Abstract
While many experts in pediatric cardiology have emphasized the importance of palliative care involvement, very few studies have assessed the influence of specialty pediatric palliative care (SPPC) involvement for children with heart disease. We conducted a systematic review using keywords related to palliative care, quality of life and care-satisfaction, and heart disease. We searched PubMed, EMBASE, CINAHL, CENTRAL and Web of Science in December 2023. Screening, data extraction and methodology followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations. Pairs of trained reviewers independently evaluated each article. All full texts excluded from the review were hand-screened for eligible references including systematic reviews in general pediatric populations. Two reviewers independently extracted: (1) study design; (2) methodology; (2) setting; (3) population; (4) intervention/exposure and control definition; (5) outcome measures; and (6) results. Of 4059 studies screened, 9 met inclusion criteria including two with overlapping patient data. Study designs were heterogenous, including only one randomized control and two historical control trials with SPPC as a prospective intervention. Overall, there was moderate to high risk of bias. Seven were single centers studies. In combined estimates, patients who received SPPC were more likely to have advance care planning documented (RR 2.7, [95%CI 1.6, 4.7], p < 0.001) and resuscitation limits (RR 4.0, [2.0, 8.1], p < 0.001), while half as likely to have active resuscitation at end-of-life ([0.3, 0.9], p = 0.032). For parental stress, receipt of SPPC improved scores by almost half a standard deviation (RR 0.48, 95%CI 0.10, 0.86) more than controls. Ultimately, we identified a paucity of high-quality data studying the influence of SPPC; however, findings correlate with literature in other pediatric populations. Findings suggest benefits of SPPC integration for patients with heart disease and their families.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Advancements in pediatric cardiology continue to improve outcomes for children with advanced heart disease (AHD) and their families [1,2,3]. These children are living longer, frequently with more medical complexity, and their families may benefit from support from palliative care [4, 5]. Palliative care provides an interdisciplinary approach to mitigate pain and suffering and to improve the emotional wellbeing and quality of life of seriously ill patients and their caregivers [6,7,8,9]. Many organizations and leaders within pediatric cardiology have acknowledged palliative care as an important aspect of care for children with AHD [5, 10, 11]. Specific needs have been described for cardiac conditions with high mortality rates [e.g., single ventricle heart disease (SVHD) and end-stage heart failure including patients being bridged with ventricular assist device (VAD)] [5, 11,12,13,14]. However overall referral rates for palliative care in pediatric heart disease lag far behind other conditions [10].
While palliative care for children with AHD can be delivered by several models [15, 16], research focuses on consultative subspecialty pediatric palliative care (SPPC) models delivered by a specialized team of interdisciplinary clinicians who provide a range of interventions/services encompassing but not limited to psychosocial support, symptom management, care coordination, hospice services and bereavement [17]. Prior systematic reviews have studied SPPC delivery in a broad range of populations, including oncology, chronic diseases, and neonatology, finding improved quality of life, improved caregiver experience, and higher rates of advance care planning (ACP) [18,19,20].
Limited empiric data characterize impacts of SPPC in pediatric heart disease, despite it being widely identified as a key area of further research [5, 10,11,12,13,14, 21,22,23,24,25]. We sought to describe the published literature assessing the influence of SPPC on the various aspects of care for children with AHD such as advanced care planning and location of death.
Methods
We utilized the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations (Supplementary Table 2), conducted this review in accordance with the Declaration of Helsinki and registered the study on PROSPERO (https://www.crd.york.ac.uk/prospero/, CRD42023484694).
Selection Criteria
We included studies meeting the following criteria: (1) study design: randomized controlled trials (RCT) and comparative observational studies, including cohort, case–control, before-after studies, and cross-sectional studies. Case series or non-comparative cross-sectional studies were excluded; (2) population: pediatric patients age < 21 with a cardiovascular disease diagnosis. Studies that did not limit to this population were included if results were stratified by diagnostic category; (3) intervention/exposure: presence of SPPC as a broad concept encompassing specialized services provided as part of a ‘palliative care team/program’; (4) control: usual/standard care or no SPPC; (5) setting: inpatient, intensive care, ambulatory, community-based, home, or hospice; (6) outcomes: any patient-level (e.g., survival, and mode of death), family-level (e.g., stress, depression), staff-level (e.g., perceptions of end-of-life experience), or health-system-level outcome (e.g., costs); and (7) publication status and language: peer-reviewed studies published in English or Spanish.
