Abstract
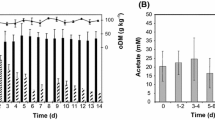
Biochemical processes in biogas plants are still not fully understood. Especially, the identification of possible bottlenecks in the complex fermentation processes during biogas production might provide potential to increase the performance of biogas plants. To shed light on the question which group of organism constitutes the limiting factor in the anaerobic breakdown of organic material, biogas sludge from different mesophilic biogas plants was examined under various conditions. Therefore, biogas sludge was incubated and analyzed in anaerobic serum flasks under an atmosphere of N2/CO2. The batch reactors mirrored the conditions and the performance of the full-scale biogas plants and were suitable test systems for a period of 24 h. Methane production rates were compared after supplementation with substrates for syntrophic bacteria, such as butyrate, propionate, or ethanol, as well as with acetate and H2+CO2 as substrates for methanogenic archaea. Methane formation rates increased significantly by 35 to 126 % when sludge from different biogas plants was supplemented with acetate or ethanol. The stability of important process parameters such as concentration of volatile fatty acids and pH indicate that ethanol and acetate increase biogas formation without affecting normally occurring fermentation processes. In contrast to ethanol or acetate, other fermentation products such as propionate, butyrate, or H2 did not result in increased methane formation rates. These results provide evidence that aceticlastic methanogenesis and ethanol-oxidizing syntrophic bacteria are not the limiting factor during biogas formation, respectively, and that biogas plant optimization is possible with special focus on methanogenesis from acetate.





Similar content being viewed by others
References
Agler MT, Spirito CM, Usack JG, Werner JJ, Angenent LT (2014) Development of a highly specific and productive process for n-caproic acid production: applying lessons from methanogenic microbiomes. Water Sci Technol 69:62–68
Ahring BK, Westermann P (1988) Product inhibition of butyrate metabolism by acetate and hydrogen in a thermophilic coculture. Appl Environ Microbiol 54:2393–2397
Barker HA (1940) Studies upon the methane fermentation. IV. The isolation and culture of Methanobacterium omelianskii. Antonie Leeuwenhoek 6:201–220
Bryant MP, Wolin EA, Wolin MJ, Wolfe RS (1967) Methanobacillus omelianskii, a symbiotic association of two species of bacteria. Arch Microbiol 59:20–31
Carrez PMC (1908) Le ferrocyanure de potassium et l'acétate de zinc comme agents de défécation des urines. Annales de chimie analytique 13:97–101
Chen Y, Cheng JJ, Creamer KS (2008) Inhibition of anaerobic digestion process: a review. Bioresour Technol 99:4044–4064
Fachverband Biogas e.V. (2012) Branchenzahlen 2011 und Prognose der Branchenentwicklung 2012/2013. http://www.biogas.org/edcom/webfvb.nsf/id/DE_PM-29-12/$file/12-11-16_Biogas%20Branchenzahlen%202011-2012-2013.pdf
Guo WQ, Ren NQ, Wang XJ, Xiang WS, Meng ZH, Ding J, Qu YY, Zhang LS (2008) Biohydrogen production from ethanol-type fermentation of molasses in an expanded granular sludge bed (EGSB) reactor. Int J Hydrog Energy 33:4981–4988
Jain SR, Mattiasson B (1998) Acclimatization of methanogenic consortia for low pH biomethanation process. Biotechnol Lett 20:771–775
Lettinga G, Van Velson AFM, Hobma SW, De Zeeuw W, Klapwijk A (1980) Use of the upflow sludge blanket (USB) reactor for biological wastewater treatment, especially for anaerobic treatment. Biotechnol Bioeng 22:699–724
Lv Z, Leite AF, Harms H, Richnow HH, Liebetrau J, Nikolausz M (2013) Influences of the substrate feeding regime on methanogenic activity in biogas reactors approached by molecular and stable isotope methods. Anaerobe. http://www.sciencedirect.com/science/article/pii/S107599641300190X?via=ihub
