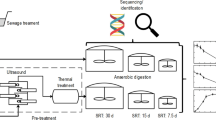
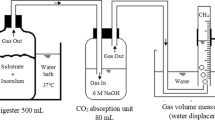
Abstract
Strategies to enhance process performance of anaerobic digestion remain of key importance to promote wider usage of this technology for integrated resource recovery from organic waste streams. Continuous inoculation of the microbial community in the digester via the feedstock could be such a cost-effective strategy. Here, anaerobic digestion of fresh waste activated sludge (WAS) was compared with sterilized WAS in response to two common process disturbances, i.e. organic overloading and increasing levels of salts, to determine the importance of feedstock inoculation. A pulse in the organic loading rate severely impacted process stability of the digesters fed sterile WAS, with a 92 ± 45% decrease in methane production, compared to a 42 ± 31% increase in the digesters fed fresh WAS, relative to methane production before the pulse. Increasing salt pulses did not show a clear difference in process stability between the digesters fed fresh and sterile WAS, and process recovery was obtained even at the highest salt pulse of 25 g Na+ L−1. Feedstock sterilization through thermal pretreatment strongly impacted the microbial community in the digesters. In conclusion, feedstock thermal pretreatment strongly impacted anaerobic digestion process stability, due to feedstock inoculation and compositional modification.





Similar content being viewed by others
References
Anthonisen AC, Loehr RC, Prakasam TBS, Srinath EG (1976) Inhibition of nitrification by ammonia and nitrous acid. J Water Pollut Control Fed 48(5):835–852
Anukam A, Mohammadi A, Naqvi M, Granström K (2019) A review of the chemistry of anaerobic digestion: methods of accelerating and optimizing process efficiency. Processes 7(8):504
Appels L, Baeyens J, Degrève J, Dewil R (2008) Principles and potential of the anaerobic digestion of waste-activated sludge. Prog Energy Combust 34(6):755–781. https://doi.org/10.1016/j.pecs.2008.06.002
Boon N, De Windt W, Verstraete W, Top EM (2002) Evaluation of nested PCR-DGGE (denaturing gradient gel electrophoresis) with group-specific 16S rRNA primers for the analysis of bacterial communities from different wastewater treatment plants. FEMS Microbiol Ecol 39(2):101–112. https://doi.org/10.1111/j.1574-6941.2002.tb00911.x
Briones A, Raskin L (2003) Diversity and dynamics of microbial communities in engineered environments and their implications for process stability. Curr Opin Biotechnol 14(3):270–276. https://doi.org/10.1016/s0958-1669(03)00065-x
Chen Y, Cheng JJ, Creamer KS (2008) Inhibition of anaerobic digestion process: a review. Bioresour Technol 99(10):4044–4064. https://doi.org/10.1016/j.biortech.2007.01.057
Conklin A, Stensel HD, Ferguson J (2006) Growth kinetics and competition between Methanosarcina and Methanosaeta in mesophilic anaerobic digestion. Water Environ Res 78(5):486–496. https://doi.org/10.2175/106143006x95393
Connelly S, Shin SG, Dillon RJ, Ijaz UZ, Quince C, Sloan WT, Collins G (2017) Bioreactor scalability: laboratory-scale bioreactor design influences performance, ecology, and community physiology in expanded granular sludge bed bioreactors. Front Microbiol 8:15. https://doi.org/10.3389/fmicb.2017.00664
Daelman MRJ, van Voorthuizen EM, van Dongen UGJM, Volcke EIP, van Loosdrecht MCM (2012) Methane emission during municipal wastewater treatment. Water Res 46(11):3657–3670. https://doi.org/10.1016/j.watres.2012.04.024
