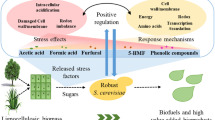
Abstract
The rising concern with the emission of greenhouse gases has boosted new incentives for biofuels production, which are less polluting than fossil fuels. Special attention has been given to the second-generation ethanol, as it is produced from abundant feedstocks which do not compete with food production, such as lignocellulosic biomass and whey. Kluyveromyces marxianus stands out in second-generation ethanol production due to its capacity of assimilating lactose, the sugar found in whey, and tolerating high temperatures used in simultaneous saccharification processes. Nonetheless, contrary to Saccharomyces cerevisiae, K. marxianus does not tolerate high ethanol concentrations. Ethanol causes a broad range of toxic effects on yeasts, acting on cell membrane and proteins, as well as inducing the generation of reactive oxygen species (ROS). The ethanol stress responses are not fully understood, mainly in non-conventional yeasts such as K. marxianus. Indeed, many molecular responses to ethanol stress are still inferred from S. cerevisiae. As such, a better understanding of the ethanol stress responses in K. marxianus may provide the basis for improving its use in the biofuel industry. Additionally, the selection of ethanol-tolerant strains by metabolic engineering is useful to provide strains with improved capacity to withstand stressful conditions, as well as to obtain new insights about the ethanol stress responses.
Key points
• It is still not totally clear why K. marxianus is less tolerant to ethanol than S. cerevisiae.
• Understanding the ethanol stress response in K. marxianus is pivotal for improving its application in the biofuel industry.
• The Metabolic engineering is expected to improve the ethanol tolerance in K. marxianus.


Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Adams CJ, Kopp MC, Larburu N, Nowak PR, Ali MMU (2019) Structure and molecular mechanism of ER stress signaling by the unfolded protein response signal activator IRE1. Front Mol Biosci 6:1–12. https://doi.org/10.3389/fmolb.2019.00011
Alvim MCT, Vital CE, Barros E, Vieira NM, da Silveira FA, Balbino TR, Diniz RHS, Brito AF, Bazzolli DMS, de Oliveira Ramos HJ, da Silveira WB (2019) Ethanol stress responses of Kluyveromyces marxianus CCT 7735 revealed by proteomic and metabolomic analyses. Anton Leeuw Int J Gen Mol Microbiol 112:827–845. https://doi.org/10.1007/s10482-018-01214-y
Bajpai P, Margaritis A (1982) Ethanol inhibition kinetics of Kluyveromyces marxianus grown on Jerusalem artichoke juice. Appl Environ Microbiol 44:1325–1329. https://doi.org/10.1128/aem.44.6.1325-1329.1982
Balat M, Balat H (2009) Recent trends in global production and utilization of bio-ethanol fuel. Appl Energy 86:2273–2282. https://doi.org/10.1016/j.apenergy.2009.03.015
Bandara A, Fraser S, Chambers PJ, Stanley GA (2009) Trehalose promotes the survival of Saccharomyces cerevisiae during lethal ethanol stress, but does not influence growth under sublethal ethanol stress. FEMS Yeast Res 9:1208–1216. https://doi.org/10.1111/j.1567-1364.2009.00569.x
Barrick JE, Yu DS, Yoon SH, Jeong H, Oh TK, Schneider D, Lenski RE, Kim JF (2009) Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature 461:1243–1247. https://doi.org/10.1038/nature08480
Buck M (1998) Trifluoroethanol and colleagues: cosolvents come of age. Recent studies with peptides and proteins. Q Rev Biophys 31:297–355. https://doi.org/10.1017/S003358359800345X
Buenrostro-Figueroa J, Tafolla-Arellano JC, Flores-Gallegos AC, Rodríguez-Herrera R, De la Garza-Toledo H, Aguilar CN (2018) Native yeasts for alternative utilization of overripe mango pulp for ethanol production. Rev Argent Microbiol 50:173–177. https://doi.org/10.1016/j.ram.2016.04.010