Maurya MS, Singh L, Sairam M, Alam SI (1994) Production of biogas from night soil: effect of temperature and volatile solids. Indian J Microbiol 34:223–228
McEniry J, Allen E, Murphy JD, O’Kiely P (2014) Grass for biogas production: the impact of silage fermentation characteristics on methane yield in two contrasting biomethane potential test systems. Renew Energy 63:524–530
McInerney MJ, Sieber JR, Gunsalus RP (2009) Syntrophy in anaerobic global carbon cycles. Curr Opin Biotechnol 20:623–632
Moestedt J, Påledal SN, Anna Schnürer A, Nordell E (2013) Biogas production from thin stillage on an industrial scale—experience and optimisation. Energies 6:5642–5655
Müller N, Worm P, Schink B, Stams AJM, Plugge CM (2010) Syntrophic butyrate and propionate oxidation processes: from genomes to reaction mechanisms. Environ Microbiol Rep 2:489–499
Nielsen HB, Uellendahl H, Ahring BK (2007) Regulation and optimization of the biogas process: propionate as a key parameter. Biomass Bioenergy 31:820–830
Nordmann W (1977) Die Überwachung der Schlammfaulung. Korrespondenz Abwasser 3
Nyns EJ (1986) Biomethanation processes. In: Rehm HJ, Reeds G (eds) Biotechnology, vol 8. VCH press, Weinheim, pp 207–268
Pindt PF, Angelidaki I, Ahring BK (2003) Dynamics of the anaerobic process: effects of volatile fatty acids. Biotechnol Bioeng 82:791–801
Rasi S, Veijanen A, Rintala J (2007) Trace compounds of biogas from different biogas production plants. Energy 32:1375–1380
Sanders FA, Bloodgood DE (1965) The effect of nitrogen to carbon ratios on anaerobic decomposition. J Water Pollut Control Fed 37:1741
Schink B (1997) Energetics of syntrophic cooperation in methanogenic degradation. Microbiol Mol Biol Rev 61:262–280
Scholten JCM, Conrad R (2000) Energetics of syntrophic propionate oxidation in defined batch and chemostat cocultures. Appl Environ Microbiol 66:2934–2942
Seitz HJ, Schink B, Conrad R (1988) Thermodynamics of hydrogen metabolism in methanogenic cocultures degrading ethanol or lactate. FEMS Microbiol Lett 55:119–124
Sharma SK, Mishra IM, Sharma MP, Saini JS (1988) Effect of particle size on biogas generation from biomass residues. Biomass 17:251–263
Sieber JR, McInerney MJ, Gunsalus RP (2012) Genomic insights into syntrophy: the paradigm for anaerobic metabolic cooperation. Annu Rev Microbiol 66:429–452
Stams AJM (1994) Metabolic interactions between anaerobic bacteria in methanogenic environments. Antonie Leeuwenhoek 66:271–294
Stams AJM, Plugge CM (2009) Electron transfer in syntrophic communities of anaerobic bacteria and archaea. Nat Rev Microbiol 7:568–577
Takizawa N, Umetsu K, Takahata H, Hoshiba H (1994) Temperature effects on continuously expending anaerobic digester with dairy manure slurry. Res Bull Obihiro Univ Nat Sci 19:31–36
Umetsu K, Takahata H, Kawamoto T (1992) Effect of temperature on mesophilic anaerobic digestion of dairy cow slurry. Res Bull Obihiro Univ 17:401–408
Wilkie A, Colleran E (1986) Pilot scale digestion of pig slurry supernatant using an upflow anaerobic filter. Environ Lett 7:65–76
Acknowledgments
We thank Joachim Clemens, Nadine Hörter, Stefanie Peters, Thomas Dickhaus, and Thomas Fülling from the companies Bioreact GmbH and Bonalytic GmbH, for their technical support and analysis of physicochemical parameters. We would also like to thank Elisabeth Schwab, Stefanie Berger, and Sebastian van Helmont for their technical assistance. This work was supported by funding from Bundesministerium für Bildung und Forschung (BMBF, project no. 03SF0421A).
Author information
Authors and Affiliations
Corresponding author
Additional information
Sarah Refai and Kati Wassmann contributed equally to this work.
Rights and permissions
About this article
Cite this article
Refai, S., Wassmann, K. & Deppenmeier, U. Short-term effect of acetate and ethanol on methane formation in biogas sludge. Appl Microbiol Biotechnol 98, 7271–7280 (2014). https://doi.org/10.1007/s00253-014-5820-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-5820-6