Abstract
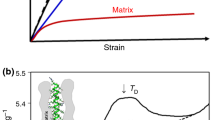
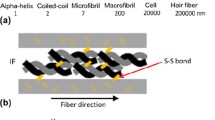
Previous work with Atomic Force Microscope (AFM) nanoindentation, on longitudinal and cross-sections of the human hair fibre, allowed for the derivation of a model for the mechanical behaviour of human hair, called the Anisotropic Index. Expanding that research further, and by applying this model, the nanomechanical behaviour of hairs from patients with the disease Trichothiodystrophy (TTD) has been examined and structural insights, gained from combining the AFM results with Differential Scanning Calorimetry (DSC) experiments and tensile measurements, suggests that TTD-affected hairs have a relatively increased amount of Keratin Intermediate Filaments, contained in compartments of differing crosslinking extent. The associated calculations of axial and transverse Young’s Moduli deliver values in good agreement with the measured fibre mechanics. Furthermore, comparing these findings with the results previously obtained from the study of hairs from patients with the disease Monilethrix, it is shown that the Anisotropic Index correlates well with the known deficiencies in both hair types obtained from such patients and allows for discerning between the Control hair and from those affected by the two diseases. AFM nanoindentation along and across the fibre axis and the Anisotropic Index thus appear to reveal structural details of hair not otherwise acquirable, whilst DSC may offer a quick and simple method for distinguishing between different severities of TTD.








Similar content being viewed by others
Data availability
All the data are given in the article.
References
Akkermans RLC, Warren PB (2004) Multiscale modelling of human hair. Phil Trans R Soc Lond A 362:1783–1793
Alibardi L (2006) Structural and immunocytochemical characterization of keratinization in vertebrate epidermis and epidermal derivatives. Int Rev Cytol 253:177–259. https://doi.org/10.1016/S0074-7696(06)53005-0
Annaidh AN, Bruyère K, Destrade M, Gilchrist MD, Otténio M (2012) Characterization of the anisotropic mechanical properties of excised human skin. J Mech Behav Biomed Mater 5:139–148. https://doi.org/10.1016/j.jmbbm.2011.08.016
Botta E, Nardo T, Lehmann AR, Egly J-M, Pedrini AM, Stefanini M (2002) Reduced level of the repair/transcription factor TFIIH in trichothiodystrophy. Hum Molec Genet 11:2919–2928. https://doi.org/10.1093/hmg/11.23.2919
Breakspear S, Noecker B, Popescu C (2018) Hair mechanical anisotropy—what does it tell us? J Cosmet Sci 69:305–313
Breakspear S, Noecker B, Popescu C (2019) Relevance and evaluation of hydrogen and disulfide bond contribution to the mechanics of hard α-keratin fibers. J Phys Chem B 123:4505–4511. https://doi.org/10.1021/acs.jpcb.9b01690
Breakspear S, Ivanov D, Noecker B, Popescu C (2021) Nanomechanical properties of Monilethrix affected hair are independent of phenotype. J Str Biol 213:107679. https://doi.org/10.1016/j.jsb.2020.107679
Chang H, Huang Z, Wen S, Chen J, Liu X, Pan M, Zhao Y (2016) The influence of crystal defects on the elastic properties of tungsten metals. Fusion Eng Des 109:321–325. https://doi.org/10.1016/j.fusengdes.2016.03.003
Cheng S, Stone J, De Berker D (2009) Trichothiodystrophy and fragile hair: the distinction between diagnostic signs and diagnostic labels in childhood hair disease. Br J Dermatol 161(6):1370–1383. https://doi.org/10.1111/j.1365-2133.2009.09403.x
Clifford CA, Seah MP (2005) Quantification issues in the identification of nanoscale regions of homopolymers using modulus measurement via AFM nanoindentation. Appl Surf Sci 252:1915–1933. https://doi.org/10.1016/j.apsusc.2005.08.090
