Abstract
Purpose
The aims of this retrospective analysis were to compare 68Ga-PSMA PET findings and low-dose CT findings (120 kV, 30 mA), and to obtain semiquantitative and quantitative 68Ga-PSMA PET data in patients with prostate cancer (PC) bone metastases.
Methods
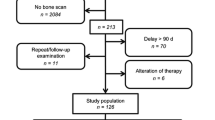
In total, 152 PET/CT scans from 140 patients were evaluated. Of these patients, 30 had previously untreated primary PC, and 110 had biochemical relapse after treatment of primary PC. All patients underwent dynamic PET/CT scanning of the pelvis and lower abdomen as well as whole-body PET/CT with 68Ga-PSMA-11. The PET/CT scans were analysed qualitatively (visually), semiquantitatively (SUV), and quantitatively based on a two-tissue compartment model and a noncompartmental approach leading to the extraction of the fractal dimension. Differences were considered significant for p values <0.05.
Results
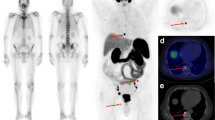
In total, 168 68Ga-PSMA-positive and 113 CT-positive skeletal lesions were detected in 37 patients (8 with primary PC, 29 with biochemical recurrence). Of these 168 lesions, 103 were both 68Ga-PSMA PET-positive and CT-positive, 65 were only 68Ga-PSMA-positive, and 10 were only CT-positive. The Yang test showed that there were significantly more 68Ga-PSMA PET-positive lesions than CT-positive lesions. Association analysis showed that PSA plasma levels were significantly correlated with several 68Ga-PSMA-11-associated parameters in bone metastases, including the degree of tracer uptake (SUVaverage and SUVmax), its transport rate from plasma to the interstitial/intracellular compartment (K1), its rate of binding to the PSMA receptor and its internalization (k3), its influx rate (Ki), and its distribution heterogeneity.
Conclusion
68Ga-PSMA PET/CT is a useful diagnostic tool in the detection of bone metastases in PC. 68Ga-PSMA PET visualizes more bone metastases than low-dose CT. PSA plasma levels are significantly correlated with several 68Ga-PSMA PET parameters.





Similar content being viewed by others
References
Langsteger W, Rezaee A, Pirich C, Beheshti M. 18F-NaF-PET/CT and 99mTc-MDP bone scintigraphy in the detection of bone metastases in prostate cancer. Semin Nucl Med. 2016;46(6):491–501.
Mottet N, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71:618–29.
Mohler JL, Kantoff PW, Armstrong AJ, et al. Prostate cancer, version 2.2014. J Natl Compr Cancer Netw. 2014;12:686–718.
Afshar-Oromieh A, Zechmann CM, Malcher A, et al. Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2014;41:11–20.
Afshar-Oromieh A, Avtzi E, Giesel FL, et al. The diagnostic value of PET/CT imaging with the (68)Ga-labelled PSMA ligand HBED-CC in the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2015;42:197–209.
Eiber M, Maurer T, Souvatzoglou M, et al. Evaluation of hybrid 68Ga-PSMA-ligand PET/CT in 248 patients with biochemical recurrence after radical prostatectomy. J Nucl Med. 2015;56:668–74.
Sachpekidis C, Eder M, Kopka K, et al. 68Ga-PSMA-11 dynamic PET/CT imaging in biochemical relapse of prostate cancer. Eur J Nucl Med Mol Imaging. 2016;43:1288–99.
Maurer T, Gschwend JE, Rauscher J, et al. Diagnostic efficacy of (68)gallium-PSMA-PET compared to conventional imaging in lymph node staging of 130 consecutive patients with intermediate to high risk prostate cancer. J Urol. 2016;195(5):1436–43.
Sachpekidis C, Kopka K, Eder M, et al. 68Ga-PSMA-11 dynamic PET/CT imaging in primary prostate cancer. Clin Nucl Med. 2016;41(11):e473–9.
Uprimny C, Kroiss AS, Decristoforo C, et al. 68Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nucl Med Mol Imaging. 2017;44(6):941–9.
Afshar-Oromieh A, Holland-Letz T, Giesel FL, et al. Diagnostic performance of 68Ga-PSMA-11 (HBED-CC) PET/CT in patients with recurrent prostate cancer: evaluation in 1007 patients. Eur J Nucl Med Mol Imaging. 2017;44(8):1258–68.
Pyka T, Okamoto S, Dahlbender M, et al. Comparison of bone scintigraphy and 68Ga-PSMA PET for skeletal staging in prostate cancer. Eur J Nucl Med Mol Imaging. 2016;43(12):2114–21.
Thomas L, Balmus C, Ahmadzadehfar H, et al. Assessment of bone metastases in patients with prostate cancer – a comparison between 99mTc-bone-scintigraphy and [68Ga]Ga-PSMA PET/CT. Pharmaceuticals (Basel). 2017;10(3):68.
Eder M, Schäfer M, Bauder-Wüst U, et al. 68Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem. 2012;23:688–97.
Eder M, Neels O, Müller M, et al. Novel preclinical and radiopharmaceutical aspects of [68Ga]Ga-PSMA-HBED-CC: a new PET tracer for imaging of prostate cancer. Pharmaceuticals (Basel). 2014;7:779–96.