Search Strategy
The search was conducted in December 2023 by a medical librarian, using PubMed, EMBASE, CINAHL, Web of Science and Cochrane Central Register of Controlled Trials (CENTRAL). The search strategy included extensive keywords related to palliative care, quality of life and care-satisfaction, and heart disease; terms included, but were not limited to, “palliative care,” “hospice care,” “terminal care,” “quality of life,” “personal satisfaction,” “mental stress,” “congenital heart defect,” and “heart disease” (Supplementary Table 3). Publication date was limited from 2000 through the search date. In EMBASE, the search was limited to exclude conference abstracts. Results were exported to, and deduplicated in EndNote [26].
Study Screening and Selection
All articles were uploaded to Covidence (https://www.covidence.org, Veritas Health Innovation, Melbourne, Australia) for screening and data extraction. Pairs of trained reviewers (JT, KS, VB, CDC, KM) independently evaluated each article generated by the search strategy by the title and abstract. Discrepancies between reviewers were discussed until a consensus was reached among study authors. All full texts excluded from the review were hand-screened for potentially eligible references including systematic reviews in general pediatric populations [19].
Data Extraction
For each article, we collected: (1) study design; (2) methodology; (2) setting; (3) study population; (4) definitions of intervention/exposure and control; (5) outcome measures; and (6) results. Two reviewers independently extracted data (JT, KS, VB, CDC), with conflicts resolved by KM. Study authors were contacted for additional data unavailable in the published articles.
Risk of Bias Assessment
Risk of bias was assessed including reported conflicts of interest and the Quality in Prognosis Studies or the revised Cochrane risk of bias for RCT tools, as appropriate [27, 27,28,29]. Domains were: (1) study participation, (2) study attrition, (3) prognostic factor measurement, (4) outcome measurement, (5) confounder measurement, (6) statistical analysis and reporting domains. A composite quality score was calculated by assigning 0–2 points for high, moderate and low risk of bias in each category (maximum of 12 as low bias, high quality).
Statistical Analysis and Meta-analysis
Total SPPC referral rates were calculated with numerator, denominator and exact binomial confidence intervals overall and restricted to decedents, excluding the prospective interventional studies.
We performed a meta-analysis to test for homogeneity of effects across studies and obtain a pooled estimate of treatment effect over the individual studies. Studies with duplicate/overlapping patient data were excluded from meta-analyses. For dichotomous outcomes, we estimated the relative risk for intervention vs control; for time-to-event outcomes (surgical and length of stay), we estimated the ratio of means or medians for intervention vs control (under an accelerated time-to-event model the ratio of means and medians are the same) [30].
For studies with continuous outcome measures that used different scoring systems we used Cohen’s d as the intervention effect in each study. Cohens d is the difference in means in intervention versus control divided by the pooled standard deviation, and thus can be interpreted as the differences in intervention versus control in terms of standard deviations with appropriate effect size cutoffs [31]. This measures an intervention effect in a study that is not dependent on the scoring system/measure itself, and thus can be compared/combined across studies. These studies with continuous outcomes were pre/post studies, so the intervention effect is Cohen’s d in terms of difference-in-differences (mean ‘pre minus post’ difference in intervention, minus mean ‘pre minus post’ difference in control). The DerSimonian and Laird test was used to test for homogeneity of intervention effect across studies [32]. If the ‘homogeneity test’ was rejected combined risk ratio probabilities were not presented. If homogeneity was established; the pooled estimate was determined as a weighted average of the intervention effect estimates from the individual studies [33]. The weights used in the pooled estimate equal the inverse of the variance of the estimated intervention effect from each study. Given the small number of studies, the meta-analysis was performed assuming a fixed effect for each study. We determined the 95% confidence interval for the pooled intervention effects, as well as a p value for the null hypothesis that the pooled intervention effect equals the null value. A forest plot was used to display the intervention effect for each individual study and the pooled estimate.
