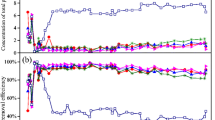
Abstract
Due to the complex nature of the wastewater (both domestic and non-domestic) composition, biological processes are widely used to remove nutrients, such as carbon (C), nitrogen (N), and phosphorous (P), which cause instability and hence contribute to the damage of water bodies. Systems with different configurations have been developed (including anaerobic, anoxic, and aerobic conditions) for the joint removal of carbon, nitrogen, and phosphorus. The goal of this research is to evaluate the extracellular activity of β-glucosidase and phosphatase enzymes in a University of Cape Town (UCT) system fed with two synthetic wastewaters of different molecular complexity. Both types of waters have medium strength characteristics similar to those of domestic wastewater with a mean C/N/P ratio of 100:13:1. The operation parameters were hydraulic retention time (HRT) of 10 h, solid retention time (SRT) of 12 days, mean concentration of the influent in terms of chemical oxygen demand (COD), total Kjeldahl nitrogen (TKN), and total phosphorus (TP) of 600, 80, and 6 mg/L, respectively. According to the results obtained, statistically significant differences have been found in the extracellular enzyme activities with the evaluated wastewaters and in the units comprising the treatment system in some of the cases. An analysis of principal components showed that the extracellular enzymatic activity has been correlated to nutrient concentration in wastewater, biomass concentration in the system, and metabolic conditions of treatment phases. Additionally, this research has allowed determining an inverse relationship between wastewater biodegradability and the extracellular enzyme activity of β-glucosidase and phosphatase. These results highlight the importance of including the analysis of biomass biochemical characteristics as control methods in wastewater treatment systems for the nutrient removal.





Similar content being viewed by others
References
APHA, AWWA, WEF (2012) Standard methods for the examination of water & wastewater, 22nd edn. American Public Health Association, American Water Works Association, Water Environment Federation, Washington
Arnosti C (2003) Microbial extracellular enzymes and their role in dissolved organic matter cycling. In: Findlay S, Sinsabaugh RL (eds) Aquatic ecosystems: interactivity of dissolved organic matter, vol 1. Aquatic ecology series. Academic Press, San Diego, pp 315–342
Bitton G (2011) Wastewater microbiology. Ecological and applied microbiology, 4th edn. Wiley, Hoboken
Chróst RJ (1991) Environmental control of the synthesis and activity of aquatic microbial ectoenzymes. In: Chróst RJ (ed) Microbial enzymes in aquiatic environments. Brock/Springer series in contemporary bioscience. Springer-Verlag New York Inc., New York, EUA, pp 29–59
Chróst RJ, Siuda W (2002) Ecology of microbial enzymes in lake ecosystems. In: Burns RG, Dick RP (eds) Enzymes in the environment. Activity, ecology and applications. Marcel Dekker, Inc., New York, pp 35–72
Chuang SH, Ouyang CF, Yuang HC, You SJ (1997) Effects of SRT and DO on nutrient removal in a combined as-biofilm process. Water Sci Technol 36:19–27
Crittenden JC, Trussell RR, Hand DW, Howe KJ, Tchobanoglous G (2012) MWH’s water treatment: principles and design, 3rd edn. Wiley, Hoboken
Cunha A, Almeida A, Coelho FJRC, Gomes NCM, Oliveira V, Santos AL (2010) Bacterial extracellular enzymatic activity in gobally changing aquatic ecosystems. In: Méndez-Vilas A (ed) Current research, technology and education topics in applied microbiology and microbial biotechnology, 2nd edn, vol 1. Formatex Research Center, Bajadoz, pp 124–135
Demarche P, Junghanns C, Nair RR, Agathos SN (2012) Harnessing the power of enzymes for environmental stewardship. Biotechnol Adv 30:933–953
Ekama GA (2015) Recent developments in biological nutrient removal. Water SA 41:515–524. doi:10.4314/wsa.v41i4.11
Ekama GA, Wentzel MC (2008) Nitrogen removal. In: Henze M, van Loosdrecht MCM, Ekama GA, Brdjanovic D (eds) Biological wastewater treatment: principles, modelling and design. IWA Publishing, London, pp 87–138
Friedler E, Butler D, Alfiya Y (2013) Wastewater composition. In: Larsen TA, Udert KM, Lienert J (eds) Source separation and decentralization for wastewater management. IWA Publishing, London, pp 241–257
Frølund B, Griebe T, Nielsen PH (1995) Enzymatic activity in the activated-sludge floc matrix. Appl Microbiol Biotechnol 43:755–761
Giraldo LC, Palacio CA, Aguirre NJ (2014) Temporal variation of the extracellular enzymatic activity (EEA): case of study: Aburrá-Medellín River, in the Valle de Aburrá in Medellín, Antioquia, Colombia. IJEP 4:58–67
Goel R, Mino T, Satoh H, Matsuo T (1998) Enzyme activities under anaerobic and aerobic conditions in activated sludge sequencing batch reactor. Water Res 32:2081–2088
