoxy-D-glucose ([18F]FDG) in cancer imaging. This study aimed to investigate the feasibility of a one-stop FDG-FAPI dual-tracer imaging protocol with dual-low activity for oncological imaging.
Methods
Nineteen patients with malignancies underwent one-stop [18F]FDG (0.37 MBq/kg) PET (PETFDG) and dual-tracer PET 30–40 and 50–60 min (hereafter, PETD30–40 and PETD50–60, respectively) after additional injection of [68Ga]Ga-DOTA-FAPI-04 (0.925 MBq/kg), with a single diagnostic CT to generate the PET/CT. The lesion detection rate and tumor-to-normal ratios (TNRs) of tracer uptake were compared between PETFDG/CT and PETD50–60/CT and between PETD50–60/CT and PETD30–40/CT. In addition, a visual scoring system was established to compare the lesion detectability.
Results
The dual-tracer PETD50–60 and PETD30–40/CT showed similar performance in detecting primary tumors but presented significantly higher lesion TNRs than PETFDG. Significantly, more metastases with higher TNRs were identified on PETD50–60 than PETFDG (491 vs. 261, P < 0.001). The dual-tracer PETD50–60 received significantly higher visual scores than single PETFDG (111 vs. 10) in demonstrating both primary tumors (12 vs. 2) and metastases (99 vs. 8). However, these differences were not significant between PETD50–60 and PETD30–40. These resulted in tumor upstaging in 44.4% patients taking PET/CT for initial assessment, and more recurrences (68 vs. 7) were identified in patients taking PET/CT for restaging, both on PETD50–60 and PETD30–40, compared to PETFDG. The reduced effective dosimetry per patient (26.2 ± 2.57 mSv) was equal to that of a single standard whole-body PET/CT.
Conclusion
The one-stop dual-tracer dual-low-activity PET imaging protocol combines the strengths of [18F]FDG and [68Ga]Ga-DOTA-FAPI-04 with shorter duration and lesser radiation and is thus clinically applicable.






Similar content being viewed by others
Data availability
The dataset used and/or analyzed in the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Change history
01 July 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00259-023-06303-3
References
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16(9):582–98.
Huang R, Pu Y, Huang S, Yang C, Yang F, Pu Y, et al. FAPI-PET/CT in cancer imaging: a potential novel molecule of the century. Front Oncol. 2022;12:854658.
Chen H, Pang Y, Wu J, Zhao L, Hao B, Wu J, et al. Comparison of [68Ga]Ga-DOTA-FAPI-04 and [18F]FDG PET/CT for the diagnosis of primary and metastatic lesions in patients with various types of cancer. Eur J Nucl Med Mol Imaging. 2020;47(8):1820–32.
Lan L, Liu H, Wang Y, Deng J, Peng D, Feng Y, et al. The potential utility of [68Ga]Ga-DOTA-FAPI-04 as a novel broad-spectrum oncological and non-oncological imaging agent—comparison with [18F]FDG. Eur J Nucl Med Mol Imaging. 2022;49(3):963–79.
Çermik TF, Ergül N, Yılmaz B, Mercanoğlu G. Tumor imaging with 68Ga-DOTA-FAPI-04 PET/CT: comparison with 18F-FDG PET/CT in 22 different cancer types. Clin Nucl Med. 2022;47(4):e333–9.
Wu J, Wang Y, Liao T, Rao Z, Gong W, Ou L, et al. Comparison of the relative diagnostic performance of [68Ga]Ga-DOTA-FAPI-04 and [18F]FDG PET/CT for the detection of bone metastasis in patients with different cancers. Front Oncol. 2021;11:737827.
Peng D, He J, Liu H, Cao J, Wang Y, Chen Y. FAPI PET/CT research progress in digestive system tumours. Digest Liver Dis. 2022;54(2):164–9.
Qin C, Liu F, Huang J, Ruan W, Liu Q, Gai Y, et al. A head-to-head comparison of 68Ga-DOTA-FAPI-04 and 18F-FDG PET/MR in patients with nasopharyngeal carcinoma: a prospective study. Eur J Nucl Med Mol Imaging. 2021;48(10):3228–37.
Roth KS, Voltin CA, van Heek L, Wegen S, Schomacker K, Fischer T, et al. Dual-tracer PET/CT protocol with [18F]-FDG and [68Ga]Ga-FAPI-46 for cancer imaging: a proof of concept. J Nucl Med. 2022;63(11):1683–6.
Badawi RD, Shi H, Hu P, Chen S, Xu T, Price PM, et al. First human imaging studies with the EXPLORER total-body PET scanner. J Nucl Med. 2019;60(3):299–303.
Alberts I, Hunermund JN, Prenosil G, Mingels C, Bohn KP, Viscione M, et al. Clinical performance of long axial field of view PET/CT: a head-to-head intra-individual comparison of the Biograph Vision Quadra with the Biograph Vision PET/CT. Eur J Nucl Med Mol Imaging. 2021;48(8):2395–404.
Liu G, Hu P, Yu H, Tan H, Zhang Y, Yin H, et al. Ultra-low-activity total-body dynamic PET imaging allows equal performance to full-activity PET imaging for investigating kinetic metrics of 18F-FDG in healthy volunteers. Eur J Nucl Med Mol Imaging. 2021;48(8):2373–83.
Sui X, Liu G, Hu P, Chen S, Yu H, Wang Y, et al. Total-body PET/computed tomography highlights in clinical practice: experiences from Zhongshan Hospital. Fudan University PET Clin. 2021;16(1):9–14.
Yu H, Gu Y, Fan W, Gao Y, Wang M, Zhu X, et al. Expert consensus on oncological [18F]FDG total-body PET/CT imaging (version 1). Eur Radiol. 2023;33(1):615–26.
Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res. 1996;82:359–74.
Qin C, Shao F, Gai Y, Liu Q, Ruan W, Liu F, et al. 68Ga-DOTA-FAPI-04 PET/MR in the evaluation of gastric carcinomas: comparison with 18F-FDG PET/CT. J Nucl Med. 2022;63(1):81–8.
Amin MB, Edge SB, Greene FL, Schilsky RL, Gaspar LE, Washington MK, et al. AJCC cancer staging manual (8th edition). New York: Springer; 2017.
ICRP. Radiation dose to patients from radiopharmaceuticals. Addendum 3 to ICRP Publication 53. ICRP Publication 106. Approved by the Commission in October 2007. Ann ICRP. 2008;38(1–2):1–197.
Giesel FL, Kratochwil C, Lindner T, Marschalek MM, Loktev A, Lehnert W, et al. 68Ga-FAPI PET/CT: biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J Nucl Med. 2019;60(3):386–92.
Christner JA, Kofler JM, McCollough CH. Estimating effective dose for CT using dose-length product compared with using organ doses: consequences of adopting International Commission on Radiological Protection publication 103 or dual-energy scanning. Am J Roentgenol. 2010;194(4):881–9.
Brix G, Lechel U, Glatting G, Ziegler SI, Munzing W, Muller SP, et al. Radiation exposure of patients undergoing whole-body dual-modality 18F-FDG PET/CT examinations. J Nucl Med. 2005;46(4):608–13.
Li Y, Jiang L, Wang H, Cai H, Xiang Y, Li L. Effective radiation dose of 18F-FDG PET/CT: how much does diagnostic CT contribute? Radiat Prot Dosim. 2019;187(2):183–90.
Huang B, Law MW, Khong PL. Whole-body PET/CT scanning: estimation of radiation dose and cancer risk. Radiology. 2009;251(1):166–74.
Hu Y, Liu G, Yu H, Wang Y, Li C, Tan H, et al. Feasibility of acquisitions using total-body PET/CT with an ultra-low 18F-FDG activity. J Nucl Med. 2022;63(6):959–65.
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42(2):328–54.
Giesel FL, Kratochwil C, Schlittenhardt J, Dendl K, Eiber M, Staudinger F, et al. Head-to-head intra-individual comparison of biodistribution and tumor uptake of 68Ga-FAPI and 18F-FDG PET/CT in cancer patients. Eur J Nucl Med Mol Imaging. 2021;48(13):4377–85.
Koerber SA, Staudinger F, Kratochwil C, Adeberg S, Haefner MF, Ungerechts G, et al. The role of 68Ga-FAPI PET/CT for patients with malignancies of the lower gastrointestinal tract: first clinical experience. J Nucl Med. 2020;61(9):1331–6.
Kratochwil C, Flechsig P, Lindner T, Abderrahim L, Altmann A, Mier W, et al. 68Ga-FAPI PET/CT: tracer uptake in 28 different kinds of cancer. J Nucl Med. 2019;60(6):801–5.
Calais J, Mona CE. Will FAPI PET/CT replace FDG PET/CT in the next decade?—point: an important diagnostic, phenotypic, and biomarker role. Am J Roentgenol. 2021;216(2):305–6.
Li L, Hu X, Ma J, Yang S, Gong W, Zhang C. A systematic review of [68Ga]Ga-DOTA-FAPI-04 and [18F]FDG PET/CT in the diagnostic value of malignant tumor bone metastasis. Front Oncol. 2022;12:978506.
Funding
This study was funded by the Shanghai Municipal Key Clinical Specialty Project (grant number: SHSLCZDZK03401 to H.S.), the Major Science and Technology Projects for Major New Drug Creation (grant number: 2019ZX09302001 to H.S.), the Shanghai Science and Technology Committee Program (grant number: 20DZ2201800 to H.S.), the Three-year Action Plan of Clinical Skills and Innovation of Shanghai Hospital Development Center (grant number: SHDC2020CR3079B to H.S.), and the Next Generation Information Infrastructure Construction Project founded by Shanghai Municipal Commission of Economy and Informatization (grant number: 201901014 to H.S.).
Author information
Authors and Affiliations
Contributions
G. Liu and W. Mao had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. G. Liu, W. Mao, J. Gu, and H. Shi were responsible for the concept and design of the study. G. Liu, W. Mao, and H. Yu were involved in data acquisition. G. Liu and Y. Hu were involved in image review, data analysis, and interpretation. G. Liu and W. Mao drafted the manuscript, and all authors revised it critically. G. Liu and Y. Hu did the statistical analysis. J. Gu and H. Shi supervised the study. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted. J. Gu and H. Shi are the guarantors.
Corresponding authors
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of Zhongshan Hospital of Fudan University (approval number: B2022-098R2).
Consent to participate
Written informed consents were obtained from included subjects for participation of this study.
Consent for publication
The authors affirm that human research participants provided informed consent for publication of the studied data and the images in Figs. 3, 4, and 5.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Miscellanea
The original online version of this article was revised: The authors regret to inform that the following statement was not included in the legend of Figure 1: “This figure was adapted with permission from a JNM article—Roth KS, Voltin CA, van Heek L, etal. J Nucl Med 2022; 63(11):1683-1686. © SNMMI (ref. 9).” The original article has been corrected.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, G., Mao, W., Yu, H. et al. One-stop [18F]FDG and [68Ga]Ga-DOTA-FAPI-04 total-body PET/CT examination with dual-low activity: a feasibility study. Eur J Nucl Med Mol Imaging 50, 2271–2281 (2023). https://doi.org/10.1007/s00259-023-06207-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-023-06207-2