Abstract
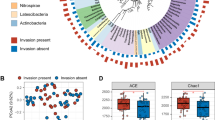
Dormancy is thought to promote biodiversity within microbial communities, but how assembly of the active community responds to changes in environmental conditions is unclear. To measure the active and dormant communities of bacteria and fungi colonizing decomposing litter in maple forests, we targeted ribosomal genes and transcripts across a natural environmental gradient. Within bacterial and fungal communities, the active and dormant communities were phylogenetically distinct, but patterns of phylogenetic clustering varied. For bacteria, active communities were significantly more clustered than dormant communities, while the reverse was found for fungi. The proportion of operational taxonomic units (OTUs) classified as active and the degree of phylogenetic clustering of the active bacterial communities declined with increasing pH and decreasing C/N. No significant correlations were found for the fungal community. The opposing pattern of phylogenetic clustering in dormant and active communities and the differential response of active communities to environmental gradients suggest that dormancy differentially structures bacterial and fungal communities.





Similar content being viewed by others
References
Segev E, Smith Y, Ben-Yehuda S (2012) RNA dynamics in aging bacterial spores. Cell 148:139–149. doi:10.1016/j.cell.2011.11.059
Jones SE, Lennon JT (2010) Dormancy contributes to the maintenance of microbial diversity. Proc Natl Acad Sci 107:5881–5886. doi:10.1073/pnas.0912765107
Lennon JT, Lennon JT, Jones SE, Jones SE (2011) Microbial seed banks: the ecological and evolutionary implications of dormancy. Nat Rev Microbiol 9:119–130. doi:10.1038/nrmicro2504
Chesson P (2000) Mechanisms of maintenance of species diversity. Annu Rev Ecol Syst 343–366
Burns JH, Strauss SY (2011) More closely related species are more ecologically similar in an experimental test. Proc Natl Acad Sci 108:5302–5307. doi:10.1073/pnas.1013003108
Martiny AC, Treseder K, Pusch G (2013) Phylogenetic conservatism of functional traits in microorganisms. ISME J 7:830–838. doi:10.1038/ismej.2012.160
Amend AS, Martiny AC, Allison SD et al (2015) Microbial response to simulated global change is phylogenetically conserved and linked with functional potential. The ISME J 1–10. doi: 10.1038/ismej.2015.96
Webb CO (2000) Exploring the phylogenetic structure of ecological communities: an example for rain forest trees. Am Nat 156:145–155. doi:10.1086/303378
Litchman E, Edwards KF, Klausmeier CA (2015) Microbial resource utilization traits and trade-offs: implications for community structure, functioning, and biogeochemical impacts at present and in the future. Front Microbiol 6:254. doi:10.3389/fmicb.2015.00254
Bertness MD, Callaway R (1994) Positive interactions in communities. Trends Ecol Evol 9:191–193. doi:10.1016/0169-5347(94)90088-4
Placella SA, Brodie EL, Firestone MK (2012) Rainfall-induced carbon dioxide pulses result from sequential resuscitation of phylogenetically clustered microbial groups. Proc Natl Acad Sci 109:10931–10936. doi:10.1073/pnas.1204306109
Angel R, Conrad R (2013) Elucidating the microbial resuscitation cascade in biological soil crusts following a simulated rain event. Environ Microbiol 15:2799–2815. doi:10.1111/1462-2920.12140
Shade A, Peter H, Allison SD et al (2012) Fundamentals of microbial community resistance and resilience. Front Microbiol 3:417. doi:10.3389/fmicb.2012.00417
Aanderud ZT, Jones SE, Fierer N, Lennon JT (2015) Resuscitation of the rare biosphere contributes to pulses of ecosystem activity. Front Microbiol 6:24. doi:10.3389/fmicb.2015.00024
van der Heijden MGA, Bardgett RD, van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310. doi:10.1111/j.1461-0248.2007.01139.x
Pregitzer KS, Burton AJ, Zak DR, Talhelm AF (2008) Simulated chronic nitrogen deposition increases carbon storage in Northern Temperate forests. Glob Chang Biol 14:142–153. doi:10.1111/j.1365-2486.2007.01465.x
