Abstract
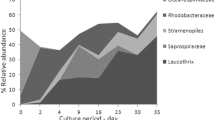
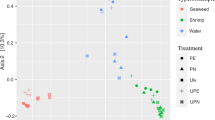
Biofloc technology is an efficient approach for intensive shrimp culture. However, the extent to which this process can influence the composition of intestinal microbial community is still unknown. Here, we surveyed the shrimp intestinal bacteria as well as the floc water from three biofloc systems with different stock densities. Our study revealed a similar variation trend in phylum taxonomy level between floc bacteria and gut microbiota. Microbial community varied notably in floc water from different stock densities, while a core genus with dominating relative abundance was detected in gut samples. Extensive variation was discovered in gut microbiota, but still clustered into groups according to stock density. Our results indicated that shrimp intestinal microbiota as well as bacteria aggregated in flocs assembled into distinct communities from different stock densities, and the intestinal communities were more similar with the surrounding environment as the increase of stock density and resulting high floc biomass. The high stock density changed the core gut microbiota by reducing the relative abundance of Paracoccus and increasing that of Nocardioides, which may negatively influence shrimp performance. Therefore, this study helps us to understand further bacteria and host interactions in biofloc system.





Similar content being viewed by others
Data availability
All data are fully available without restriction.
References
Arnold SJ, Sellars MJ, Crocos PJ, Coman GJ (2005) Response of juvenile brown tiger shrimp (Penaeus esculentus) to intensive culture conditions in a flow through tank system with three-dimensional artificial substrate. Aquaculture 246(1–4):231–238. https://doi.org/10.1016/j.aquaculture.2005.01.020
Avnimelech Y (1999) Carbon/nitrogen ratio as a control element in aquaculture systems. Aquaculture 176(3):227–235. https://doi.org/10.1016/S0044-8486(99)00085-X
Avnimelech Y (2009) Biofloc technology. A practical guide book. The World Aquaculture Society, Baton Rouge
Bianchi MAG, Bianchi AJM (1982) Statistical sampling of bacterial strains and its use in bacterial diversity measurement. Microb Ecol 8(1):61–69. https://doi.org/10.1007/bf02011462
Burford MA, Thompson PJ, McIntosh RP, Bauman RH, Pearson DC (2004) The contribution of flocculated material to shrimp (Litopenaeus vannamei) nutrition in a high-intensity, zero-exchange system. Aquaculture 232(1–4):525–537. https://doi.org/10.1016/s0044-8486(03)00541-6
Cardona E, Gueguen Y, Magre K, Lorgeoux B, Piquemal D, Pierrat F, Noguier F, Saulnier D (2016) Bacterial community characterization of water and intestine of the shrimp Litopenaeus stylirostris in a biofloc system. BMC Microbiol 16(1):157. https://doi.org/10.1186/s12866-016-0770-z
Chakravarthy SS, Pande S, Kapoor A, Nerurkar AS (2011) Comparison of denitrification between Paracoccus sp. and Diaphorobacter sp. Appl Biochem Biotechnol 165(1):260–269. https://doi.org/10.1007/s12010-011-9248-5
Chen W-M, Moulin L, Bontemps C, Vandamme P, Béna G, Boivin-Masson C (2003) Legume symbiotic nitrogen fixation by β -Proteobacteria is widespread in nature. J Bacteriol 185(24):7266–7272. https://doi.org/10.1128/jb.185.24.7266-7272.2003
Crab R, Lambert A, Defoirdt T, Bossier P, Verstraete W (2010) The application of bioflocs technology to protect brine shrimp (Artemia franciscana) from pathogenic Vibrio harveyi. J Appl Microbiol 109:1643–1649. https://doi.org/10.1111/j.1365-2672.2010.04791.x
Crab R, Defoirdt T, Bossier P, Verstraete W (2012) Biofloc technology in aquaculture: beneficial effects and future challenges. Aquaculture 356-357:351–356. https://doi.org/10.1016/j.aquaculture.2012.04.046
de Lorenzo MA, Candia EWS, Schleder DD, Rezende PC, Seiffert WQ, do Nascimento Vieira F (2016) Intensive hatchery performance of Pacific white shrimp in the biofloc system under three different fertilization levels. Aquac Eng 72-73:40–44. https://doi.org/10.1016/j.aquaeng.2016.04.001