De Meester S, Demeyer J, Velghe F, Peene A, Van Langenhove H, Dewulf J (2012) The environmental sustainability of anaerobic digestion as a biomass valorization technology. Bioresour Technol 121:396–403. https://doi.org/10.1016/j.biortech.2012.06.109
De Vrieze J, Verstraete W (2016) Perspectives for microbial community composition in anaerobic digestion: from abundance and activity to connectivity. Environ Microbiol 18(9):2797–2809. https://doi.org/10.1111/1462-2920.13437
De Vrieze J, Raport L, Willems B, Verbrugge S, Volcke E, Meers E, Angenent LT, Boon N (2015a) Inoculum selection influences the biochemical methane potential of agro-industrial substrates. Microb Biotechnol 8(5):776–786. https://doi.org/10.1111/1751-7915.12268
De Vrieze J, Saunders AM, He Y, Fang J, Nielsen PH, Verstraete W, Boon N (2015b) Ammonia and temperature determine potential clustering in the anaerobic digestion microbiome. Water Res 75(0):312–323. https://doi.org/10.1016/j.watres.2015.02.025
De Vrieze J, Christiaens MER, Verstraete W (2017) The microbiome as engineering tool: manufacturing and trading between microorganisms. New Biotechnol 39(Part B):206–214. https://doi.org/10.1016/j.nbt.2017.07.001
De Vrieze J, Pinto AJ, Sloan WT, Ijaz UZ (2018) The active microbial community more accurately reflects the anaerobic digestion process: 16S rRNA (gene) sequencing as a predictive tool. Microbiome 6:13–13. https://doi.org/10.1186/s40168-018-0449-9
Demitry ME (2016) Anaerobic digestion process stability and the extension of the ADM1 for municipal sludge co- digested with bakery waste. All Graduate Theses and Dissertations. 4945. https://digitalcommons.usu.edu/etd/4945
Duran M, Tepe N, Yurtsever D, Punzi VL, Bruno C, Mehta RJ (2006) Bioaugmenting anaerobic digestion of biosolids with selected strains of Bacillus, Pseudomonas, and Actinomycetes species for increased methanogenesis and odor control. Appl Microbiol Biotechnol 73(4):960–966. https://doi.org/10.1007/s00253-006-0548-6
Eftaxias A, Diamantis V, Aivasidis A (2018) Anaerobic digestion of thermal pre-treated emulsified slaughterhouse wastes (TESW): effect of trace element limitation on process efficiency and sludge metabolic properties. Waste Manag 76:357–363. https://doi.org/10.1016/j.wasman.2018.02.032
Eftaxias A, Gkalimanis P, Koskinari MD, Koumara A, Diamantis V, Aivasidis A (2019) Use of diluted poultry manure as a low-cost emulsifier for anaerobic digestion of used cooking oil. Water Pract Technol 14(4):937–945. https://doi.org/10.2166/wpt.2019.074
Enerdate (2018) Global Energy Statistical Yearbook 2018
Esser DK-H, Marx DWH, Lisowsky PDT (2006) DNA decontamination: novel DNA-ExitusPlus™ in comparison with conventional reagents. BioTechniques 40(2):238–239. https://doi.org/10.2144/06402af01
Feijoo G, Soto M, Méndez R, Lema JM (1995) Sodium inhibition in the anaerobic digestion process: antagonism and adaptation phenomena. Enzym Microb Technol 17(2):180–188. https://doi.org/10.1016/0141-0229(94)00011-F
Fotidis IA, Wang H, Fiedel NR, Luo G, Karakashev DB, Angelidaki I (2014) Bioaugmentation as a solution to increase methane production from an ammonia-rich substrate. Environ Sci Technol 48(13):7669–7676. https://doi.org/10.1021/es5017075
Fountoulakis MS, Petousi I, Manios T (2010) Co-digestion of sewage sludge with glycerol to boost biogas production. Waste Manag 30(10):1849–1853. https://doi.org/10.1016/j.wasman.2010.04.011