Results
Study Characteristics
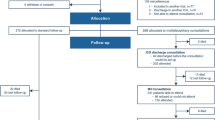
Of 4059 studies screened and 65 assessed for eligibility at full-text, nine met study inclusion criteria including two with overlapping patient data (Fig. 1) [34,35,36,37,38,39,40,41,42]. All studies were from the United States. Six were retrospective cohort observational studies with chart review to determine SPPC involvement as part of clinical care, including two with additional cross-sectional survey components (one of staff and one of bereaved parents) [35,36,37,38,39, 41]. Three involved SPPC consultation as a prospective intervention (two historical control trials and one randomized control trial) [34, 40, 42]. Studying outcomes in cardiac disease according to SPPC involvement was the primary aim in six (66.7%) [35,36,37, 40,41,42]. Seven (77.8%) were single center studies [35,36,37, 39,40,41,42]. Only one included all disease groups and sub-defined results for cardiac patients [34]. Of the remaining, five included all cardiac diseases [35,36,37,38,39] (three focused on cardiac intensive care unit [CICU] patients) [35, 36, 39], one was in VAD patients [41] and two were in neonates [40, 42] (one restricted to SVHD) [40]. Population sample sizes are shown (Table 1). Overall, most studies had moderate- to high-risk for bias (Table 1). While the prospective interventional study populations were well-matched, no study performed multivariable analyses [34, 40, 42].
SPPC Referral Rates
Overall SPPC referral rate was 3.7% [95%CI 3.3, 4.1] (n = 326/8885), compared to 40.1% when restricted to decedents [95%CI 35.4, 40.1] (n = 165/411). Among studies of decedents, this ranged from 15.9% in the oldest study to 61.5% in the VAD study.
Outcomes by SPPC
Clinical outcomes examined according to SPPC involvement are summarized in Table 2. Synthesis by clinical outcome is shown for findings restricted to decedents (Table 3) and overall (Table 4).
Patient-Level
Length of stay (LOS) was examined in six studies [34,35,36,37, 40, 42], survival (four) [35, 36, 40, 41], mode of death (four) [35,36,37, 41], location of death (three) [35,36,37], cardiac interventions (surgical vs medical, two studies) [35, 36], ACP (two) [37, 41], resuscitation limitations (two) [37, 41], intensity of medical therapies (one) [37]. Combined risk ratios and forest plots for key outcomes examined in multiple studies are shown (Fig. 2). Patients who received SPPC were 2.7 times more likely to have ACP documented (95%CI 1.6, 4.7, p < 0.001), 4.0 times more likely to have resuscitation limits (2.0, 8.1, p < 0.001), half as likely to have active resuscitation as the mode of death at end-of-life (EOL) (0.3, 0.9, p = 0.032). LOS and survival were not adjusted by disease characteristics in any study. In both, the ‘homogeneity test’ was rejected with discordant direction of associations between retrospective and prospective interventional results.
Forest Plots of Relative Risk for Intervention versus Controls for Patient-Level Outcomes. Forest plots demonstrating that combined analyses of relevant studies show statistically increased advanced care planning documentation, increased limitations on resuscitation, and reduced active resuscitation at end-of-life
Parent-, Staff- and System-Level
Survey response studies evaluated bereaved parent perceptions of a “good death” [38], parental stress [40, 42], depression [40, 42], anxiety [42], coping [40] and measures of quality of life [40]. For parental stress, receipt of a SPPC intervention improved the scores by almost a half a standard deviation (0.48, 05%CI 0.10, 0.86) pre to post more than controls in this combined estimate (Fig. 3). Only one study examined staff perceptions of quality of dying and death with mixed results overall, but involvement of palliative care increased staff-rated quality of life for the 7 days prior to death [39]. Two examined costs/cost distribution finding a reduction in hospital charges with transition to outpatient care [34, 37].
Forest Plot of Relative Risk for Intervention versus Controls for Parental Stress. Forest plot demonstrates statistically significantly improved parental stress based on two relevant studies. Relative risk (RR) estimates provided per study (first author and year named) and combined (pooled estimates, diamond symbol) with 95% confidence intervals (CI) (whiskers) with line at a RR of 1. Active Resuscitation at End-of-Life has a different RR scale
Notable Excluded Full-Text Studies
Of 54 articles excluded at full-text, 17 were non-empiric review articles. Of these, eleven specifically discussed the role of SPPC in cardiac patients [5, 10,11,12,13,14, 21,22,23,24,25].