Campos BB, Diniz RHS, Da Silveira FA, Ribeiro Júnior JI, Fietto LG, MacHado JC, Da Silveira WB (2019) Elephant grass (Pennisetum purpureum Schumach) is a promising feedstock for ethanol production by the thermotolerant yeast Kluyveromyces marxianus CCT 7735. Braz J Chem Eng 36:43–49. https://doi.org/10.1590/0104-6632.20190361s20170263
Caspeta L, Chen Y, Ghiaci P, Feizi A, Baskov S, Hallström BM, Petranovic D, Nielsen J (2014) Altered sterol composition renders yeast thermotolerant. Science 80(346):75–78. https://doi.org/10.1126/SCIENCE.1258137
Celton M, Sanchez I, Goelzer A, Fromion V, Camarasa C, Dequin S (2012) A comparative transcriptomic, fluxomic and metabolomic analysis of the response of Saccharomyces cerevisiae to increases in NADPH oxidation. BMC Genomics 13https://doi.org/10.1186/1471-2164-13-317
Charoenbhakdi S, Dokpikul T, Burphan T, Techo T, Auesukaree C (2016) Vacuolar H+-ATPase protects Saccharomyces cerevisiae cells against ethanol induced oxidative and cell wall stresses. Appl Environ Microbiol 82:3121–3130. https://doi.org/10.1128/AEM.00376-16
Chen B, Retzlaff M, Roos T, Frydman J (2011) Cellular strategies of protein quality control. Cold Spring Harb Perspect Biol 3:a004374. https://doi.org/10.1101/cshperspect.a004374
Costa DA, de Souza CJA, Costa PA, Rodrigues MQRB, dos Santos AF, Lopes MR, Genier HLA, Silveira WB, Fietto LG (2014) Physiological characterization of thermotolerant yeast for cellulosic ethanol production. Appl Microbiol Biotechnol 98:3829–40. https://doi.org/10.1007/s00253-014-5580-3
Cray JA, Russell JT, Timson DJ, Singhal RS, Hallsworth JE (2013) A universal measure of chaotropicity and kosmotropicity. Environ Microbiol 15:287–296. https://doi.org/10.1111/1462-2920.12018
Curiel JA, Salvadó Z, Tronchoni J, Morales P, Rodrigues AJ, Quirós M, Gonzalez (2016) Identification of target genes to control acetate yield during aerobic fermentation with Saccharomyces cerevisiae. Microb Cell Fact 15:156. https://doi.org/10.1186/s12934-016-0555-y
Dasgupta D, Suman SK, Pandey D, Ghosh D, Khan R, Agrawal D, Jain RK, Vadde VT, Adhikari DK (2013) Design and optimization of ethanol production from bagasse pith hydrolysate by a thermotolerant yeast Kluyveromyces sp. IIPE453 using response surface methodology. Springer Plus 2:1–10. https://doi.org/10.1186/2193-1801-2-159
de Smidt O, du Preez JC, Albertyn J (2012) Molecular and physiological aspects of alcohol dehydrogenases in the ethanol metabolism of Saccharomyces cerevisiae. FEMS Yeast Res 12:33–47. https://doi.org/10.1111/j.1567-1364.2011.00760.x
de Souza CJA, Costa DA, Rodrigues MQRB, dos Santos AF, Lopes MR, Abrantes ABP, dos Santos Costa P, Silveira WB, Passos FML, Fietto LG (2012) The influence of presaccharification, fermentation temperature and yeast strain on ethanol production from sugarcane bagasse. Bioresour Technol 109:63–69. https://doi.org/10.1016/j.biortech.2012.01.024
Deparis Q, Claes A, Foulquié-Moreno MR, Thevelein JM (2017) Engineering tolerance to industrially relevant stress factors in yeast cell factories. FEMS Yeast Res 17:1–35. https://doi.org/10.1093/femsyr/fox036
Dequin S, Casaregola S (2011) The genomes of fermentative Saccharomyces. C R Biol 334:687–693. https://doi.org/10.1016/j.crvi.2011.05.019
Devanand T, Krishnaswamy S, Vemparala S (2019) Interdigitation of lipids induced by membrane–active proteins. J Membr Biol 252:331–342. https://doi.org/10.1007/s00232-019-00072-7
Ding J, Huang X, Zhang L, Zhao N, Yang D, Zhang K (2009) Tolerance and stress response to ethanol in the yeast Saccharomyces cerevisiae. Appl Microbiol Biotechnol 85:253–263. https://doi.org/10.1007/s00253-009-2223-1