Crossley JAA, Gibson CT, Mapledoram LD, Huson MG, Myhra S, Pham DK, Sofield CJ, Turner PS, Watson GS (2000) Atomic force microscopy analysis of wool fibre surfaces in air and under water. Micron 31:659–667. https://doi.org/10.1016/S0968-4328(99)00076-1
Danilatos G, Feughelman M (1980) The microfibril-matrix relationships in the mechanical properties of keratin fibers. Part II: the mechanical properties of the matrix during the extension of an alpha-keratin fiber. Text Res J 50:568–574. https://doi.org/10.1177/004051758005000908
Danciulescu C (2004) Investigations of the relative conformational stability of protein mutants by molecular dynamics simulation. Dissertation, RWTH Aachen
Faghri S, Tamura D, Kraemer KH, DiGiovanna JJ (2008) Trichothiodystrophy: a systematic review of 112 published cases characterises a wide spectrum of clinical manifestations. J Med Genet 45(10):609–621. https://doi.org/10.1136/jmg.2008.058743
Ferrando J, Mir-Bonafé JM, Cepeda-Valdés R, Domínguez A, Ocampo-Candiani J, García-Veigas J, Gómez-Flores M, Salas-Alanis JC (2012) Further insights in Trichothiodystrophy: A clinical, microscopic, and ultrastructural study of 20 cases and literature review. Int J Trichology 4(3):158–163. https://doi.org/10.4103/0974-7753.100075
Feughelman M (1982) The physical properties of alpha-keratin fibres. J Soc Cosmet Chem 33:385–406
Feughelman M (1989) A note on the water-impenetrable component of α-keratin fibres. Text Res J 59:739–742. https://doi.org/10.1177/004051758905901205
Feughelman M (2002) Natural protein fibers. J Appl Polym Sci 83:489–507. https://doi.org/10.1002/app.2255
Feughelman M, Robinson MS (1967) The tensile behavior of wool fibers in liquid nitrogen. Text Res J 37:705–706. https://doi.org/10.1177/004051756703700812
Filippov A, Gorb SN (2013) Frictional-anisotropy-based systems in biology: structural diversity and numerical model. Sci Rep 3:1240. https://doi.org/10.1038/srep01240
Fraser RDB, Parry DAD (2017) Intermediate filament structure in fully differentiated (oxidised) trichocyte keratin. J Str Biol 200:45–53. https://doi.org/10.1016/j.jsb.2017.09.003
Fudge DS, Gosline JM (2004) Molecular design of the alpha-keratin composite: insights from a matrix-free model, hagfish slime threads. Proc R Soc Lond B 271:291–299. https://doi.org/10.1098/rspb.2003.2591
Gillespie JM, Marshall RC (1983) A comparison of the proteins of normal and trichothiodystrophic human hair. J Invest Dermatol 80:195–202. https://doi.org/10.1111/1523-1747.ep12534032
Gummer CL, Dawber RPR, Price VH (1984) Trichothiodystrophy: an electron-histochemical study of the hair shaft. Br J Dermatol 110:439–449. https://doi.org/10.1111/j.1365-2133.1984.tb04659.x
Hearle JWS (2000) A critical review of the structural mechanics of wool and hair fibres. Int J Biol Macromol 27:123–138. https://doi.org/10.1016/s0141-8130(00)00116-1
Hearle JWS (2007) Protein fibers: structural mechanics and future opportunities. J Mater Sci 42:8010–8019
Hertz H (1881) Ueber die berührung fester elastischer Koerper. J Reine Angew Math 92:156–171
Istrate D, Popescu C, Er Rafik M, Möller M (2013) The effect of pH on the thermal stability of fibrous hard alpha-keratins. Polym Degrad Stab 98:542–549. https://doi.org/10.1016/j.polymdegradstab.2012.12.001
Itin PH, Sarasin A, Pittelkow MR (2001) Trichothiodystrophy: Update on the sulfur deficient brittle hair syndromes. J Am Acad Dermatol 44:891–923. https://doi.org/10.1067/mjd.2001.114294
Johnson KL (1985) Contact mechanics. Cambridge University Press, Cambridge, UK