Discussion
This comprehensive systematic review and meta-analysis summarizes the published literature on the role of SPPC in pediatric heart disease. Overall, we identified limited empiric data, generally low in quality, solely from the US, and mostly single center studies with small sample sizes. Even with a paucity of studies, meta-analysis identified benefits of SPPC at both a patient- and family-level for decedents and survivors, with individual studies supporting system-level in-hospital cost reduction and improved staff rating of quality of life prior to death. Pediatric patients who received SPPC were less likely to experience active resuscitation at EOL, and were more likely to have documentation of ACP and resuscitation limits. Additionally, SPPC involvement was found to decrease parental stress. Identified gaps and results offer opportunities for future research and interventions to improve holistic care for children with cardiac disease.
Data supporting benefits of SPPC for children with heart disease mirror results in other populations. The evaluated studies show significant increases in ACP documentation and limits to resuscitation for pediatric patients with AHD who received SPPC, compared to those who did not receive SPPC. This is similar to findings in oncology patients and children with complex chronic conditions who subsequently die as inpatients [18, 43]. One included study evaluated the perspectives of parents of children heart disease who died and found they are more likely to perceive a “good death” experience if they felt prepared, participated in ACP, and reported non-cure-oriented goals-of care [38]. This is consistent with research in parents of children with other complex chronic conditions that shows that parents highly value ACP early in the illness course and that this improves parent-reported EOL outcomes [44].
Of pediatric cardiac patients who died, those who received SPPC were half as likely to experience active resuscitation at EOL. Similar observations were noted in general pediatric patients and children with cancer with SPPC involvement who were both less likely to experience resuscitative events prior to death [18, 45]. Importantly, one included study showed active CPR negatively influenced a family’s perception of a “good death” experience for their child with heart disease [38]. Taken together, core principles of palliative care may promote goal-concordant EOL experiences in this population.
Although EOL and SPPC are often falsely conflated, our meta-analysis demonstrated benefits of SPPC extend beyond decedents. Among parents, parental stress decreased with the receipt of SPPC services [40, 42]. Patients with SVHD have among the highest risk of mortality in AHD, so unsurprisingly this population received increased attention. The randomized control trial of early SPPC intervention for mothers of children with SVHD found decreased maternal anxiety and improved communication and family relationships [40] while parental depression and anxiety did not decrease with the intervention in all-comer neonates with AHD [42]. As part of a quality improvement initiative aiming to improve collaboration between one institution’s Single Ventricle (SV) team and SPPC team, a preliminary survey of Heart Center staff showed that 88% of respondents agreed or strongly agreed that routine involvement of the SPPC team with SV patients has improved the overall psychosocial and/or decision-making support provided to families [46]. While this study lacked a control, it suggests that providers of pediatric cardiac care see benefit from routine involvement for families of children with SVHD.
AHD is a leading cause of disease-related death in US children, and issues around EOL for these patients deserve attention [47]. Despite identifying eleven review articles specifically discussing the role of palliative care in the pediatric cardiac population emphasizing this as a key emerging area and the benefits described, overall SPPC referral rates were low [5, 10,11,12,13,14, 21,22,23,24,25]. Though SPPC involvement has been increasing in children with AHD [37], referral rates have historically been low compared to other pediatric complex conditions [18, 45]. Barriers to SPPC involvement include concerns from pediatric cardiologists of “undermining parental hope” and parents perceiving they are “giving up” [48, 49]. Notably, in a study evaluating features of SPPC involvement for children with AHD, most families expressed that life prolongation was a priority at the time of initial consultation, which suggests that parents can retain hope and concurrently receive SPPC services [19]. Our evidence suggests that SPPC involvement leads to increased ACP, modes/locations of death that may be more goal-concordant, and improvements in psychosocial support for families of children with AHD. Comparable benefits of SPPC to other patient populations should prompt improved integration of SPPC into the care of pediatric patients with AHD to improve the experience of patients and families as early as immediately after diagnosis, up to end-of-life.
