Abstract
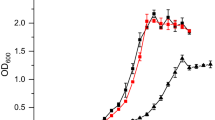
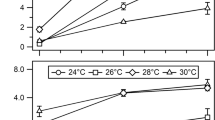
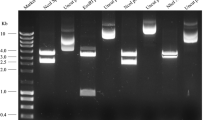
The advantage of using acetogens such as Acetobacterium woodii as biocatalysts converting the cheap substrate and greenhouse gas carbon dioxide (CO2) into value-added chemicals comes together with the disadvantage of a low overall ATP gain due to the bioenergetics associated with the Wood-Ljungdahl pathway. Expanding the product spectrum of recombinant A. woodii strains to compounds with high ATP-demanding biosynthesis is therefore challenging. As a least invasive strategy for improved ATP generation, the exploitation of the arginine deiminase pathway (ADI) was examined under native conditions and via using heterologously expressed genes in A. woodii. Several promoters were analyzed for application of different gene expression levels in A. woodii using β-glucuronidase assays. Heterologous expression of the ADI pathway genes from Clostridium autoethanogenum was controlled using either the constitutive pta-ack promoter from Clostridium ljungdahlii or a tightly regulated tetracycline-inducible promoter Ptet. Unlike constitutive expression, only induced expression of the ADI pathway genes led to a 36% higher maximal OD600 when using arginine (OD600 3.4) as nitrogen source and a 52% lower acetate yield per biomass compared to cells growing with yeast extract as nitrogen source (OD600 2.5). In direct comparison, a 69% higher maximal OD600 and about 60% lower acetate yield per biomass in induced to non-induced recombinant A. woodii cells was noticed when using arginine. Our data suggests the application of the ADI pathway in A. woodii for expanding the product spectrum to compounds with high ATP-demanding biosynthesis.




Similar content being viewed by others
References
Al-Hinai MA, Fast AG, Papoutsakis ET (2012) Novel system for efficient isolation of Clostridium double-crossover allelic exchange mutants enabling markerless chromosomal gene deletions and DNA integration. Appl Environ Microbiol 78:8112–8121. https://doi.org/10.1128/AEM.02214-12
Banerjee A, Leang C, Ueki T, Nevin KP, Lovley DR (2014) Lactose-inducible system for metabolic engineering of Clostridium ljungdahlii. Appl Environ Microbiol 80:2410–2416. https://doi.org/10.1128/AEM.03666-13
Bengelsdorf FR, Beck MH, Erz C, Hoffmeister S, Karl MM, Riegler P, Wirth S, Poehlein A, Weuster-Botz D, Dürre P (2018) Bacterial anaerobic synthesis gas (syngas) and CO2 + H2 fermentation. In: Advances in Applied Microbiology, vol 103. Elsevier, Amsterdam, pp 143–221
Braun K, Gottschalk G (1981) Effect of molecular hydrogen and carbon dioxide on chemo-organotrophic growth of Acetobacterium woodii and Clostridium aceticum. Arch Microbiol 128:294–298
Carrier MJ, Nugent ME, Tacon WCA, Primrose SB (1983) High expression of cloned genes in E. coli and its consequences. Trends Biotechnol 1:109–113. https://doi.org/10.1016/0167-7799(83)90033-1
Davison DC, Elliott WH (1952) Enzymic reaction between arginine and fumarate in plant and animal tissues. Nature 169:313–314. https://doi.org/10.1038/169313a0
de Souza Pinto Lemgruber R, Valgepea K, Tappel R, Behrendorff JB, Palfreyman RW, Plan M, Hodson MP, Simpson SD, Nielsen LK, Köpke M, Marcellin E (2019) Systems-level engineering and characterisation of Clostridium autoethanogenum through heterologous production of poly-3-hydroxybutyrate (PHB). Metab Eng 53:14–23. https://doi.org/10.1016/j.ymben.2019.01.003
Emerson DF, Woolston BM, Liu N, Donnelly M, Currie DH, Stephanopoulos G (2018) Enhancing hydrogen-dependent growth of and carbon dioxide fixation by Clostridium ljungdahlii through nitrate supplementation. Biotechnol Bioeng 116:294–306. https://doi.org/10.1002/bit.26847
Erz C (2017) 2,3-Butanediol production using acetogenic bacteria. PhD thesis, University of Ulm, Germany