Deng YL, Ruan YJ, Zhu SM, Guo XS, Han ZY, Ye ZY, Liu G, Shi MM (2017) The impact of DO and salinity on microbial community in poly(butylene succinate) denitrification reactors for recirculating aquaculture system wastewater treatment. AMB Express 7(1):113. https://doi.org/10.1186/s13568-017-0412-3
Dhariwal A, Chong J, Habib S, King IL, Agellon LB, Xia J (2017) MicrobiomeAnalyst: a web-based tool for comprehensive statistical, visual and meta-analysis of microbiome data. Nucleic Acids Res 45(W1):W180–W188. https://doi.org/10.1093/nar/gkx295
Ebeling JM, Timmons MB, Bisogni JJ (2006) Engineering analysis of the stoichiometry of photoautotrophic, autotrophic, and heterotrophic removal of ammonia–nitrogen in aquaculture systems. Aquaculture 257(1):346–358. https://doi.org/10.1016/j.aquaculture.2006.03.019
Emerenciano M, Cuzon G, Paredes A, Gaxiola G (2013) Evaluation of biofloc technology in pink shrimp Farfantepenaeus duorarum culture: growth performance, water quality, microorganisms profile and proximate analysis of biofloc. Aquac Int 21(6):1381–1394. https://doi.org/10.1007/s10499-013-9640-y
Farzanfar A (2006) The use of probiotics in shrimp aquaculture. FEMS Immunol Med Microbiol 48(2):149–158. https://doi.org/10.1111/j.1574-695X.2006.00116.x
Fernandez-Gomez B, Richter M, Schuler M, Pinhassi J, Acinas SG, Gonzalez JM, Pedros-Alio C (2013) Ecology of marine Bacteroidetes: a comparative genomics approach. ISME J 7(5):1026–1037. https://doi.org/10.1038/ismej.2012.169
Fóes GK, Fróes C, Krummenauer D, Poersch L, Wasielesky W (2011) Nursery of pink shrimp Farfantepenaeus paulensis in biofloc technology culture system: survival and growth at different stocking densities. J Shellfish Res 30(2):367–373. https://doi.org/10.2983/035.030.0224
Giatsis C, Sipkema D, Smidt H, Heilig H, Benvenuti G, Verreth J, Verdegem M (2015) The impact of rearing environment on the development of gut microbiota in tilapia larvae. Sci Rep 5:18206. https://doi.org/10.1038/srep18206
Giatsis C, Sipkema D, Smidt H, Verreth J, Verdegem M (2014) The colonization dynamics of the gut microbiota in tilapia larvae. PLoS One 9(7):e103641. https://doi.org/10.1371/journal.pone.0103641
Hargreaves JA (2006) Photosynthetic suspended-growth systems in aquaculture. Aquac Eng 34(3):344–363. https://doi.org/10.1016/j.aquaeng.2005.08.009
Haslun J, Correia E, Strychar K, Morris T, Samocha T (2012) Characterization of bioflocs in a no water exchange super-intensive system for the production of food size pacific white shrimp Litopenaeus vannamei. Int J Aquacult 2(6):29–38
Hou D, Huang Z, Zeng S, Liu J, Weng S, He J (2018) Comparative analysis of the bacterial community compositions of the shrimp intestine, surrounding water and sediment. J Appl Microbiol 125(3):792–799. https://doi.org/10.1111/jam.13919
Hu X, Du H, Ren C, Xu Y (2016) Illuminating anaerobic microbial community and cooccurrence patterns across a quality gradient in Chinese liquor fermentation pit muds. Appl Environ Microbiol 82(8):2506–2515. https://doi.org/10.1128/aem.03409-15
Johnson CN, Barnes S, Ogle J, Grimes DJ, Chang YJ, Peacock AD, Kline L (2008) Microbial community analysis of water, foregut, and hindgut during growth of pacific white shrimp, Litopenaeus vannamei, in closed-system aquaculture. J World Aquacult Soc 39(2):251–258
Kersters K, De Vos P, Gillis M, Swings J, Vandamme P, Stackebrandt E (2006) Introduction to the Proteobacteria. Springer, New York, pp 3–37
Kirchman DL (2002) The ecology of Cytophaga–Flavobacteria in aquatic environments. FEMS Microbiol Ecol 39(2):91–100. https://doi.org/10.1111/j.1574-6941.2002.tb00910.x
Krummenauer D, Samocha T, Poersch L, Lara G, Wasielesky W (2014) The reuse of water on the culture of Pacific white shrimp, Litopenaeus vannamei, in BFT system. J World Aquacult Soc 45(1):3–14. https://doi.org/10.1111/jwas.12093
Lee L, Zainal N, Azman A, Eng S, Goh B, Yin W, Ab Mutalib N, Chan K (2014) Diversity and antimicrobial activities of Actinobacteria isolated from tropical mangrove sediments in Malaysia. Sci World J 2014:14. https://doi.org/10.1155/2014/698178