Limitations and Strengths
There are limitations to consider when interpreting results. Studies are heterogenous in included populations (e.g., CICU decedents vs VAD patients) and the outcome measures examined with no standardized approach to evaluating EOL metrics in pediatric AHD. Overall studies were of low-quality with only 1 RCT, limiting ability to make causal inferences. All studies were conducted in the US in English language, and all were single center apart from one study evaluating parental perspectives from two institutions [38] and a state-based SPPC program implementation study [34], limiting generalizability. Clinicians may be more likely to consult SPPC for children with higher illness severity and/or challenging social situations; we could not assess the impact of these and other potential confounders on mortality and LOS. The fact that benefits of SPPC were observed in retrospective studies where selection bias for referral may exist could suggest larger true benefits. While definitions of SPPC differed between study designs, variable care delivery between hospitals is universal. This review was unable to examine individual interventions or frequency of interventions. Furthermore, patients in the control group may have had effective “primary” palliative care (comprehensive care with a palliative approach delivered by the primary cardiology/intensive care team) which cannot be quantified. When aggregated, statistical findings were stronger for several outcomes despite only two included studies, indicating small sample sizes may also preclude accurate conclusions without meta-analyses. We were unable to include hospice involvement in the meta-analysis due to different definitions of hospice in the included studies, i.e., home with hospice care versus institutional hospice care. This limitation is significant, as dying at home or under hospice conditions is one of the potential outcomes of interest of SPPC involvement described in other pediatric populations [18].
Future Directions
More work is needed in exploring logistics of optimal SPPC delivery models across different cardiac care environments including when and how subspecialty teams should to be involved versus primary models of delivery. Considering this must span prenatal, intensive care, inpatient, and outpatient involvement, there needs to be parallel efforts to both enhance primary palliative care through training of cardiology and critical care fellows [50, 51] and to consider novel integration approaches [15].
Additional studies are necessary to deepen the body of literature describing the impact of SPPC on the care of children with AHD. Outcomes in retrospective studies primarily centered around SPPC impact on EOL, so an opportunity exists for further research to evaluate the impacts on more holistic benefits extending to survivors or bereavement support after death. Additionally, EOL outcomes studied for pediatric patients are often extrapolated from adult EOL care priorities, such as the location of death being at home. Parental perspectives about preference for location of death has not been studied in pediatric heart disease, and future studies should identify whether preferred location of death was elicited, and if the ultimate location of death was concordant with that preference. There is a dearth of studies evaluating the perspectives of children and adolescents with heart disease receiving SPPC services.
Barriers to more generalizable research through multicenter studies include limitations to ICD-10 coding and cardiac registry data collection. Collaboration with pediatric cardiology and cardiac surgery registries could move the field forward by significantly facilitating larger scale studies [52, 53]. Defining high-quality pediatric EOL care is a priority offering opportunities for standardization of outcomes, which would allow for improved analysis of SPPC impact across institutions [54]. Despite quality metrics for EOL care in oncology [54, 55], no measures are defined for children with AHD, which is a critical gap demanding further research.
Conclusion
Our systematic review and meta-analysis shows benefits of SPPC for these patients, at both a patient- and family-level with decreased parental stress, less active resuscitation at EOL, and increased ACP documentation and resuscitation limits compared to those without SPPC. Children with AHD are a unique subpopulation of pediatric patients with potentially limited lifespans and high intensity of care through EOL. Overall we identified a paucity of high-quality data studying the influence of SPPC, however findings correlate with literature in other pediatric populations. Results illuminate gaps and future opportunities for research.
Data Availability
Systematic review extraction and metanalysis data will be made available on request.
