Abstract
Background
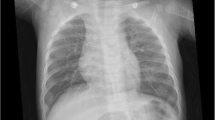
Developmental pulmonary vein pulmonary vein stenosis in the setting of prematurity is a rare and poorly understood condition. Diagnosis can be challenging in the setting of chronic lung disease of prematurity. High-resolution non-contrast chest computed tomography (CT) is the conventional method of evaluating neonates for potential structural changes contributing to severe lung dysfunction and pulmonary hypertension but may miss pulmonary venous stenosis due to the absence of contrast and potential overlap in findings between developmental pulmonary vein pulmonary vein stenosis and lung disease of prematurity.
Objective
To describe the parenchymal changes of pediatric patients with both prematurity and pulmonary vein stenosis, correlate them with venous disease and to describe the phenotypes associated with this disease.
Materials and methods
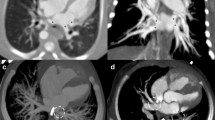
A 5-year retrospective review of chest CT angiography (CTA) imaging in patients with catheterization-confirmed pulmonary vein stenosis was performed to identify pediatric patients (< 18 years) who had a history of prematurity (< 35 weeks gestation). Demographic and clinical data associated with each patient were collected, and the patients’ CTAs were re-reviewed to evaluate pulmonary veins and parenchyma. Patients with post-operative pulmonary vein stenosis and those with congenital heart disease were excluded. Data was analyzed and correlated for descriptive purposes.
Results
A total of 17 patients met the inclusion criteria (12 female, 5 male). All had pulmonary hypertension. There was no correlation between mild, moderate, and severe grades of bronchopulmonary dysplasia and the degree of pulmonary vein stenosis. There was a median of 2 (range 1–4) diseased pulmonary veins per patient. In total, 41% of the diseased pulmonary veins were atretic. The right upper and left upper lobe pulmonary veins were the most frequently diseased (n = 13/17, 35%, n = 10/17, 27%, respectively). Focal ground glass opacification, interlobular septal thickening, and hilar soft tissue enlargement were always associated with the atresia of an ipsilateral vein.
Conclusion
Recognition of the focal parenchymal changes that imply pulmonary vein stenosis, rather than chronic lung disease of prematurity changes, may improve the detection of a potentially treatable source of pulmonary hypertension, particularly where nonangiographic studies result in a limited direct venous assessment.








Similar content being viewed by others
References
Backes CH, Nealon E, Armstrong AK et al (2018) Pulmonary vein stenosis in infants: a systematic review, meta-analysis, and meta-regression. J Pediatr 198:36-45.e3
Mahgoub L, Kaddoura T, Kameny AR, Lopez Ortego P, Vanderlaan RD, Kakadekar A, Dicke F, Rebeyka I, Calderone CA, Redington A, Del Cerro MJ, Fineman J, Adatia I (2017) Pulmonary vein stenosis of ex-premature infants with pulmonary hypertension and bronchopulmonary dysplasia, epidemiology, and survival from a multicenter cohort. Pediatr Pulmonol 52(8):1063–1070. https://doi.org/10.1002/ppul.23679
Vyas-Read S, Varghese NP, Suthar D, Backes C, Lakshminrusimha S, Petit CJ, Levy PT (2022) Prematurity and pulmonary vein stenosis: the role of parenchymal lung disease and pulmonary vascular disease children (basel). 9(5):713https://doi.org/10.3390/children9050713
Thebaud B, Abman SH (2007) Bronchopulmonary dysplasia: where have all the vessels gone? Roles of angiogenic growth factors in chronic lung disease. Am J Respir Crit Care Med 175:978–985