Liu L, Hu Z, Dai X, Avnimelech Y (2014) Effects of addition of maize starch on the yield, water quality and formation of bioflocs in an integrated shrimp culture system. Aquaculture 418-419:79–86. https://doi.org/10.1016/j.aquaculture.2013.10.005
Luo G, Avnimelech Y, Pan Y-f, Tan H-x (2013) Inorganic nitrogen dynamics in sequencing batch reactors using biofloc technology to treat aquaculture sludge. Aquac Eng 52:73–79. https://doi.org/10.1016/j.aquaeng.2012.09.003
Ma B, Wang H, Dsouza M, Lou J, He Y, Dai Z, Brookes PC, Xu J, Gilbert JA (2016) Geographic patterns of co-occurrence network topological features for soil microbiota at continental scale in eastern China. ISME J 10(8):1891–1901. https://doi.org/10.1038/ismej.2015.261
Mello BL, Alessi AM, McQueen-Mason S, Bruce NC, Polikarpov I (2016) Nutrient availability shapes the microbial community structure in sugarcane bagasse compost-derived consortia. Sci Rep 6:38781. https://doi.org/10.1038/srep38781
Mitri S, Clarke E, Foster KR (2016) Resource limitation drives spatial organization in microbial groups. ISME J 10(6):1471–1482. https://doi.org/10.1038/ismej.2015.208
Moss SM, Moss DR, Arce SM, Lightner DV, Lotz JM (2012) The role of selective breeding and biosecurity in the prevention of disease in penaeid shrimp aquaculture. J Invertebr Pathol 110(2):247–250. https://doi.org/10.1016/j.jip.2012.01.013
Nayak SK (2010) Role of gastrointestinal microbiota in fish. Aquac Res 41(11):1553–1573. https://doi.org/10.1111/j.1365-2109.2010.02546.x
Nicholson JK, Holmes E, Kinross J, Burcelin R, Gibson G, Jia W, Pettersson S (2012) Host-gut microbiota metabolic interactions. Science 336(6086):1262–1267. https://doi.org/10.1126/science.1223813
Olafsen JA (2001) Interactions between fish larvae and bacteria in marine aquaculture. Aquaculture 200(1–2):223–247. https://doi.org/10.1016/S0044-8486(01)00702-5
Oliveros J (2007) An interactive tool for comparing lists with Venn’s diagrams. http://bioinfogp.cnb.csic.es/tools/venny/index.html. Accessed 12 November 2018
Prauser H (1976) Nocardioides, a new genus of the order Actinomycetales. Int J Syst Evol Microbiol 26(1):58–65
Rawls JF, Mahowald MA, Ley RE, Gordon JI (2006) Reciprocal gut microbiota transplants from zebrafish and mice to germ-free recipients reveal host habitat selection. Cell 127(2):423–433. https://doi.org/10.1016/j.cell.2006.08.043
Ray AJ, Dillon KS, Lotz JM (2011) Water quality dynamics and shrimp (Litopenaeus vannamei) production in intensive, mesohaline culture systems with two levels of biofloc management. Aquac Eng 45(3):127–136. https://doi.org/10.1016/j.aquaeng.2011.09.001
Ringø E, Zhou Z, Vecino JLG, Wadsworth S, Romero J, Krogdahl Å, Olsen RE, Dimitroglou A, Foey A, Davies S, Owen M, Lauzon HL, Martinsen LL, De Schryver P, Bossier P, Sperstad S, Merrifield DL (2016) Effect of dietary components on the gut microbiota of aquatic animals. A never-ending story? Aquac Nutr 22(2):219–282. https://doi.org/10.1111/anu.12346
Round JL, Mazmanian SK (2009) The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol 9(5):313
Samocha TM (2010) Use of intensive and super-intensive nursery systems. The shrimp book, theory and practice of Penaeid shrimp aquaculture. Nottingham, UK, pp 247–280
Schveitzer R, Arantes R, Costódio PFS, do Espírito Santo CM, Arana LV, Seiffert WQ, Andreatta ER (2013) Effect of different biofloc levels on microbial activity, water quality and performance of Litopenaeus vannamei in a tank system operated with no water exchange. Aquac Eng 56:59–70. https://doi.org/10.1016/j.aquaeng.2013.04.006
Stokstad E (2010) Down on the shrimp farm. Science 328(5985):1504
Tzeng T-D, Pao Y-Y, Chen P-C, Weng FC-H, Jean WD, Wang D (2015) Effects of post phylogeny and habitats on gut microbiomes of oriental river prawn (Macrobrachium nipponense). PLoS One 10(7):e0132860. https://doi.org/10.1371/journal.pone.0132860
Vadstein O, Bergh Ø, Gatesoupe F-J, Galindo-Villegas J, Mulero V, Picchietti S, Scapigliati G, Makridis P, Olsen Y, Dierckens K, Defoirdt T, Boon N, De Schryver P, Bossier P (2013) Microbiology and immunology of fish larvae. Rev Aquac 5:S1–S25. https://doi.org/10.1111/j.1753-5131.2012.01082.x