References
Lynn MM, Salemi JL, Kostelyna SP et al (2022) Lesion-specific congenital heart disease mortality trends in children: 1999 to 2017. Pediatrics 150:e2022056294. https://doi.org/10.1542/peds.2022-056294
Gaies M, Pasquali SK, Banerjee M et al (2019) Improvement in pediatric cardiac surgical outcomes through interhospital collaboration. J Am Coll Cardiol 74:2786–2795. https://doi.org/10.1016/j.jacc.2019.09.046
Hagel JA, Sperotto F, Laussen PC et al (2021) Shock index, coronary perfusion pressure, and rate pressure product as predictors of adverse outcome after pediatric cardiac surgery. Pediatr Crit Care Med J Soc Crit Care Med World Fed Pediatr Intensive Crit Care Soc 22:e67–e78. https://doi.org/10.1097/PCC.0000000000002524
Yu JA, Schenker Y, Maurer SH et al (2019) Pediatric palliative care in the medical neighborhood for children with medical complexity. Fam Syst Health J Collab Fam Healthc 37:107–119. https://doi.org/10.1037/fsh0000414
Blume ED, Kirsch R, Cousino MK et al (2023) Palliative care across the life span for children with heart disease: a scientific statement from the american heart association. Circ Cardiovasc Qual Outcomes. https://doi.org/10.1161/HCQ.0000000000000114
Center to Advance Palliative Care (2016) Pediatric Palliative Care Field Guide. In: Cent. Adv. Palliat. Care. https://www.capc.org/documents/download/257/. Accessed 25 Apr 2024
World Health Organization (2023) Palliative care for children. In: World Health Organ. https://www.who.int/europe/news-room/fact-sheets/item/palliative-care-for-children. Accessed 25 Apr 2024
Norris SE, McCabe ME (2023) Hospice and palliative medicine: pediatric essentials. Pediatr Rev 44:255–264. https://doi.org/10.1542/pir.2020-004259
Center to Advance Palliative Care. The Value of Palliative Care. In: Cent. Adv. Palliat. Care. https://www.capc.org/the-case-for-palliative-care/. Accessed 1 May 2024
Morell E, Moynihan K, Wolfe J, Blume ED (2019) Palliative care and paediatric cardiology: current evidence and future directions. Lancet Child Adolesc Health 3:502–510. https://doi.org/10.1016/S2352-4642(19)30121-X
Kirk R, Dipchand AI, Rosenthal DN et al (2014) The International Society for Heart and Lung Transplantation guidelines for the management of pediatric heart failure: executive summary. J Heart Lung Transplant 33:888–909. https://doi.org/10.1016/j.healun.2014.06.002
Kaufman BD, Cohen HJ (2019) Palliative care in pediatric heart failure and transplantation. Curr Opin Pediatr 31:611–616. https://doi.org/10.1097/MOP.0000000000000799
Hope KD, Bhat PN, Dreyer WJ et al (2021) Pediatric palliative care in the heart failure, ventricular assist device and transplant populations: supporting patients. Fam Clin Teams Child 8:468. https://doi.org/10.3390/children8060468
Cousino MK, Lord BT, Blume ED (2022) State of the science and future research directions in palliative and end-of-life care in paediatric cardiology: a report from the Harvard Radcliffe Accelerator Workshop. Cardiol Young 32:431–436. https://doi.org/10.1017/S104795112100233X
Moynihan KM, Snaman JM, Kaye EC et al (2019) Integration of pediatric palliative care into cardiac intensive care: a champion-based model. Pediatrics 144:e20190160. https://doi.org/10.1542/peds.2019-0160
Sreedhar SS, Kraft C, Friebert S (2020) Primary palliative care: skills for all clinicians. Curr Probl Pediatr Adolesc Health Care 50:100814. https://doi.org/10.1016/j.cppeds.2020.100814
Benini F, Papadatou D, Bernadá M et al (2022) International standards for pediatric palliative care: from IMPaCCT to GO-PPaCS. J Pain Symptom Manag 63:e529–e543. https://doi.org/10.1016/j.jpainsymman.2021.12.031
Kaye EC, Weaver MS, DeWitt LH et al (2021) The impact of specialty palliative care in pediatric oncology: a systematic review. J Pain Symptom Manag 61:1060-1079.e2. https://doi.org/10.1016/j.jpainsymman.2020.12.003
Marcus KL, Balkin EM, Al-Sayegh H et al (2018) Patterns and outcomes of care in children with advanced heart disease receiving palliative care consultation. J Pain Symptom Manag 55:351–358. https://doi.org/10.1016/j.jpainsymman.2017.08.033