Diniz RHS, Rodrigues MQRB, Fietto LG, Passos FML, Silveira WB (2013) Optimizing and validating the production of ethanol from cheese whey permeate by Kluyveromyces marxianus UFV-3. Biocatal Agric Biotechnol 3:111–117. https://doi.org/10.1016/j.bcab.2013.09.002
Diniz RHS, Villada JC, Alvim MCT, Vidigal PMP, Vieira NM, Lamas-Maceiras M, Cerdán ME, González-Siso MI, Lahtvee PJ, da Silveira WB (2017) Transcriptome analysis of the thermotolerant yeast Kluyveromyces marxianus CCT 7735 under ethanol stress. Appl Microbiol Biotechnol 101:6969–6980. https://doi.org/10.1007/s00253-017-8432-0
Dos Santos VC, Bragança CRS, Passos FJV, Passos FML (2013) Kinetics of growth and ethanol formation from a mix of glucose/xylose substrate by Kluyveromyces marxianus UFV-3. Anton Leeuwen Int J Gen Mol Microbiol 103:153–161. https://doi.org/10.1007/s10482-012-9794-z
Dragosits M, Mattanovich D (2013) Adaptive laboratory evolution – principles and applications for biotechnology. Microb Cell Fact 12 VN-r:64 https://doi.org/10.1186/1475-2859-12-64
Eardley J, Timson DJ (2020) Yeast cellular stress: impacts on bioethanol production. Fermentation 6:109. https://doi.org/10.3390/fermentation6040109
Ferreira PG, da Silveira FA, dos Santos RCV, Genier HLA, Diniz RHS, Ribeiro JI, Fietto LG, Passos FML, da Silveira WB (2015) Optimizing ethanol production by thermotolerant Kluyveromyces marxianus CCT 7735 in a mixture of sugarcane bagasse and ricotta whey. Food Sci Biotechnol 24:1421–1427. https://doi.org/10.1007/s10068-015-0182-0
Fong SS, Burgard AP, Herring CD, Knight EM, Blattner FR, Maranas CD, Palsson BO (2005) In silico design and adaptive evolution of Escherichia coli for production of lactic acid. Biotechnol Bioeng 91:643–648. https://doi.org/10.1002/bit.20542
Fu X, Li P, Zhang L, Li S (2019) Understanding the stress responses of Kluyveromyces marxianus after an arrest during high-temperature ethanol fermentation based on integration of RNA-Seq and metabolite data. Appl Microbiol Biotechnol 103:2715–2729. https://doi.org/10.1007/s00253-019-09637-x
Gao J, Yuan W, Li Y, Xiang R, Hou S, Zhong S, Bai F (2015) Transcriptional analysis of Kluyveromyces marxianus for ethanol production from inulin using consolidated bioprocessing technology. Biotechnol Biofuels 8:1–17. https://doi.org/10.1186/s13068-015-0295-y
Gomez M, Pérez-Gallardo R V., Sánchez LA, Díaz-Pérez AL, Cortés-Rojo C, Carmen VM, Saavedra-Molina A, Lara-Romero J, Jiménez-Sandoval S, Rodríguez F, Rodríguez-Zavala JS, Campos-García J (2014) Malfunctioning of the iron-sulfur cluster assembly machinery in saccharomyces cerevisiae produces oxidative stress via an iron-dependent mechanism, causing dysfunction in respiratory complexes. PLoS One 9https://doi.org/10.1371/journal.pone.0111585
Goshima T, Negi K, Tsuji M, Inoue H, Yano S, Hoshino T, Matsushika A (2013) Ethanol fermentation from xylose by metabolically engineered strains of Kluyveromyces marxianus. J Biosci Bioeng 116:551–554. https://doi.org/10.1016/j.jbiosc.2013.05.010
Güneşer O, Karagül-Yüceer Y, Wilkowska A, Kregiel D (2016) Volatile metabolites produced from agro-industrial wastes by Na-alginate entrapped Kluyveromyces marxianus. Braz J Microbiol 47:965–972. https://doi.org/10.1016/j.bjm.2016.07.018
Halbleib K, Pesek K, Covino R, Hofbauer HF, Wunnicke D, Hänelt I, Hummer G, Ernst R (2017) Activation of the unfolded protein response by lipid bilayer stress. Mol Cell 67:673-684.e8. https://doi.org/10.1016/j.molcel.2017.06.012