Garcia ML, Angenent LT (2009) Interaction between temperature and ammonia in mesophilic digesters for animal waste treatment. Water Res 43(9):2373–2382. https://doi.org/10.1016/j.watres.2009.02.036
González Arias J, Sánchez ME, Gómez X (2018) Enhancing anaerobic digestion: the effect of carbon conductive materials, vol 4
Greenberg AE, Clesceri LS, Eaton AD (1992) Standard methods for the examination of water and wastewater. American Public Health Association Publications, Washington
Hagos K, Zong J, Li D, Liu C, Lu X (2017) Anaerobic co-digestion process for biogas production: progress, challenges and perspectives. Renew Sust Energ Rev 76:1485–1496. https://doi.org/10.1016/j.rser.2016.11.184
Hierholtzer A, Akunna JC (2014) Modelling start-up performance of anaerobic digestion of saline-rich macro-algae. Water Sci Technol 69(10):2059–2065. https://doi.org/10.2166/wst.2014.100
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54(2):427–432. https://doi.org/10.2307/1934352
Holm-Nielsen JB, Lomborg CJ, Oleskowicz-Popiel P, Esbensen KH (2008) On-line near infrared monitoring of glycerol-boosted anaerobic digestion processes: evaluation of process analytical technologies. Biotechnol Bioeng 99(2):302–313. https://doi.org/10.1002/bit.21571
Holm-Nielsen JB, Al Seadi T, Oleskowicz-Popiel P (2009) The future of anaerobic digestion and biogas utilization. Bioresour Technol 100(22):5478–5484. https://doi.org/10.1016/j.biortech.2008.12.046
Hubenov V, Mihaylova SN, Simeonov I (2015) Anaerobic co-digestion of waste fruits and vegetables and swine manure in a pilot-scale bioreactor. Bulg Chem Commun 47(3):788–792
Ismail SB, de La Parra CJ, Temmink H, van Lier JB (2010) Extracellular polymeric substances (EPS) in upflow anaerobic sludge blanket (UASB) reactors operated under high salinity conditions. Water Res 44(6):1909–1917. https://doi.org/10.1016/j.watres.2009.11.039
Kacprzak A, Krzystek L, Ledakowicz S (2010) Co-digestion of agricultural and industrial wastes. Chem Pap 64(2):127–131. https://doi.org/10.2478/s11696-009-0108-5
Kirkegaard RH, McIlroy SJ, Kristensen JM, Nierychlo M, Karst SM, Dueholm MS, Albertsen M, Nielsen PH (2017) The impact of immigration on microbial community composition in full-scale anaerobic digesters. Sci Rep 7:11. https://doi.org/10.1038/s41598-017-09303-0
Klindworth A, Pruesse E, Schweer T, Peplies J, Quast C, Horn M, Glockner FO (2013) Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res 41(1):11. https://doi.org/10.1093/nar/gks808
Kugelman IJ, McCarty PL (1965) Cation toxicity and stimulation in anaerobic waste treatment. J Water Pollut Control Fed 37(1):97–116
Langer SG, Ahmed S, Einfalt D, Bengelsdorf FR, Kazda M (2015) Functionally redundant but dissimilar microbial communities within biogas reactors treating maize silage in co-fermentation with sugar beet silage. Microb Biotechnol 8(5):828–836. https://doi.org/10.1111/1751-7915.12308
Lebiocka M, Montusiewicz A, Cydzik-Kwiatkowska A (2018) Effect of bioaugmentation on biogas yields and kinetics in anaerobic digestion of sewage sludge. Int J Environ Res Public Health 15(8):1717. https://doi.org/10.3390/ijerph15081717
Li Y, Li L, Sun Y, Yuan Z (2018) Bioaugmentation strategy for enhancing anaerobic digestion of high C/N ratio feedstock with methanogenic enrichment culture. Bioresour Technol 261:188–195. https://doi.org/10.1016/j.biortech.2018.02.069
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15(12):550–550. https://doi.org/10.1186/s13059-014-0550-8