Viau VE, Souza DM, Rodríguez EM, Wasielesky W, Abreu PC, Ballester ELC (2013) Biofilm feeding by postlarvae of the pink shrimp Farfantepenaeus brasiliensis (Decapoda, Penaidae). Aquac Res 44(5):783–794. https://doi.org/10.1111/j.1365-2109.2011.03087.x
Vinatea L, Gálvez AO, Browdy CL, Stokes A, Venero J, Haveman J, Lewis BL, Lawson A, Shuler A, Leffler JW (2010) Photosynthesis, water respiration and growth performance of Litopenaeus vannamei in a super-intensive raceway culture with zero water exchange: interaction of water quality variables. Aquac Eng 42(1):17–24. https://doi.org/10.1016/j.aquaeng.2009.09.001
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73(16):5261–5267. https://doi.org/10.1128/aem.00062-07
Wasielesky W, Atwood H, Stokes A, Browdy CL (2006) Effect of natural production in a zero exchange suspended microbial floc based super-intensive culture system for white shrimp Litopenaeus vannamei. Aquaculture 258(1):396–403. https://doi.org/10.1016/j.aquaculture.2006.04.030
Xiong J, Wang K, Wu J, Qiuqian L, Yang K, Qian Y, Zhang D (2015) Changes in intestinal bacterial communities are closely associated with shrimp disease severity. Appl Microbiol Biotechnol 99(16):6911–6919. https://doi.org/10.1007/s00253-015-6632-z
Xu W-J, Morris TC, Samocha TM (2016) Effects of C/N ratio on biofloc development, water quality, and performance of Litopenaeus vannamei juveniles in a biofloc-based, high-density, zero-exchange, outdoor tank system. Aquaculture 453:169–175. https://doi.org/10.1016/j.aquaculture.2015.11.021
Xu W-J, Pan L-Q (2012) Effects of bioflocs on growth performance, digestive enzyme activity and body composition of juvenile Litopenaeus vannamei in zero-water exchange tanks manipulating C/N ratio in feed. Aquaculture 356-357:147–152. https://doi.org/10.1016/j.aquaculture.2012.05.022
Yang G, Tian X, Dong S, Peng M, Wang D (2015) Effects of dietary Bacillus cereus G19, B. cereus BC-01, and Paracoccus marcusii DB11 supplementation on the growth, immune response, and expression of immune-related genes in coelomocytes and intestine of the sea cucumber (Apostichopus japonicus Selenka). Fish Shellfish Immunol 45(2):800–807. https://doi.org/10.1016/j.fsi.2015.05.032
Zhao P, Huang J, Wang X-H, Song X-L, Yang C-H, Zhang X-G, Wang G-C (2012) The application of bioflocs technology in high-intensive, zero exchange farming systems of Marsupenaeus japonicus. Aquaculture 354-355:97–106. https://doi.org/10.1016/j.aquaculture.2012.03.034
Zhu SM, Deng YL, Ruan YJ, Guo XS, Shi MM, Shen JZ (2015) Biological denitrification using poly(butylene succinate) as carbon source and biofilm carrier for recirculating aquaculture system effluent treatment. Bioresour Technol 192. https://doi.org/10.1016/j.biortech.2015.06.021
Acknowledgements
We would like to thank the reviewers and editors for their insightful comments. We also thank Xiaomei Yue (Business Economics Group, Wageningen University) for helping with data analysis and visualization.
Funding
This study was funded by Natural Science Fund of China (31402348 and 31670377), Zhejiang Province Science and Technology Projects (2015C03010) and Program for Liaoning Excellent Talents in University (LR2015009). We also thank the reviewers for their insightful comments and suggestions.
Author information
Authors and Affiliations
Contributions
Conceptualization: YD and YR; data collection and analysis: YD, XX, XY, and HL; drafting the manuscript: YD and YR; review and editing: YD, XX, GC, JY, and YR. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This paper does not contain any studies with human participants or vertebrate animals performed by any of the authors.
Consent for publication
This manuscript does not contain any individual person’s data.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Deng, Y., Xu, X., Yin, X. et al. Effect of stock density on the microbial community in biofloc water and Pacific white shrimp (Litopenaeus vannamei) gut microbiota. Appl Microbiol Biotechnol 103, 4241–4252 (2019). https://doi.org/10.1007/s00253-019-09773-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-09773-4