Gómez-Silván C, Arevalo J, Perez J, Gonzalez-Lopez J, Rodelas B (2013) Linking hydrolytic activities to variables influencing a submerged membrane bioreactor (MBR) treating urban wastewater under real operating conditions. Water Res 47:66–78
Henry JG, Heinke GW, Burton I (2008) Environmental science and engineering, 2nd edn. Prentice Hall International, New Jersey
Henze M, Harremoës P, Arvin E, Janses JC (1997) Wastewater treatment: biological and chemical processes. Environmental engineering, 2nd. edn. Springer Berlin Heidelberg, New York
Hoffman M, Decho AW (1999) Extracellular enzymes within microbial biofilms and the role of the extracellular polymer matrix. In: Wingender J, Neu TR, Flemming HC (eds) Microbial extracelullar polymeric substances. Characterization, structure and function. Springer, Berlin, pp 217–230
Kirchman DL (2003) The contribution of monomers and other low-molecular weight compounds to the flux of dissolved organic material in aquatic ecosystems. In: Findlay S, Sinsabaugh RL (eds) Aquatic ecosystems: interactivity of dissolved organic matter, vol 1. Academic Press, San Diego, pp 218–241
Kreutz JA, Bockenhuser I, Wacht M, Fischer K (2016) A 1-year study of the activities of seven hydrolases in a communal wastewater treatment plant: trends and correlations. Appl Microbiol Biotechnol 100:6903–6915
Levine AD, Tchobanoglous G, Asano T (1991) Characterization of the size distribution of contaminants in wastewater: treatment and reuse implications. Water Res 25:911–922
Mara D (2004) Domestic wastewater treatment in developing countries. Earthscan, London
Marxsen J, Tippmann P, Heininger P, Preuss G, Remde A (1998) Enzymaktivität. In: Remde A, Tippmann P (eds) Mikrobiologische Charakterisierung aquatischer Sedimente: Methodensammlung. vol (Hrsg.) Vereinigung für Allgemeine und Angewandte Mikrobiologie (VAAM). Oldenbourg Verlag GmbH, München, pp 87–114
Monclús H, Sipma J, Ferrero G, Comas J, Rodriguez-Roda I (2010) Optimization of biological nutrient removal in a pilot plant UCT-MBR treating municipal wastewater during start-up. Desalination 250:592–597. doi:10.1016/j.desal.2009.09.030
Orhon D, Babuna FG, Karahan O (2009) Industrial wastewater treatment by activated sludge. IWA Publishing, London
R Development Core Team (2015) R: A language and environment for statistical computing, 3.2.3 edn. R foundation for statistical computing, Vienna
Rejmánková E, Sirová D (2007) Wetland macrophyte decomposition under different nutrient conditions: relationships between decomposition rate, enzyme activities and microbial biomass. Soil Biol Biochem 39:526–538. doi:10.1016/j.soilbio.2006.08.022
Saldarriaga JC, Garrido JM, Hoyos DA, Correa MA (2010) Remoción simultánea de carbono, nitrógeno y fósforo de aguas residuales en un sistema híbrido UCT modificado. DYNA 77:39–48
Seviour RJ (2009) An overview of the microbes in activated sludge. In: Seviour RJ, Nielsen PH (eds) Microbial ecology of activated sludge. IWA Publishing, London, pp 1–56
Smolders GJF, Van der Meij J, van Loosdrecht MCM, Heijnen JJ (1994) Stoichiometric model of the aerobic metabolism of the biological phosphorus removal process. Biotechnol Bioeng 44:837–848
Szilveszter S, Miklossy I, Meszaros A, Abraham B, Raduly B, Robescu DN, Lanyi S (2010) Enzyme activities and distribution in activated sludge in a lab-scale sequencing batch reactor (SBR). UPB Sci Bull 72:141–150
UN-Water (2015) Wastewater management: A UN-Water analytical brief. World Meteorological Organization, Geneve
Wentzel MC, Comeau Y, Ekama GA, Van Loosdrecht MCM, Brdjanovic D (2008) Enhanced biological phosphorus removal. In: Henze M, Van Loosdrecht MCM, Ekama GA, Brdjanovic D (eds) Biological wastewater treatment: principles, modelling and design. IWA Publishing, London, pp 156–220
Wetzel RG (2001) Limnology. Lake and river ecosystems. Academic Press, San Diego
Wilczeck S, Fischer H, Pusch MT (2005) Regulation and seasonal dynamics of extracelullar enzyme activities in the sediments of a large lowland river. Microb Ecol 50:253–267
Xie C, Lu R, Huang Y, Wang Q, Xu X (2010) Effects of ions and phosphates on alkaline phosphatase activity in aerobic activated sludge system. Bioresour Technol 101:3394–3399
Acknowledgements
This research was funded with resources provided by Grupo de Investigación en Ingeniería y Gestión Ambiental (GIGA) associated to the School of Engineering of Universidad de Antioquia. Furthermore, authors would like to thank to the Environmental Studies Laboratory of Universidad de Antioquia for the support provided to achieve this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Berrio-Restrepo, J.M., Saldarriaga, J.C., Correa, M.A. et al. Extracellular enzymatic activity of two hydrolases in wastewater treatment for biological nutrient removal. Appl Microbiol Biotechnol 101, 7385–7396 (2017). https://doi.org/10.1007/s00253-017-8423-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8423-1