Schäfer M, Bauder-Wüst U, Leotta K, Zoller F, Mier W, et al. A dimerized urea-based inhibitor of the prostate-specific membrane antigen for 68Ga-PET imaging of prostate cancer. EJNMMI Res. 2012;2:23.
Strauss LG, Conti PS. The applications of PET in clinical oncology. J Nucl Med. 1991;32:623–48.
Burger C, Buck A. Requirements and implementations of a flexible kinetic modeling tool. J Nucl Med. 1997;38:1818–23.
Mikolajczyk K, Szabatin M, Rudnicki P, Grodzki M, Burger C. A JAVA environment for medical image data analysis: initial application for brain PET quantitation. Med Inf. 1998;23:207–14.
Sokoloff L, Smith CB. Basic principles underlying radioisotopic methods for assay of biochemical processes in vivo. In: Greitz T, Ingvar DH, Widén L, editors. The metabolism of the human brain studied with positron emission tomography. New York: Raven Press; 1983. p. 123–48.
Miyazawa H, Osmont A, Petit-Taboué MC, et al. Determination of 18F-fluoro-2-deoxy-D-glucose rate constants in the anesthetized baboon brain with dynamic positron tomography. J Neurosci Methods. 1993;50:263–72.
Ohtake T, Kosaka N, Watanabe T, et al. Noninvasive method to obtain input function for measuring tissue glucose utilization of thoracic and abdominal organs. J Nucl Med. 1991;32:1432–8.
Galli G, Indovina L, Calcagni ML, Mansi L, Giordano A. The quantification with FDG as seen by a physician. Nucl Med Biol. 2013;40:720–30.
Guo N, Lang L, Gao H, Niu G, Kiesewetter DO, et al. Quantitative analysis and parametric imaging of 18F-labeled monomeric and dimeric RGD peptides using compartment model. Mol Imaging Biol. 2012;14:743–52.
Dimitrakopoulou-Strauss A, Strauss LG, Burger C, et al. On the fractal nature of positron emission tomography (PET) studies. World J Nucl Med. 2003;4:306–13.
Krzywinski M, Altman N, Blainey P. Points of significance: nested designs. For studies with hierarchical noise sources, use a nested analysis of variance approach. Nat Methods. 2014;11(10):977–8.
Yang Z, Sun X, Hardin JWA. Note on the tests for clustered matched-pair binary data. Biom J. 2010;52:638–52.
Ibrahim T, Flamini E, Mercatali L, Sacanna E, Serra P, Amadori D. Pathogenesis of osteoblastic bone metastases from prostate cancer. Cancer. 2010;116(6):1406–18.
Cook GJ, Azad G, Padhani AR. Bone imaging in prostate cancer: the evolving roles of nuclear medicine and radiology. Clin Transl Imaging. 2016;4(6):439–47.
Fonager RF, Zacho HD, Langkilde NC, Petersen LJ. (18)F-fluoride positron emission tomography/computed tomography and bone scintigraphy for diagnosis of bone metastases in newly diagnosed, high-risk prostate cancer patients: study protocol for a multicentre, diagnostic test accuracy study. BMC Cancer. 2016;16:10.
Mannweiler S, Amersdorfer P, Trajanoski S, Terrett JA, King D, Mehes G. Heterogeneity of prostate-specific membrane antigen (PSMA) expression in prostate carcinoma with distant metastasis. Pathol Oncol Res. 2009;15:167–72.
Miyamoto DT, Sequist LV, Lee RJ. Circulating tumour cells – monitoring treatment response in prostate cancer. Nat Rev Clin Oncol. 2014;11:401–12.
Baum RP, Kulkarni HR, Schuchardt C, Singh A, Wirtz M, et al. 177Lu-Labeled prostate-specific membrane antigen radioligand therapy of metastatic castration-resistant prostate cancer: safety and efficacy. J Nucl Med. 2016;57:1006–13.
Rahbar K, Ahmadzadehfar H, Kratochwil C, Haberkorn U, Schäfers M, et al. German multicenter study investigating 177Lu-PSMA-617 radioligand therapy in advanced prostate cancer patients. J Nucl Med. 2017;58:85–90.
Kratochwil C, Bruchertseifer F, Giesel FL, Weis M, Verburg FA, et al. 225Ac-PSMA-617 for PSMA-targeted α-radiation therapy of metastatic castration-resistant prostate cancer. J Nucl Med. 2016;57:1941–4.
Kratochwil C, Bruchertseifer F, Rathke H, Bronzel M, Apostolidis C, et al. Targeted α-therapy of metastatic castration-resistant prostate cancer with 225Ac-PSMA-617: dosimetry estimate and empiric dose finding. J Nucl Med. 2017;58:1624–31.
Dey P. Basic principles and applications of fractal geometry in pathology: a review. Anal Quant Cytol Histol. 2005;27:284–90.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Sachpekidis, C., Bäumer, P., Kopka, K. et al. 68Ga-PSMA PET/CT in the evaluation of bone metastases in prostate cancer. Eur J Nucl Med Mol Imaging 45, 904–912 (2018). https://doi.org/10.1007/s00259-018-3936-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-018-3936-0