Zuniga-Villanueva G, Widger K, Medeiros C et al (2021) Specialized pediatric palliative care in neonates with life-limiting illness: a systematic review. Am J Perinatol 38:e318–e329. https://doi.org/10.1055/s-0040-1710031
Bergsträsser E, Lukose S, Zimmermann K, Oxenius A (2022) Palliative care in children with advanced heart disease in a tertiary care environment: a mini review. Front Cardiovasc Med 9:863031. https://doi.org/10.3389/fcvm.2022.863031
Wan A, Weingarten K, Rapoport A (2020) Palliative care?! But this child’s not dying: the burgeoning partnership between pediatric cardiology and palliative care. Can J Cardiol 36:1041–1049. https://doi.org/10.1016/j.cjca.2020.04.041
Bertaud S, Lloyd DFA, Laddie J, Razavi R (2016) The importance of early involvement of paediatric palliative care for patients with severe congenital heart disease. Arch Dis Child 101:984–987. https://doi.org/10.1136/archdischild-2015-309789
Mazwi ML, Henner N, Kirsch R (2017) The role of palliative care in critical congenital heart disease. Semin Perinatol 41:128–132. https://doi.org/10.1053/j.semperi.2016.11.006
Afonso NS, Ninemire MR, Gowda SH et al (2021) Redefining the relationship: palliative care in critical perinatal and neonatal cardiac patients. Child Basel Switz 8:548. https://doi.org/10.3390/children8070548
Bramer WM, Giustini D, de Jonge GB et al (2016) De-duplication of database search results for systematic reviews in EndNote. J Med Libr Assoc 104:240–243. https://doi.org/10.3163/1536-5050.104.3.014
Hayden JA, van der Windt DA, Cartwright JL et al (2013) Assessing bias in studies of prognostic factors. Ann Intern Med 158:280–286. https://doi.org/10.7326/0003-4819-158-4-201302190-00009
Sterne JAC, Savović J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898. https://doi.org/10.1136/bmj.l4898
Higgins JP, Sterne JA, Savovic J et al (2016) A revised tool for assessing risk of bias in randomized trials. Cochrane Database Syst Rev 10:29–31
Dey T, Lipsitz SR, Cooper Z et al (2022) Regression modeling of time-to-event data with censoring. Nat Methods 19:1513–1515. https://doi.org/10.1038/s41592-022-01689-8
Sullivan GM, Feinn R (2012) Using effect size-or why the p value is not enough. J Grad Med Educ 4:279–282. https://doi.org/10.4300/JGME-D-12-00156.1
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188. https://doi.org/10.1016/0197-2456(86)90046-2
Chang B, Waternaux C, Lipsitz S (2001) Meta-analysis of binary data: which within study variance estimate to use? Stat Med 20:1947–1956. https://doi.org/10.1002/sim.823
Gans D, Hadler MW, Chen X et al (2016) Cost analysis and policy implications of a pediatric palliative care program. J Pain Symptom Manag 52:329–335. https://doi.org/10.1016/j.jpainsymman.2016.02.020
Delgado-Corcoran C, Wawrzynski SE, Bennett EE et al (2020) Palliative care in children with heart disease treated in an ICU. Pediatr Crit Care Med 21:423–429. https://doi.org/10.1097/PCC.0000000000002271
Delgado-Corcoran C, Bennett EE, Bodily SA et al (2021) Prevalence of specialised palliative care consultation for eligible children within a paediatric cardiac ICU. Cardiol Young 31:1458–1464. https://doi.org/10.1017/S1047951121000433
Moynihan KM, Heith CS, Snaman JM et al (2021) Palliative care referrals in cardiac disease. Pediatrics 147:e2020018580. https://doi.org/10.1542/peds.2020-018580
Moynihan KM, Ziniel SI, Johnston E et al (2022) A “good death” for children with cardiac disease. Pediatr Cardiol 43:744–755. https://doi.org/10.1007/s00246-021-02781-0
Bailey V, Beke DM, Snaman JM et al (2022) Assessment of an instrument to measure interdisciplinary staff perceptions of quality of dying and death in a pediatric cardiac intensive care unit. JAMA Netw Open 5:e2210762. https://doi.org/10.1001/jamanetworkopen.2022.10762
Hancock HS, Pituch K, Uzark K et al (2018) A randomised trial of early palliative care for maternal stress in infants prenatally diagnosed with single-ventricle heart disease. Cardiol Young 28:561–570. https://doi.org/10.1017/S1047951117002761