Eisenlord SD, Freedman Z, Zak DR et al (2013) Microbial mechanisms mediating increased soil C storage under elevated atmospheric N deposition. Appl Environ Microbiol 79:1191–1199. doi:10.1128/AEM.03156-12
Hesse CN, Mueller RC, Vuyisich M et al (2015) Forest floor community metatranscriptomes identify fungal and bacterial responses to N deposition in two maple forests. Front Microbiol 6:337. doi:10.3389/fmicb.2015.00337
Claesson MJ, Wang Q, O'Sullivan O et al (2010) Comparison of two next-generation sequencing technologies for resolving highly complex microbiota composition using tandem variable 16S rRNA gene regions. Nucleic Acids Res 38:e200–e200. doi:10.1093/nar/gkq873
Schloss PD, Westcott SL, Ryabin T et al (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541. doi:10.1128/AEM.01541-09
Caporaso JG, Kuczynski J, Stombaugh J et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. doi:10.1038/nmeth.f.303
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461. doi:10.1093/bioinformatics/btq461
Cole JR, Wang Q, Cardenas E et al (2009) The Ribosomal Database Project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res 37:D141–D145. doi:10.1093/nar/gkn879
Liu KL, Liu KL, Porras-Alfaro A et al (2012) Accurate, rapid taxonomic classification of fungal large-subunit rRNA genes. Appl Environ Microbiol 78:1523–1533. doi:10.1128/AEM.06826-11
Meyer F, Paarmann D, D'Souza M et al (2008) The metagenomics RAST server—a public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinf 9:386. doi:10.1186/1471-2105-9-386
Kembel SW, Wu M, Eisen JA, Green JL (2012) Incorporating 16S gene copy number information improves estimates of microbial diversity and abundance. PLoS Comput Biol 8:e1002743. doi:10.1371/journal.pcbi.1002743
Celio GJ, Padamsee M, Dentinger BTM et al (2007) Assembling the Fungal Tree of Life: constructing the Structural and Biochemical Database. Mycologia 98:850–859. doi:10.3852/mycologia.98.6.850
Matsen FA, Kodner RB, Armbrust EV (2010) pplacer: linear time maximum-likelihood and Bayesian phylogenetic placement of sequences onto a fixed reference tree. BMC Bioinf 11:538. doi:10.1186/1471-2105-11-538
Letunic I, Bork P (2011) Interactive Tree Of Life v2: online annotation and display of phylogenetic trees made easy. Nucleic Acids Res 39:W475–W478. doi:10.1093/nar/gkr201
Kembel SW, Cowan PD, Helmus MR et al (2010) Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26:1463–1464. doi:10.1093/bioinformatics/btq166
Oksanen J, Blanchet FG, Kindt R et al (2013) Vegan: community ecology package. Community Ecol
Lozupone C, Knight R (2005) UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71:8228–8235. doi:10.1128/AEM.71.12.8228-8235.2005
McMurdie PJ, Holmes S (2013) Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8:e61217. doi:10.1371/journal.pone.0061217
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Evans SE, Wallenstein MD (2013) Climate change alters ecological strategies of soil bacteria. Ecol Lett 17:155–164. doi:10.1111/ele.12206
Kembel SW (2009) Disentangling niche and neutral influences on community assembly: assessing the performance of community phylogenetic structure tests. Ecol Lett 12:949–960. doi:10.1111/j.1461-0248.2009.01354.x
Gotelli NJ (2000) Null model analysis of species co-occurrence patterns. Ecology 81:2606–2621. doi:10.1890/0012-9658(2000)081[2606:NMAOSC]2.0.CO;2
Rousk J, Bååth E, Brookes PC et al (2010) Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J 4:1340–1351. doi:10.1038/ismej.2010.58
Strickland MS, Rousk J (2010) Soil biology & biochemistry. Soil Biol Biochem 42:1385–1395. doi:10.1016/j.soilbio.2010.05.007
Goslee SC, Urban DL (2007) The ecodist package for dissimilarity-based analysis of ecological data. J Stat Softw 22:1–19
Damon C, Lehembre F, Oger-Desfeux C et al (2012) Metatranscriptomics reveals the diversity of genes expressed by eukaryotes in forest soils. PLoS ONE 7:e28967. doi:10.1371/journal.pone.0028967.t004
DeAngelis KM, Firestone MK (2012) Phylogenetic clustering of soil microbial communities by 16S rRNA but not 16S rRNA genes. Appl Environ Microbiol 78:2459–2461. doi:10.1128/AEM.07547-11