Lynch N, Daniels L, Parkin GF (1987) Bioaugmentation of stressed anaerobic filters with methanogenic enrichment cultures. In: Proceedings of the 42nd Industrial Waste Conference. (pp. 285-296). West Lafayette, Indiana: Purdue University
Ma J, Van Wambeke M, Carballa M, Verstraete W (2008) Improvement of the anaerobic treatment of potato processing wastewater in a UASB reactor by co-digestion with glycerol. Biotechnol Lett 30(5):861–867. https://doi.org/10.1007/s10529-007-9617-x
Mata-Alvarez J, Mace S, Llabres P (2000) Anaerobic digestion of organic solid wastes. An overview of research achievements and perspectives. Bioresour Technol 74(1):3–16. https://doi.org/10.1016/s0960-8524(00)00023-7
Mata-Alvarez J, Dosta J, Macé S, Astals S (2011) Codigestion of solid wastes: a review of its uses and perspectives including modeling. Crit Rev Biotechnol 31(2):99–111. https://doi.org/10.3109/07388551.2010.525496
McMahon KD, Martin HG, Hugenholtz P (2007) Integrating ecology into biotechnology. Curr Opin Biotechnol 18(3):287–292. https://doi.org/10.1016/j.copbio.2007.04.007
McMurdie PJ, Holmes S (2013) Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS One 8(4):e61217. https://doi.org/10.1371/journal.pone.0061217
McMurdie PJ, Holmes S (2014) Waste not, want not: why rarefying microbiome data is inadmissible. PLoS Comput Biol 10(4):12. https://doi.org/10.1371/journal.pcbi.1003531
Mischopoulou M, Kalamaras SD, Naidis P, Kotsopoulos TA, Samaras P (2017) Start-up and steady-state results of a UASB reactor treating high pH baker's yeast molasses wastewater for methane production. J Chem Technol Biotechnol 92(4):775–780. https://doi.org/10.1002/jctb.5055
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara R.B., Simpson GL, Solymos P, Stevens MHH, Wagner H (2016) Vegan: community ecology package. R package version 2.3–4
Papadimitriou EK (2010) Hydrolysis of organic matter during autoclaving of commingled household waste. Waste Manag 30(4):572–582. https://doi.org/10.1016/j.wasman.2009.11.019
Pianka ER (1970) On r- and K-Selection. Am Nat 104(940):592–597
R Development Core Team (2013) R: a language and environment for statistical computing, 3.0 edn. R Foundation for Statistical Computing, Vienna
Rinzema A, van Lier J, Lettinga G (1988) Sodium inhibition of acetoclastic methanogens in granular sludge from a UASB reactor. Enzym Microb Technol 10(1):24–32. https://doi.org/10.1016/0141-0229(88)90094-4
Sanders HL (1968) Marine benthic diversity: a comparative study. Am Nat 102(925):243–282. https://doi.org/10.1086/282541
Saravanane R, Murthy DVS, Krishnaiah K (2001a) Bioaugmentation and anaerobic treatment of pharmaceutical effluent in fluidized bed reactor. J Environ Sci Health, Part A 36(5):779–791. https://doi.org/10.1081/ESE-100103760
Saravanane R, Murthy DVS, Krishnaiah K (2001b) Bioaugmentation and treatment of cephalexin drug-based pharmaceutical effluent in an upflow anaerobic fluidized bed system. Bioresour Technol 76(3):279–281. https://doi.org/10.1016/S0960-8524(00)00121-8
Scarlat N, Dallemand J-F, Fahl F (2018) Biogas: developments and perspectives in Europe. Renew Energy 129:457–472. https://doi.org/10.1016/j.renene.2018.03.006
Schauer-Gimenez AE, Zitomer DH, Maki JS, Struble CA (2010) Bioaugmentation for improved recovery of anaerobic digesters after toxicant exposure. Water Res 44(12):3555–3564. https://doi.org/10.1016/j.watres.2010.03.037