Flitsch SK (2016) Identifikation und Charakterisierung von Regulatoren der Lösungsmittelbildung in Clostridium acetobutylicum. PhD thesis, University of Ulm, Germany
Flüchter S, Follonier S, Schiel-Bengelsdorf B, Bengelsdorf FR, Zinn M, Dürre P (2019) Anaerobic production of poly(3-hydroxybutyrate) and its precursor 3-hydroxybutyrate from synthesis gas by autotrophic clostridia. Biomacromolecules. https://doi.org/10.1021/acs.biomac.9b00342
Green MR, Sambrook J (2012) Molecular cloning: a laboratory manual, 4th edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Grimm V, Gleinser M, Neu C, Zhurina D, Riedel CU (2014) Expression of fluorescent proteins in bifidobacteria for analysis of host-microbe interactions. Appl Environ Microbiol 80:2842–2850. https://doi.org/10.1128/AEM.04261-13
Groher A, Weuster-Botz D (2016) Comparative reaction engineering analysis of different acetogenic bacteria for gas fermentation. J Biotechnol 228:82–94. https://doi.org/10.1016/j.jbiotec.2016.04.032
Heap JT, Pennington OJ, Cartman ST, Minton NP (2009) A modular system for Clostridium shuttle plasmids. J Microbiol Methods 78:79–85. https://doi.org/10.1016/j.mimet.2009.05.004
Hoffmeister S, Gerdom M, Bengelsdorf FR, Linder S, Flüchter S, Öztürk H, Blümke W, May A, Fischer R-J, Bahl H, Dürre P (2016) Acetone production with metabolically engineered strains of Acetobacterium woodii. Metab Eng 36:37–47. https://doi.org/10.1016/j.ymben.2016.03.001
Jackson MJ, Beaudet AL, O’Brien WE (1986) Mammalian urea cycle enzymes. Annu Rev Genet 20:431–464. https://doi.org/10.1146/annurev.ge.20.120186.002243
LanzaTech (2018) World’s first commercial waste gas to ethanol plant starts up. https://www.lanzatech.com/2018/06/08/worlds-first-commercial-waste-gas-ethanol-plant-starts/2018/. Accessed 11 July 2019
Larsen R, Buist G, Kuipers OP, Kok J (2004) ArgR and AhrC are both required for regulation of arginine metabolism in Lactococcus lactis. J Bacteriol 186:1147–1157. https://doi.org/10.1128/JB.186.4.1147-1157.2004
Larsen R, Kok J, Kuipers OP (2005) Interaction between ArgR and AhrC controls regulation of arginine metabolism in Lactococcus lactis. J Biol Chem 280:19319–19330. https://doi.org/10.1074/jbc.M413983200
Liu S-Q, Pilone GJ (1998) A review: arginine metabolism in wine lactic acid bacteria and its practical significance. J Appl Microbiol 84:315–327. https://doi.org/10.1046/j.1365-2672.1998.00350.x
Monod J (1949) The growth of bacterial cultures. Annu Rev Microbiol 3:371–394. https://doi.org/10.1146/annurev.mi.03.100149.002103
Mumberg D, Müller R, Funk M (1994) Regulatable promoters of Saccharomyces cerevisiae: comparison of transcriptional activity and their use for heterologous expression. Nucleic Acids Res 22:5767–5768
Noens EEE, Lolkema JS (2017) Convergent evolution of the arginine deiminase pathway: the ArcD and ArcE arginine/ornithine exchangers. Microbiol Open 6:e00412. https://doi.org/10.1002/mbo3.412
Noens EEE, Kaczmarek MB, Żygo M, Lolkema JS (2015) ArcD1 and ArcD2 arginine/ornithine exchangers encoded in the arginine deiminase pathway gene cluster of Lactococcus lactis. J Bacteriol 197:3545–3553. https://doi.org/10.1128/JB.00526-15
O’Brien J, Wright GD (2011) An ecological perspective of microbial secondary metabolism. Curr Opin Biotechnol 22:552–558. https://doi.org/10.1016/j.copbio.2011.03.010
Poehlein A, Schmidt S, Kaster A-K, Goenrich M, Vollmers J, Thürmer A, Bertsch J, Schuchmann K, Voigt B, Hecker M, Daniel R, Thauer RK, Gottschalk G, Müller V (2012) An ancient pathway combining carbon dioxide fixation with the generation and utilization of a sodium ion gradient for ATP synthesis. PLoS One 7:e33439. https://doi.org/10.1371/journal.pone.0033439
Poolman B, Driessen AJ, Konings WN (1987) Regulation of arginine-ornithine exchange and the arginine deiminase pathway in Streptococcus lactis. J Bacteriol 169:5597–5604