Kadir M, Wang X, Zhu B, Liu J, Harland D, Popescu C (2017) The structure of the ‘‘amorphous” matrix of keratins. J Str Biol 198:116–123. https://doi.org/10.1016/j.jsb.2017.04.001
Katz JL (1980) Anisotropy of Young’s modulus of bone. Nature 283:106–107. https://doi.org/10.1038/283106a0
Korge BP, Healy E, Munro CS, Pünter C, Birch-Machin M, Holmes SC, Darlington S, Hamm H, Messenger AG, Rees JL, Traupe H (1998) A mutational hotspot in the 2B domain of human hair basic keratin 6 (hHb6) in monilethrix patients. J Invest Dermatol 111:896–899. https://doi.org/10.1046/j.1523-1747.1998.00362.x
Liang C, Kraemer KH, Morris A, Schiffmann R, Price VH, Menefee E, DiGiovanna JJ (2005) Characterization of tiger tail banding and hair shaft abnormalities in trichothiodystrophy. J Am Acad Dermatol 52:224–232. https://doi.org/10.1016/j.jaad.2004.09.013
Liu H, Bryson WG (2002) A three-component model of the wool fibre - effects of morphology, elasticity and intermediate filament arrangement on fibre stiffness. J Text Inst 93:121–131. https://doi.org/10.1080/00405000208630557
Mamada A, Ishihama M, Fukuda R, Inoue S (2008) Changes in hair properties by Eucalyptus extract. J Cosmet Sci 59:481–496
Nickell S, Hermann M, Essenpreis M, Farrell TJ, Krämer U, Patterson MS (2000) Anisotropy of light propagation in human skin. Phys Med Biol 45:2873. https://doi.org/10.1088/0031-9155/45/10/310
Osada M, Gniadecka M, Brandrup F, Kjærsgård Hansen L, Wulf HC (2003) Raman spectroscopy analysis of protein structure of hair in patients with trichothiodystrophy. Br J Dermatol 148:600–601. https://doi.org/10.1046/j.1365-2133.2003.05209_8.x
Popescu C, Gummer C (2016) DSC of human hair: a tool for claim support or incorrect data analysis? Int J Cosm Sci 38:433–439. https://doi.org/10.1111/ics.12306
Popescu C, Hoecker H (2007) Hair—the most sophisticated biological composite material. Chem Soc Rev 36:1282–1291. https://doi.org/10.1039/b604537p
Price VH, Odom RB, Ward WG, Jones FT (1980) Trichothiodystrophy: sulphur-deficient brittle hair as a marker for a neuroectodermal symptom complex. Arch Dermatol 116:1375–1384. https://doi.org/10.1001/archderm.1980.01640360049017
Reuss A (1929) Berechnung der fließgrenze von Mischkristallen auf Grund der Plastizitätsbedingung für Einkristalle. ZAMM-Z Angew Math Me 9:49–58
Rice RH, Wong VJ, Price VH, Hohl D, Pinkerton KE (1996) Cuticle cell defects in lamellar ichthyosis hair and anomalous hair shaft syndromes visualized after detergent extraction. Anat Rec 246:433–440. https://doi.org/10.1002/(SICI)1097-0185(199612)246:4%3c433::AID-AR2%3e3.0.CO;2-W
Robbins CR (2012) Chemical and physical behavior of human hair, 5th edn. Springer Verlag, Berlin Heidelberg
Rockland LB (1960) Saturated salt solutions for static control of relative humidity between 5° and 40 °C. Anal Chem 32:1375–1376
Rogers M (1995) Hair shaft abnormalities: Part I. Australas J Dermatol 36:179–184. https://doi.org/10.1111/j.1440-0960.1995.tb00969.x
Rouse JG, Van Dyke ME (2010) A review of keratin-based biomaterials for biomedical applications. Materials 3:999–1014. https://doi.org/10.3390/ma3020999
Sass JO, Skladal D, Zelger B, Romani N, Utermann B (2004) Trichothiodystrophy: quantification of cysteine in human hair and nails by application of sodium azide-dependent oxidation to cysteic acid. Arch Dermatol Res 296:188–191. https://doi.org/10.1007/s00403-004-0483-2
Seshadri IP, Bhushan B (2008) In situ tensile deformation characterization of human hair with Atomic Force Microscopy. Acta Mater 56:774–781. https://doi.org/10.1016/j.actamat.2007.10.033