Baldrian P, Kolařík M, Štursová M et al (2012) Active and total microbial communities in forest soil are largely different and highly stratified during decomposition. ISME J 6:248–258. doi:10.1038/ismej.2011.95
Webb CO, Ackerly DD, McPeek MA, Donoghue MJ (2002) Phylogenies and community ecology. Annu Rev Ecol Syst 475–505
Cavender-Bares J, Kozak KH, Fine PVA, Kembel SW (2009) The merging of community ecology and phylogenetic biology. Ecol Lett 12:693–715. doi:10.1111/j.1461-0248.2009.01314.x
Mayfield MM, Mayfield MM, Levine JM, Levine JM (2010) Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecol Letter 13:1085–1093. doi:10.1111/j.1461-0248.2010.01509.x
Rousk J, Brookes PC, Baath E (2009) Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. Appl Environ Microbiol 75:1589–1596. doi:10.1128/AEM.02775-08
Gray JV, Petsko GA, Johnston GC et al (2004) “Sleeping Beauty”: Quiescence in Saccharomyces cerevisiae. Microbiol Mol Biol Rev 68:187–206. doi:10.1128/MMBR.68.2.187-206.2004
Novodvorska M, Hayer K, Pullan ST, Wilson R (2013) Transcriptional landscape of Aspergillus niger at breaking of conidial dormancy revealed by RNA-sequencing. BMC Genomics 14:246–250
Hofmann GE, Todgham AE (2010) Living in the now: physiological mechanisms to tolerate a rapidly changing environment. Annu Rev Physiol 72:127–145. doi:10.1146/annurev-physiol-021909-135900
de Boer W, Folman LB, Summerbell RC, Boddy L (2005) Living in a fungal world: impact of fungi on soil bacterial niche development. FEMS Microbiol Rev 29:795–811. doi:10.1016/j.femsre.2004.11.005
Barnard RL, Osborne CA, Firestone MK (2013) Responses of soil bacterial and fungal communities to extreme desiccation and rewetting. ISME J 7:2229–2241. doi:10.1038/ismej.2013.104
Feofilova EP, Ivashechkin AA, Alekhin AI, Sergeeva YE (2011) Fungal spores: dormancy, germination, chemical composition, and role in biotechnology (review). Appl Biochem Microbiol 48:1–11. doi:10.1134/S0003683812010048
Rensing L, Monnerjahn C, Meyer U (1998) Differential stress gene expression during the development of Neurospora crassa and other fungi. FEMS Microbiol Lett 168:159–166
Blazewicz SJ, Barnard RL, Daly RA, Firestone MK (2013) Evaluating rRNA as an indicator of microbial activity in environmental communities: limitations and uses. ISME J 7:2061–2068. doi:10.1038/ismej.2013.102
Buckley DH, Schmidt TM (2003) Diversity and dynamics of microbial communities in soils from agro-ecosystems. Environ Microbiol 5:441–452
Sukenik A, Kaplan-Levy RN, Welch JM, Post AF (2011) Massive multiplication of genome and ribosomes in dormant cells (akinetes) of Aphanizomenon ovalisporum (Cyanobacteria). ISME J 6:670–679. doi:10.1038/ismej.2011.128
Rousk J, Brookes PC, Bååth E (2010) Investigating the mechanisms for the opposing pH relationships of fungal and bacterial growth in soil. Soil Biol Biochem 42:926–934. doi:10.1016/j.soilbio.2010.02.009
Giorgio PAD, Scarborough G (1995) Increase in the proportion of metabolically active bacteria along gradients of enrichment in freshwater and marine plankton: implications for estimates of bacterial growth and production rates. J Plankton Res 17:1905–1924. doi:10.1093/plankt/17.10.1905
Acknowledgments
We thank Zachary Freedman and Sarah Eisenlord for sample collection, John Dunbar, Blaire Steven, and Cedar Hesse for helpful discussions, and two anonymous reviewers whose comments and suggestions greatly improved the manuscript. Funding for this study came from the Department of Energy Biological and Environmental Research program, the Los Alamos National Laborarory LDRD program and the National Science Foundation Long-Term Ecological Research program. This is LANL unclassified report LA-UR-14-25588.
Conflict of Interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 3254 kb)
Rights and permissions
About this article
Cite this article
Mueller, R.C., Gallegos-Graves, L., Zak, D.R. et al. Assembly of Active Bacterial and Fungal Communities Along a Natural Environmental Gradient. Microb Ecol 71, 57–67 (2016). https://doi.org/10.1007/s00248-015-0655-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-015-0655-y