Hernández-Elvira M, Torres-Quiroz F, Escamilla-Ayala A, Domínguez-Martin E, Escalante R, Kawasaki L, Ongay-Larios L, Coria R (2018) The unfolded protein response pathway in the yeast Kluyveromyces lactis. A comparative view among yeast species. Cells 7:106–7:106https://doi.org/10.3390/CELLS7080106
Hesham AEL, Wambui V, Ogola JOH, Maina JM (2014) Phylogenetic analysis of isolated biofuel yeasts based on 5.8S-ITS rDNA and D1/D2 26S rDNA sequences. J Genet Eng Biotechnol 12:37–43. https://doi.org/10.1016/j.jgeb.2014.01.001
Ho C, Grousl T, Shatz O, Jawed A, Ruger-Herreros C, Semmelink M, Zahn R, Richter K, Bukau B, Mogk A (2019) Cellular sequestrases maintain basal Hsp70 capacity ensuring balanced proteostasis. Nat Commun 10:4851. https://doi.org/10.1038/s41467-019-12868-1
IEA IEA, McCaherty J, Huschitt E, Schwarck R, Wilson C, Cooper G (2021) Essential Energy: 2021 Ethanol Industry Outlook. Renew Fuels Assoc Ellisvile, MO, USA
IEA IEA, Koehler N, McCaherty J, Wilson C, Cooper G, Schwarck R, Kemmet N, Baker R, Mcafee E, Drook R, Markham S, Sitzmann C, Friedberg J, Ricketts M, Christensen S, Boyle P, Harder S, Keiser K, Woodside C, Roe S, Wilson C (2020) Focus forward. 2020 RFA’s ethanol industry outlook. Renew Fuels Assoc Ellisvile, MO, USA
Jhariya U, Dafale NA, Srivastava S, Bhende RS, Kapley A, Purohit HJ (2021) Understanding ethanol tolerance mechanism in Saccharomyces cerevisiae to enhance the bioethanol production: current and future prospects. Bioenergy Res 14:670–688. https://doi.org/10.1007/s12155-020-10228-2
Jing H, Liu H, Zhang L, Gao J, Song H, Tan X (2018) Ethanol induces autophagy regulated by mitochondrial ROS in Saccharomyces cerevisiae. J Microbiol Biotechnol 28:1982–1991. https://doi.org/10.4014/jmb.1806.06014
Jung YR, Park JM, Heo SY, Hong WK, Lee SM, Oh BR, Park SM, Seo JW, Kim CH (2015) Cellulolytic enzymes produced by a newly isolated soil fungus Penicillium sp. TG2 with potential for use in cellulosic ethanol production. Renew Energy 76:66–71. https://doi.org/10.1016/j.renene.2014.10.064
Kádár Z, Szengyel Z, Réczey K (2004) Simultaneous saccharification and fermentation (SSF) of industrial wastes for the production of ethanol. Ind Crops Prod 20:103–110. https://doi.org/10.1016/j.indcrop.2003.12.015
Koutinas M, Menelaou M, Nicolaou EN (2014) Development of a hybrid fermentation-enzymatic bioprocess for the production of ethyl lactate from dairy waste. Bioresour Technol 165:343–349. https://doi.org/10.1016/j.biortech.2014.03.053
LaCroix RA, Palsson BO, Fiest AM (2017) A model for designing adaptive laboratory evolution experiments. Appl Enviromental Microbiol 83:1–14. https://doi.org/10.1128/AEM.03115-16
Lee S, Lee D, Kim T, Kim B (2005) Metabolic engineering of Escherichia coli for enhanced production of succinic acid, based on genome comparison and in silico gene knockout simulation. Appl Enviromental Microbiol 71:7880–7887. https://doi.org/10.1128/AEM.71.12.7880
Li L, Ye Y, Pan L, Zhu Y, Zheng S, Lin Y (2009) The induction of trehalose and glycerol in Saccharomyces cerevisiae in response to various stresses. Biochem Biophys Res Commun 4:778–783
Li P, Fu X, Chen M, Zhang L, Li S (2019) Proteomic profiling and integrated analysis with transcriptomic data bring new insights in the stress responses of Kluyveromyces marxianus after an arrest during high-temperature ethanol fermentation. Biotechnol Biofuels 12:1–13. https://doi.org/10.1186/s13068-019-1390-2
Li P, Fu X, Li S, Zhang L (2018) Engineering TATA-binding protein Spt15 to improve ethanol tolerance and production in Kluyveromyces marxianus. Biotechnol Biofuels 11:1–13. https://doi.org/10.1186/s13068-018-1206-9
Limayem A, Ricke SC (2012) Lignocellulosic biomass for bioethanol production: current perspectives, potential issues and future prospects. Prog Energy Combust Sci 38:449–467. https://doi.org/10.1016/j.pecs.2012.03.002