Knoll C, Kaufman B, Chen S et al (2020) Palliative care engagement for pediatric ventricular assist device patients: a single-center experience. ASAIO J 66:929–932. https://doi.org/10.1097/MAT.0000000000001092
Callahan K, Steinwurtzel R, Brumarie L et al (2019) Early palliative care reduces stress in parents of neonates with congenital heart disease: validation of the “Baby, Attachment, Comfort Interventions.” J Perinatol 39:1640–1647. https://doi.org/10.1038/s41372-019-0490-y
Spraker-Perlman HL, Tam RP, Bardsley T et al (2019) The impact of pediatric palliative care involvement in the care of critically ill patients without complex chronic conditions. J Palliat Med. https://doi.org/10.1089/jpm.2018.0469
DeCourcey DD, Silverman M, Oladunjoye A, Wolfe J (2019) Advance care planning and parent-reported end-of-life outcomes in children, adolescents, and young adults with complex chronic conditions*. Crit Care Med 47:101–108. https://doi.org/10.1097/CCM.0000000000003472
Trowbridge A, Walter JK, McConathey E et al (2018) Modes of death within a children’s hospital. Pediatrics 142:e20174182. https://doi.org/10.1542/peds.2017-4182
Davis JAM, Bass A, Humphrey L et al (2020) Early integration of palliative care in families of children with single ventricle congenital heart defects: a quality improvement project to enhance family support. Pediatr Cardiol 41:114–122. https://doi.org/10.1007/s00246-019-02231-y
Centers for Disease Control and Prevention, National Center for Injury Prevention and Control (2023) Leading Causes of Death and Injury. CDC. https://www.cdc.gov/injury/wisqars/leadingcauses.html. Accessed 19 Feb 2024
Morell E, Thompson J, Rajagopal S et al (2021) Congenital cardiothoracic surgeons and palliative care: a national survey study. J Palliat Care 36:17–21. https://doi.org/10.1177/0825859719874765
Balkin EM, Sleeper LA, Kirkpatrick JN et al (2018) Physician perspectives on palliative care for children with advanced heart disease: a comparison between pediatric cardiology and palliative care physicians. J Palliat Med 21:773–779. https://doi.org/10.1089/jpm.2017.0612
Das N, Brown A, Harris TH (2024) Delivering serious news in pediatric cardiology—a pilot program. Pediatr Cardiol. https://doi.org/10.1007/s00246-024-03440-w
Brock KE, Tracewski M, Allen KE et al (2019) Simulation-based palliative care communication for pediatric critical care fellows. Am J Hosp Palliat Care 36:820–830. https://doi.org/10.1177/1049909119839983
O’Keefe S, Czaja AS (2021) Validation of administrative codes for palliative care consultation among critically Ill children. Hosp Pediatr 11:179–182. https://doi.org/10.1542/hpeds.2020-001263
Pediatric Cardiac Critical Care Consortium (2023) PC4 Revised Data Definitions Manual. In: Pediatr. Card. Crit. Care Consort. PC4. https://pc4.arbormetrix.com/Registry/static/pc4/content/PC4_Revised_Data_Definitions_Manual_FAQ_v3.pdf. Accessed 2 Mar 2024
Johnston EE, Martinez I, Wolfe J, Asch SM (2021) Quality measures for end-of-life care for children with cancer: a modified Delphi approach. Cancer 127:2571–2578. https://doi.org/10.1002/cncr.33546
Ananth P, Lindsay M, Nye R et al (2022) End-of-life care quality for children with cancer who receive palliative care. Pediatr Blood Cancer 69:e29841. https://doi.org/10.1002/pbc.29841
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
Conceptualization: Drs Moynihan, Delgado-Corcoran, Ting and Songer. Literature search: Ms Rotman. Data analysis: Dr Lipsitz. Screening and Extraction: Drs Moynihan, Delgado-Corcoran, Ting and Songer and Ms Bailey. Drafted initial manuscript: Drs Moynihan, Ting and Songer. Critically revised the work for important intellectual content: Drs Delgado-Corcoran, Lipsitz and Rosenberg, and Ms Bailey.
Corresponding author
Ethics declarations
Competing interests
The authors have no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ting, J., Songer, K., Bailey, V. et al. Impact of Subspecialty Pediatric Palliative Care on Children with Heart Disease; A Systematic Review and Meta-analysis. Pediatr Cardiol (2024). https://doi.org/10.1007/s00246-024-03535-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00246-024-03535-4