Smith JR (1997) Use of atomic force microscopy for high-resolution non-invasive structural studies of human hair. J Soc Cosmet Chem 48:199–208
Smith JR, Swift JA (2005) Maple syrup urine disease hair reveals the importance of 18-methyleicosanoic acid in cuticular delamination. Micron 36(3):261–266
Smith JR, Breakspear S, Fletcher RJR, Campbell SA (2005) AFM in surface finishing: part IV Force–distance curves. Trans Inst Met Fin 83(2):63–67. https://doi.org/10.1179/002029605X38979
Sneddon IN (1965) The relation between load and penetration in the axisymmetric Boussinesq problem for a punch of arbitrary profile. Int J Eng Sci 3:47–57. https://doi.org/10.1016/0020-7225(65)90019-4
Van Neste D (1991) Trichothiodystrophy: recent advances. Eur J Pediat Dermatol 1:45–50
Voigt W (1889) Ueber die Beziehung zwischen den beiden Elasticitätsconstanten isotroper Körper. Ann Phys-Berlin 274:573–587
Wang L, Song J, Ortiz C, Boyce MC (2009) Anisotropic design of a multilayered biological exoskeleton. J Mater Res 24:3477–3494. https://doi.org/10.1557/jmr.2009.0443
Wang B, Yang W, McKittrick J, Meyers MA (2016) Keratin: Structure, mechanical properties, occurrence in biological organisms, and efforts at bioinspiration. Prog Mater Sci 76:229–318. https://doi.org/10.1016/j.pmatsci.2015.06.001
Watt C, D'Arcy RL (1979) 26—Water-vapour adsorption Isotherms of wool. J Text Inst 70(7):298–307. https://doi.org/10.1080/00405007908658854
Whiting DA, Dy LC (2006) Office diagnosis of hair shaft defects. Semin Cutan Med Surg 25(1):24–34. https://doi.org/10.1016/j.sder.2006.01.004
Winter H, Rogers MA, Gebhardt M, Wollina U, Boxall L, Chitayat D, Babul-Hirji R, Stevens HP, Zlotogorski A, Schweizer J (1997) A new mutation in the type II hair cortex keratin hHb1 involved in the inherited hair disorder monilethrix. Hum Genet 101:165–169. https://doi.org/10.1007/s004390050607
Winter H, Clark RD, Taras-Wahlberg T, Rogers MA, Schweizer J (1999) Monilethrix: a novel mutation (Glu402Lys) in the helix termination motif and the first causative mutation (Asn114Asp) in the helix initiation motif of the type II hair keratin hHb6. J Invest Dermatol 113:263–266. https://doi.org/10.1046/j.1523-1747.1999.00685.x
Acknowledgements
The authors wish to thank Dr. Ingrid Hausser of the Institute of Pathology, University of Heidelberg, Germany, for providing the Trichothiodystrophy-affected hair samples used in this study and to Dr. Lutz Langbein, formerly of German Cancer Research Center (DKFZ) Heidelberg, Germany, for fruitful discussions and advice.
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Contributions
SB conceptualization, methodology, data curation, formal analysis, investigation, and writing. BN conceptualization, methodology, data curation, formal analysis, investigation, and writing. CP conceptualization, methodology, formal analysis, investigation, and writing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
Research does not involve Human Participants and/or Animals. The samples used for this work are hair fibres collected from hair-cut salon.
Informed consent
The authors do not deem any need of informed consent as there are no identifiable personal data/materials used in this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Breakspear, S., Noecker, B. & Popescu, C. Mechanical anisotropy of hair affected by genetic diseases highlights structural information related to differential crosslinking in keratins. Eur Biophys J 52, 53–67 (2023). https://doi.org/10.1007/s00249-023-01635-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-023-01635-2