Limtong S, Sringiew C, Yongmanitchai W (2007) Production of fuel ethanol at high temperature from sugar cane juice by a newly isolated Kluyveromyces marxianus. Bioresour Technol 98:3367–3374. https://doi.org/10.1016/j.biortech.2006.10.044
Ma M, Liu ZL (2010) Mechanisms of ethanol tolerance in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 87:829–845. https://doi.org/10.1007/s00253-010-2594-3
Madeira-Jr JV, Gombert AK (2018) Towards high-temperature fuel ethanol production using Kluyveromyces marxianus: on the search for plug-in strains for the Brazilian sugarcane-based biorefinery. Biomass Bioenergy 119:217–228. https://doi.org/10.1016/j.biombioe.2018.09.010
Martin SR, Esposito V, Rios PDL, Pastore A, Temussi PA (2008) Cold denaturation of yeast Frataxin offers the clue to understand the effect of alcohols on protein stability. J Am Chem Soc 130:9963–9970. https://doi.org/10.1021/JA803280E
Mehmood N, Alayoubi R, Husson E, Jacquard C, Büchs J, Sarazin C, Gosselin I (2018) Kluyveromyces marxianus, an attractive yeast for ethanolic fermentation in the presence of imidazolium ionic liquids. Int J Mol Sci 19. https://doi.org/10.3390/ijms19030887
Ming M, Wang X, Lian L, Zhang H, Gao W, Zhu B, Low D (2019) Metabolic responses of Saccharomyces cerevisiae to ethanol stress using gas chromatography-mass spectrometry. Mol Omics 15:216. https://doi.org/10.1039/c9mo00055k
Mizushima N, Yoshimori T, Ohsumi Y (2011) The role of atg proteins in autophagosome formation. Annu Rev Cell Dev Biol 27:107–132. https://doi.org/10.1146/annurev-cellbio-092910-154005
Mo W, Wang M, Zhan R, Yu Y, He Y, Lu H (2019) Kluyveromyces marxianus developing ethanol tolerance during adaptive evolution with significant improvements of multiple pathways. Biotechnol Biofuels 12:1–15. https://doi.org/10.1186/s13068-019-1393-z
Moncan M, Mnich K, Blomme A, Almanza A, Samali A, Gorman AM (2021) Regulation of lipid metabolism by the unfolded protein response. J Cell Mol Med 25:1359–1370. https://doi.org/10.1111/jcmm.16255
Moreno AD, Ibarra D, Ballesteros I, González A, Ballesteros M (2013) Comparing cell viability and ethanol fermentation of the thermotolerant yeast Kluyveromyces marxianus and Saccharomyces cerevisiae on steam-exploded biomass treated with laccase. Bioresour Technol 135:239–245. https://doi.org/10.1016/j.biortech.2012.11.095
Naik SN, Goud VV, Rout PK, Dalai AK (2010) Production of first and second generation biofuels: a comprehensive review. Renew Sustain Energy Rev 14:578–597. https://doi.org/10.1016/j.rser.2009.10.003
Nakamura K, Shinomiya N, Orikasa Y, Oda Y (2012) Efficient production of ethanol from saccharified crops mixed with cheese whey by the flex yeast Kluyveromyces marxianus KD-15. Food Sci Technol Res 18:235–242. https://doi.org/10.3136/fstr.18.235
Navarro-Tapia E, Pérez-Torrado R, Querol A (2017) Ethanol effects involve non-canonical unfolded protein response activation in yeast cells. Front Microbiol 8:1–12. https://doi.org/10.3389/fmicb.2017.00383
Nikolaidis A, Andreadis M, Moschakis T (2017) Effect of heat, pH, ultrasonication and ethanol on the denaturation of whey protein isolate using a newly developed approach in the analysis of difference-UV spectra. Food Chem 232:425–433. https://doi.org/10.1016/j.foodchem.2017.04.022
Novitskiy G, Traore K, Wang L, Trush MA, Mezey E (2006) Effects of ethanol and acetaldehyde on reactive oxygen species production in rat hepatic stellate cells. Alcohol Clin Exp Res 30:1429–1435. https://doi.org/10.1111/j.1530-0277.2006.00171.x