Ransom EM, Ellermeier CD, Weiss DS (2015) Use of mCherry red fluorescent protein for studies of protein localization and gene expression in Clostridium difficile. Appl Environ Microbiol 81:1652–1660. https://doi.org/10.1128/AEM.03446-14
Ratner S (1954) Urea synthesis and metabolism of arginine and citrulline. Adv Enzymol Relat Subj Biochem 15:319–387
Schiel-Bengelsdorf B, Dürre P (2012) Pathway engineering and synthetic biology using acetogens. FEBS Lett 586:2191–2198. https://doi.org/10.1016/j.febslet.2012.04.043
Schiel-Bengelsdorf B, Montoya J, Linder S, Dürre P (2013) Butanol fermentation. Environ Technol 34:1691–1710. https://doi.org/10.1080/09593330.2013.827746
Schrumpf B, Schwarzer A, Kalinowski J, Pühler A, Eggeling L, Sahm H (1991) A functionally split pathway for lysine synthesis in Corynebacterium glutamicum. J Bacteriol 173:4510–4516
Schuchmann K, Müller V (2014) Autotrophy at the thermodynamic limit of life: a model for energy conservation in acetogenic bacteria. Nat Rev Microbiol 12:809–821. https://doi.org/10.1038/nrmicro3365
Sorek R, Cossart P (2010) Prokaryotic transcriptomics: a new view on regulation, physiology and pathogenicity. Nat Rev Genet 11:9–16. https://doi.org/10.1038/nrg2695
Trip H, Mulder NL, Lolkema JS (2013) Cloning, expression, and functional characterization of secondary amino acid transporters of Lactococcus lactis. J Bacteriol 195:340–350. https://doi.org/10.1128/JB.01948-12
Valgepea K, Loi KQ, Behrendorff JB, de Lemgruber RSP, Plan M, Hodson MP, Köpke M, Nielsen LK, Marcellin E (2017) Arginine deiminase pathway provides ATP and boosts growth of the gas-fermenting acetogen Clostridium autoethanogenum. Metab Eng 41:202–211. https://doi.org/10.1016/j.ymben.2017.04.007
Wagner S, Klepsch MM, Schlegel S, Appel A, Draheim R, Tarry M, Hogbom M, van Wijk KJ, Slotboom DJ, Persson JO, de Gier J-W (2008) Tuning Escherichia coli for membrane protein overexpression. Proc Natl Acad Sci U S A 105:14371–14376. https://doi.org/10.1073/pnas.0804090105
Woolston BM, Emerson DF, Currie DH, Stephanopoulos G (2018) Rediverting carbon flux in Clostridium ljungdahlii using CRISPR interference (CRISPRi). Metab Eng 48:243–253. https://doi.org/10.1016/j.ymben.2018.06.006
Zhang J, Liu Y-J, Cui G-Z, Cui Q (2015a) A novel arabinose-inducible genetic operation system developed for Clostridium cellulolyticum. Biotechnol Biofuels 8:36. https://doi.org/10.1186/s13068-015-0214-2
Zhang Y, Grosse-Honebrink A, Minton NP (2015b) A universal mariner transposon system for forward genetic studies in the genus Clostridium. PLoS One 10:e0122411. https://doi.org/10.1371/journal.pone.0122411
Zúñiga M, Pérez G, González-Candelas F (2002) Evolution of arginine deiminase (ADI) pathway genes. Mol Phylogenet Evol 25:429–444
Acknowledgments
We thank Eric Ransom and David Weiss for kindly providing plasmid pDSW1728. We thank Sophie Moormann, Julia Nothelfer, Judith Cammerer, and Lucille Exler for conducting enzymatic assays. The provision of the theophylline riboswitch sequence by Gerd Seibold and Christian Rückert is gratefully acknowledged. We thank the reviewers of this paper for constructive criticism and numerous helpful comments.
Funding
This study was funded by the ERA-IB5 program (project CO2CHEM, grant 031A566A).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Matthias H. Beck declares that he has no conflict of interest. Maximilian Flaiz declares that he has no conflict of interest. Frank R. Bengelsdorf declares that he has no conflict of interest. Peter Dürre declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 519 kb)
Rights and permissions
About this article
Cite this article
Beck, M.H., Flaiz, M., Bengelsdorf, F.R. et al. Induced heterologous expression of the arginine deiminase pathway promotes growth advantages in the strict anaerobe Acetobacterium woodii. Appl Microbiol Biotechnol 104, 687–699 (2020). https://doi.org/10.1007/s00253-019-10248-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10248-9