Shi X, Lin J, Zuo J, Li P, Li X, Guo X (2017) Effects of free ammonia on volatile fatty acid accumulation and process performance in the anaerobic digestion of two typical bio-wastes. J Environ Sci 55:49–57. https://doi.org/10.1016/j.jes.2016.07.006
Shin J, Cho S-K, Lee J, Hwang K, Chung JW, Jang H-N, Shin SG (2019) Performance and microbial community dynamics in anaerobic digestion of waste activated sludge: impact of immigration. Energies 12(3):573
Sloan WT, Lunn M, Woodcock S, Head IM, Nee S, Curtis TP (2006) Quantifying the roles of immigration and chance in shaping prokaryote community structure. Environ Microbiol 8(4):732–740. https://doi.org/10.1111/j.1462-2920.2005.00956.x
Sundberg C, Al-Soud WA, Larsson M, Alm E, Yekta SS, Svensson BH, Sørensen SJ, Karlsson A (2013) 454 pyrosequencing analyses of bacterial and archaeal richness in 21 full-scale biogas digesters. FEMS Microbiol Ecol 85(3):612–626. https://doi.org/10.1111/1574-6941.12148
Tampio E, Ervasti S, Paavola T, Heaven S, Banks C, Rintala J (2014) Anaerobic digestion of autoclaved and untreated food waste. Waste Manag 34(2):370–377. https://doi.org/10.1016/j.wasman.2013.10.024
Tepe N, Yurtsever D, Duran M, Mehta RJ, Bruno C, Punzi VL (2008) Odor control during post-digestion processing of biosolids through bioaugmentation of anaerobic digestion. Water Sci Technol 57(4):589–594. https://doi.org/10.2166/wst.2008.008
Venkiteshwaran K, Bocher B, Maki J, Zitomer D (2016) Relating anaerobic digestion microbial community and process function. Microbiol Insights 8(Suppl 2):37–44. https://doi.org/10.4137/MBI.S33593
Wang S, Hou X, Su H (2017) Exploration of the relationship between biogas production and microbial community under high salinity conditions. Sci Rep 7:1149. https://doi.org/10.1038/s41598-017-01298-y
Westerholm M, Schnürer A (2019) Microbial responses to different operating practices for biogas production systems biogas [working title]. IntechOpen
Zhang W, Werner JJ, Agler MT, Angenent LT (2014) Substrate type drives variation in reactor microbiomes of anaerobic digesters. Bioresour Technol 151:397–401. https://doi.org/10.1016/j.biortech.2013.10.004
Zhang Y, Alam MA, Kong X, Wang Z, Li L, Sun Y, Qi W (2017a) Effect of salinity on the microbial community and performance on anaerobic digestion of marine macroalgae. J Chem Technol Biotechnol 92(9):2392–2399. https://doi.org/10.1002/jctb.5246
Zhang Y, Li L, Kong X, Zhen F, Wang Z, Sun Y, Dong P, Lv P (2017b) Inhibition effect of sodium concentrations on the anaerobic digestion performance of Sargassum species. Energy Fuel 31(7):7101–7109. https://doi.org/10.1021/acs.energyfuels.7b00557
Acknowledgements
The authors would like to thank Tim Lacoere for his contribution to the molecular analyses and Aquafin for their assistance with sample collection. We thank Pieter Candry and Karel Folens for critically reading the manuscript.
Funding
Cindy Ka Y Law received support from the ELECTRA project “Electricity Driven Low Energy and Chemical Input Technology for Accelerated Bioremediation” financed by the H2020 of the European Commission under Grant number GA 826244. Jo De Vrieze is supported as a postdoctoral fellow by the Research Foundation Flanders (FWO-Vlaanderen).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Law, C., De Henau, R. & De Vrieze, J. Feedstock thermal pretreatment selectively steers process stability during the anaerobic digestion of waste activated sludge. Appl Microbiol Biotechnol 104, 3675–3686 (2020). https://doi.org/10.1007/s00253-020-10472-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10472-8