Odat O, Matta S, Khalil H, Kampranis SC, Pfau R, Tsichlis PN, Makris AM (2007) Old yellow enzymes, highly homologous FMN oxidoreductases with modulating roles in oxidative stress and programmed cell death in yeast *. J Biol Chem 282:36010–36023. https://doi.org/10.1074/JBC.M704058200
Ortiz-Merino RA, Varela JA, Coughlan AY, Hoshida H, da Silveira WB, Wilde C, Kuijpers NGA, Geertman JM, Wolfe KH, Morrissey JP (2018) Ploidy variation in Kluyveromyces marxianus separates dairy and non-dairy isolates. Front Genet 9:1–16. https://doi.org/10.3389/fgene.2018.00094
Packer MS, Liu DR (2015) Methods for the directed evolution of proteins. Nat Rev Genet 16:379–394. https://doi.org/10.1038/nrg3927
Pennisi E (2013) The man who bottled evolution. Science 80(342):790–793
Pereira FB, Guimarães PMR, Teixeira JA, Domingues L (2011) Robust industrial Saccharomyces cerevisiae strains for very high gravity bio-ethanol fermentations. J Biosci Bioeng 112:130–136. https://doi.org/10.1016/j.jbiosc.2011.03.022
Pilap W, Thanonkeo S, Klanrit P, Thanonkeo P (2018) The potential of the newly isolated thermotolerant Kluyveromyces marxianus for high-temperature ethanol production using sweet sorghum juice. 3 Biotech 8 https://doi.org/10.1007/s13205-018-1161-y
Piškur J, Rozpedowska E, Polakova S, Merico A, Compagno C (2006) How did Saccharomyces evolve to become a good brewer? Trends Genet 22:183–186. https://doi.org/10.1016/j.tig.2006.02.002
Portnoy VA, Bezdan D, Zengler K (2011) Adaptive laboratory evolution—harnessing the power of biology for metabolic engineering. Curr Opin Biotechnol 22:590–594. https://doi.org/10.1016/j.copbio.2011.03.007
Read A, Schröder M (2021) The unfolded protein: an overview. Biology 10(5):384
Reed KB, Wagner JM, d’Oelsnitz S, Wiggers JM, Alper HS (2019) Improving ionic liquid tolerance in Saccharomyces cerevisiae through heterologous expression and directed evolution of an ILT1 homolog from Yarrowia lipolytica. J Ind Microbiol Biotechnol 46:1715–1724. https://doi.org/10.1007/s10295-019-02228-9
Reggiori F, Klionsky DJ (2013) Autophagic processes in yeast: mechanism, machinery and regulation. Genetics 194:341–361. https://doi.org/10.1534/genetics.112.149013
Rugthaworn P, Murata Y, Machida M, Apiwatanapiwat W, Hirooka A, Thanapase W, Dangjarean H, Ushiwaka S, Morimitsu K, Kosugi A, Arai T, Vaithanomsat P (2014) Growth inhibition of thermotolerant yeast, Kluyveromyces marxianus, in hydrolysates from cassava pulp. Appl Biochem Biotechnol 173:1197–1208. https://doi.org/10.1007/s12010-014-0906-2
Saini P, Beniwal A, Kokkiligadda A, Vij S (2018) Response and tolerance of yeast to changing environmental stress during ethanol fermentation. Process Biochem 72:1–12. https://doi.org/10.1016/j.procbio.2018.07.001
Saini P, Beniwal A, Kokkiligadda A, Vij S (2017) Evolutionary adaptation of Kluyveromyces marxianus strain for efficient conversion of whey lactose to bioethanol. Process Biochemhttps://doi.org/10.1016/j.procbio.2017.07.013
Sandoval-Nuñez D, Arellano-Plaza M, Gschaedler A, Arrizon J, Amaya-Delgado L (2018) A comparative study of lignocellulosic ethanol productivities by Kluyveromyces marxianus and Saccharomyces cerevisiae. Clean Technol Environ Policy 20:1491–1499. https://doi.org/10.1007/s10098-017-1470-6
Sansonetti S, Curcio S, Calabrò V, Iorio G (2009) Bio-ethanol production by fermentation of ricotta cheese whey as an effective alternative non-vegetable source. Biomass Bioenergy 33:1687–1692. https://doi.org/10.1016/j.biombioe.2009.09.002
Saratale GD, Saratale RG, Ghodake GS, Jiang YY, Chang JS, Shin HS, Kumar G (2017) Solid state fermentative lignocellulolytic enzymes production, characterization and its application in the saccharification of rice waste biomass for ethanol production: An integrated biotechnological approach. J Taiwan Inst Chem Eng 76:51–58. https://doi.org/10.1016/j.jtice.2017.03.027