Song MK, Bae EJ, Jeong SI, Kang IS, Kim NK, Choi JY et al (2013) Clinical characteristics and prognostic factors of primary pulmonary vein stenosis or atresia in children. Ann Thorac Surg 95(1):229–234
Jadcherla AV, Backes CH, Cua CL, Smith CV, Levy PT, Ball MK (2021) Primary pulmonary vein stenosis: a new look at a rare but challenging disease. NeoReviews 22(5):e296-308
Kalfa D, Belli E, Bacha E, Lambert V, di Carlo D, Kostolny M, Salminen J, Nosal M, Poncelet A, Horer J, Berggren H, Yemets I, Hazekamp M, Maruszewski B, Sarris G, Pozzi M, Ebels T, Lacour-Gayet F (2017) European congenital heart surgeons association. Primary pulmonary vein stenosis: outcomes, risk factors, and severity score in a multicentric study. Ann Thorac Surg 104(1):182–189. https://doi.org/10.1016/j.athoracsur.2017.03.022
Frank DB, Levy PT, Stiver CA, Boe BA, Baird CW, Callahan RM et al (2021) Primary pulmonary vein stenosis during infancy: state of the art review. J Perinatol Off J Calif Perinat Assoc 41(7):1528–1539
Nasr VG, Callahan R, Wichner Z, Odegard KC, DiNardo JA (2019) Intraluminal pulmonary vein stenosis in children: a “new” lesion. Anesth Analg 129(1):27–40
Callahan R, Kieran MW, Baird CW, Colan SD, Gauvreau K, Ireland CM et al (2018) Adjunct targeted biologic inhibition agents to treat aggressive multivessel intraluminal pediatric pulmonary vein stenosis. J Pediatr 198:29-35.e5
Sena L, Callahan R, Sleeper LA, Beroukhim RS (2021) Prognostic significance of computed tomography findings in pulmonary vein stenosis. Children 8:402
Callahan R, Gauvreau K, Marshall AC, Sena LM, Baird CW, Ireland CM, McEnaney K, Bjornlund EC, Mendonca JT, Jenkins KJ (2021) Outcomes in establishing individual vessel patency for pediatric pulmonary vein stenosis. Children 2021
Brody AS, Guillerman RP, Hay TC et al (2010) Neuroendocrine cell hyperplasia of infancy: diagnosis with high-resolution CT. AJR Am J Roentgenol 194:238–244
Weinman JP, White CJ, Liptzin DR, Deterding RR, Galambos C, Browne LP (2018) High-resolution CT findings of pulmonary interstitial glycogenosis. Pediatr Radiol 48:1066–1072. https://doi.org/10.1007/s00247-018-4138-4
LeMoine BD, Browne LP, Liptzin DR, Deterding RR, Galambos C, Weinman JP (2019) High-resolution computed tomography findings of thyroid transcription factor 1 deficiency (NKX2-1 mutations). Pediatr Radiol 49(7):869–875. https://doi.org/10.1007/s00247-019-04388-3
Barrera CA, Saul D, Rapp JB, Smith CL, White AM, Biko DM, Otero HJ (2020) Diagnostic performance of CT angiography to detect pulmonary vein stenosis in children. Int J Cardiovasc Imaging 36(1):141–147
Lee EY, Callahan R, Vargas SO, Jenkins KJ, Park HJ, Gauthier Z, Winant AJ (2021) Extravascular MDCT findings of pulmonary vein stenosis in children with cardiac septal defect children (Basel). 8(8):667
Guillerman RP (2010) Imaging of childhood interstitial lung disease. Pediatr Allergy Immunol Pulmonol 23(1):43–68
Mižíková I, Morty RE (2015) The extracellular matrix in bronchopulmonary dysplasia: target and source. Front Med (Lausanne) 23(2):91. https://doi.org/10.3389/fmed.2015.00091
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
O’Callaghan, B., Zablah, J.E., Weinman, J.P. et al. Computed tomographic parenchymal lung findings in premature infants with pulmonary vein stenosis. Pediatr Radiol 53, 1874–1884 (2023). https://doi.org/10.1007/s00247-023-05673-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-023-05673-y