Schabort DTWP, Letebele PK, Steyn L, Kilian SG, du Preez JC (2016) Differential RNA-seq, multi-network analysis and metabolic regulation analysis of Kluyveromyces marxianus reveals a compartmentalised response to xylose. PLoS One 11:e0156242. https://doi.org/10.1371/JOURNAL.PONE.0156242
Silveira FA, de Oliveira Soares DL, Bang KW, Balbino TR, de Moura Ferreira MA, Diniz RHS, de Lima LA, Brandão MM, Villas-Bôas SG, da Silveira WB (2020) Assessment of ethanol tolerance of Kluyveromyces marxianus CCT 7735 selected by adaptive laboratory evolution. Appl Microbiol Biotechnol 104:7483–7494. https://doi.org/10.1007/s00253-020-10768-9
Silveira WB, Passos FJV, Mantovani HC, Passos FML (2005) Ethanol production from cheese whey permeate by Kluyveromyces marxianus UFV-3: a flux analysis of oxido-reductive metabolism as a function of lactose concentration and oxygen levels. Enzyme Microb Technol 36:930–936. https://doi.org/10.1016/j.enzmictec.2005.01.018
Sims REH, Mabee W, Saddler JN, Taylor M (2010) An overview of second generation biofuel technologies. Bioresour Technol 101:1570–1580. https://doi.org/10.1016/j.biortech.2009.11.046
Šoštarić N, Arslan A, Carvalho B, Plech M, Voordeckers K, Verstrepen KJ, Van Noort V (2021) Integrated multi-omics analysis of mechanisms underlying yeast ethanol tolerance. J Proteome Res 20:3840–3852. https://doi.org/10.1021/acs.jproteome.1c00139
Stanley D, Bandara A, Fraser S, Chambers PJ, Stanley GA (2010) The ethanol stress response and ethanol tolerance of Saccharomyces cerevisiae. J Appl Microbiol 109:13–24. https://doi.org/10.1111/j.1365-2672.2009.04657.x
Stanley D, Fraser S, Chambers PJ, Rogers P, Stanley GA (2010) Generation and characterisation of stable ethanol-tolerant mutants of Saccharomyces cerevisiae. J Ind Microbiol Biotechnol 37:139–149. https://doi.org/10.1007/s10295-009-0655-3
Steensels J, Snoek T, Meersman E, Nicolino MP, Voordeckers K, Verstrepen KJ (2014) Improving industrial yeast strains: exploiting natural and artificial diversity. FEMS Microbiol Rev 38:947–995. https://doi.org/10.1111/1574-6976.12073
Tavares B, Felipe M das G de A, dos Santos JC, Pereira FM, Gomes SD, Sene L (2019) An experimental and modeling approach for ethanol production by Kluyveromyces marxianus in stirred tank bioreactor using vacuum extraction as a strategy to overcome product inhibition. Renew Energy 131:261–267https://doi.org/10.1016/j.renene.2018.07.030
Tinôco D, Genier HLA, da Silveira WB (2021) Technology valuation of cellulosic ethanol production by Kluyveromyces marxianus CCT 7735 from sweet sorghum bagasse at elevated temperatures. Renew Energy 173:188–196. https://doi.org/10.1016/j.renene.2021.03.132
Tokuyama K, Toya Y, Horinouchi T, Furusawa C, Matsuda F, Shimizu H (2018) Application of adaptive laboratory evolution to overcome a flux limitation in an Escherichia coli production strain. Biotechnol Bioeng 115:1542–1551. https://doi.org/10.1002/bit.26568
Tomás-Pejó E, Oliva JM, González A, Ballesteros I, Ballesteros M (2009) Bioethanol production from wheat straw by the thermotolerant yeast Kluyveromyces marxianus CECT 10875 in a simultaneous saccharification and fermentation fed-batch process. Fuel 88:2142–2147. https://doi.org/10.1016/j.fuel.2009.01.014
Trotter EW, Collinson EJ, Dawes IW, Grant CM (2006) Old yellow enzymes protect against acrolein toxicity in the yeast Saccharomyces cerevisiae. Appl Environ Microbiol 72:4885–4892. https://doi.org/10.1128/AEM.00526-06
Vamvakas SS, Kapolos J (2020) Factors affecting yeast ethanol tolerance and fermentation efficiency. World J Microbiol Biotechnol 36:1–8. https://doi.org/10.1007/s11274-020-02881-8
Vriesekoop F, Haass C, Pamment NB (2009) The role of acetaldehyde and glycerol in the adaptation to ethanol stress of Saccharomyces cerevisiae and other yeasts. FEMS Yeast Res 9:365–371. https://doi.org/10.1111/j.1567-1364.2009.00492.x
Wang D, Wu D, Yang X, Hong J (2018) Transcriptomic analysis of thermotolerant yeast: Kluyveromyces marxianus in multiple inhibitors tolerance. RSC Adv 8:14177–14192. https://doi.org/10.1039/c8ra00335a
Wu WH, Hung WC, Lo KY, Chen YH, Wan HP, Cheng KC (2016) Bioethanol production from taro waste using thermo-tolerant yeast Kluyveromyces marxianus K21. Bioresour Technol 201:27–32. https://doi.org/10.1016/j.biortech.2015.11.015
Yan J, Wei Z, Wang Q, He M, Li S, Irbis C (2015) Bioethanol production from sodium hydroxide/hydrogen peroxide-pretreated water hyacinth via simultaneous saccharification and fermentation with a newly isolated thermotolerant Kluyveromyces marxianus strain. Bioresour Technol 193:103–109. https://doi.org/10.1016/j.biortech.2015.06.069
Yan T, Zhao Y (2020) Acetaldehyde induces phosphorylation of dynamin-related protein 1 and mitochondrial dysfunction via elevating intracellular ROS and Ca2+ levels. Redox Biol 28:101381. https://doi.org/10.1016/j.redox.2019.101381
Yang KM, Lee NR, Woo JM, Choi W, Zimmermann M, Blank LM, Park JB (2012) Ethanol reduces mitochondrial membrane integrity and thereby impacts carbon metabolism of Saccharomyces cerevisiae. FEMS Yeast Res 12:675–684. https://doi.org/10.1111/j.1567-1364.2012.00818.x
Yoshida M, Kato S, Fukuda S, Izawa S (2021) Acquired resistance to severe ethanol stress in Saccharomyces cerevisiae Protein Quality Control. Appl Environ Microbiol 87:1–17. https://doi.org/10.1128/AEM.02353-20
Yu CY, Jiang BH, Duan KJ (2013) Production of bioethanol from carrot pomace using the thermotolerant yeast Kluyveromyces marxianus. Energies 6:1794–1801. https://doi.org/10.3390/en6031794
Yuan WJ, Chang BL, Ren JG, Liu JP, Bai FW, Li YY (2012) Consolidated bioprocessing strategy for ethanol production from Jerusalem artichoke tubers by Kluyveromyces marxianus under high gravity conditions. J Appl Microbiol 112:38–44. https://doi.org/10.1111/j.1365-2672.2011.05171.x
Zhao XQ, Bai FW (2009) Mechanisms of yeast stress tolerance and its manipulation for efficient fuel ethanol production. J Biotechnol 144:23–30. https://doi.org/10.1016/j.jbiotec.2009.05.001
Zoppellari F, Bardi L (2013) Production of bioethanol from effluents of the dairy industry by Kluyveromyces marxianus. N Biotechnol 30:607–613. https://doi.org/10.1016/j.nbt.2012.11.017
Funding
This work was supported by the Brazilian National Council for Scientific and Technological Development (CNPq); the Foundation for Research Support of the State of Minas Gerais (FAPEMIG); and the Coordination for the Improvement of Higher Education Personnel (CAPES, Finance Code 001)
Author information
Authors and Affiliations
Contributions
M.A.M.F., F.A.S., and W.B.S. wrote the review. W.B.S. designed the structure of the review. M.A.M.F. edited the review and elaborated the figures. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have read and approved the manuscript for publication.
Conflict of interest
The authors have declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Moura Ferreira, M.A., da Silveira, F.A. & da Silveira, W.B. Ethanol stress responses in Kluyveromyces marxianus: current knowledge and perspectives. Appl Microbiol Biotechnol 106, 1341–1353 (2022). https://doi.org/10.1007/s00253-022-11799-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-11799-0