Abstract
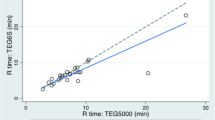
Thromboelastography (TEG) is a point-of-care test (POCT) used to analyze the hemostatic properties of whole blood. TEG® 5000and TEG® 6s (Haemonetics Corp, USA) measure the same parameters describing clot viscoelasticity using different methodologies. The purpose of this study was to evaluate agreement between TEG5000 and TEG6s measurements. We analyzed prospectively collected tests resulting from paired blood samples in cardiac surgery pediatric patients at one hour (T0) and 24 h (T1) postoperatively. Each citrated sample was utilized for TEG® 5000 and TEG ®6s. Six specific TEG parameters were analyzed and compared: R kaolin time (RK), R kaolin heparinase (RKH) time, K kaolin time (KK), K kaolin heparinase time KH (KKH), Maximum Amplitude kaolin (MAK), Maximal Amplitude Kaolin Heparinase (MAKH). We enrolled 30 patients. Median (interquartile range) patients’ age was 206 (20–597) days. All surgical patients underwent correction except 5 who were palliated. At T0, RK and RKH showed an average (standard deviation) % bias of 15.8 (31) and 16.1 (28), respectively, with similar results at T1. A % bias of -6 (23) and − 6 [15] in MAK was found at T0 and T1, respectively. Similarly, MAKH % bias was 1.5 (22) and 7.6 (29) at T0 and T1, respectively. At both timepoints, low % biases (< ± 6%) were demonstrated in KK and KKH. All parameters showed improved coagulation from T0 to T1, but without significant interaction between type of device and time. Analysis of the entire pool of 60 paired samples showed no agreement in diagnostic performance (within the range vs. outside the range) in 12 (20%), 5 (9.8%), 1 (1.7%), 4 (7.8%), 9 (15%), and 5 (9.8%) cases for RK, RKH, MAK, MAKH, KK and KKH, respectively. We observed substantial agreement in MAK and KK in a cohort of pediatric patients undergoing uncomplicated cardiac surgery. Our findings suggest that TEG®5000 and TEG®6s are interchangeable for assessing these parameters.

Similar content being viewed by others
References
Williams GD, Bratton SL, Nielsen NJ, Ramamoorthy C (1998) Fibrinolysis in pediatric patients undergoing cardiopulmonary bypass. J Cardiothorac Vasc Anesth 12(6):633–638. https://doi.org/10.1016/s1053-0770(98)90233-6
Ignjatovic V, Than J, Summerhayes R, Newall F, Horton S, Cochrane A, Monagle P (2011) Hemostatic response in paediatric patients undergoing cardiopulmonary bypass surgery. Pediatr Cardiol 32(5):621–627. https://doi.org/10.1007/s00246-011-9929-4
Ravn HB, Andreasen JB, Hvas AM (2017) Does whole blood coagulation analysis reflect developmental haemostasis? Blood Coagul Fibrinolysis 28(3):218–223. https://doi.org/10.1097/MBC.0000000000000585
Chan KL, Summerhayes RG, Ignjatovic V, Horton SB, Monagle PT (2007) Reference values for kaolin-activated thromboelastography in healthy children. Anesth Analg 105(6):1610–1613. https://doi.org/10.1213/01.ane.0000287645.26763.be
Kettner SC, Pollak A, Zimpfer M, Seybold T, Prusa AR, Herkner K, Kuhle S (2004) Heparinase-modified thrombelastography in term and preterm neonates. Anesth Analg 98(6):1650–1652. https://doi.org/10.1213/01.ANE.0000115149.25496.DD
Perez-Ferrer A, Vicente-Sanchez J, Carceles-Baron MD, Van der Linden P, Faraoni D (2015) Early thromboelastometry variables predict maximum clot firmness in children undergoing cardiac and non- cardiac surgery. Br J Anaesth 115(6):896–902. https://doi.org/10.1093/bja/aev369.PMID
Faraoni D, Meier J, New HV, Van der Linden PJ, Hunt BJ (2019) Patient blood management for neonates and children undergoing cardiac surgery: 2019 NATA Guidelines. J Cardiothorac Vasc Anesth 33(12):3249–3263. https://doi.org/10.1053/j.jvca.2019.03.036
Rizza A, Ricci Z, Pezzella C, Favia I, Di Felice G, Ranucci M, Cogo P (2017) Kaolin- activated thromboelastography and standard coagulation assays in cyanotic and acyanotic infants undergoing complex cardiac surgery: a prospective cohort study. Paediatr Anaesth 27(2):170–180. https://doi.org/10.1111/pan.13038
Emani S, Emani VS, Diallo FB, Dutta P, Matte GS, Nathan M, Ibla JC, Emani SM (2022) Comparison of Thromboelastography Devices TEG®6S Point of Care device vs. TEG®5000 in Pediatric Patients undergoing cardiac surgery. J Extra Corpor Technol 54:42–49. https://doi.org/10.1182/ject-42-49
Gurbel PA, Bliden KP, Tantry US, Monroe AL, Muresan AA, Brunner NE, Lopez-Espina CG, Delmenico PR, Cohen E, Raviv G, Haugen DL, Ereth MH (2016) First report of the point-of-care TEG: a technical validation study of the TEG-6S system. Platelets 27(7):642–649. https://doi.org/10.3109/09537104.2016.1153617
Wong Q, Byrne KP, Robinson SC (2020) Clinical agreement and interchangeability of TEG5000 and TEG6s during cardiac surgery. Anaesth Intensive Care 48(1):43–52. https://doi.org/10.1177/0310057X19897657
Robson JL, Dj Watts A, McCulloch TJ, Paleologos MS, Mortimer RA, Ca Kam P (2019) Correlation and agreement between the TEG® 5000 and the TEG® 6s during liver transplant surgery. Anaesth Intensive Care 47(1):32–39. https://doi.org/10.1177/0310057X18811731
Lloyd-Donald P, Churilov L, Zia F, Bellomo R, Hart G, McCall P, Mårtensson J, Glassford N, Weinberg L (2019) Assessment of agreement and interchangeability between the TEG5000 and TEG6S thromboelastography haemostasis analysers: a prospective validation study. BMC Anesthesiol 30(1):45. https://doi.org/10.1186/s12871-019-0717-7
Wegner J, Popovsky MA (2010) Clinical utility of thromboelastography: one size does not fit all. Semin Thromb Hemost 36(07):699–706. https://doi.org/10.1055/s-0030-1265286
Moynihan KM, Johnson K, Rane M, Norman A, Humphreys S, Stocker C, Gibbons K, Roy J (2021) Pediatric Thromboelastograph 6s and Laboratory Coagulation reference values. Arch Pathol Lab Med 145(11):1413–1423. https://doi.org/10.5858/arpa.2020-0647-OA
Funding
No financial support was required for this study.
Author information
Authors and Affiliations
Contributions
All authors wrote the manuscript and reviewed all text and figures. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no potential conflicts of interest to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Benegni, S., Giorni, C., Raggi, V. et al. Diagnostic Agreement Between TEG5000 and TEG6S in the Assessment of Hemostasis in Pediatric Cardiac Surgery: A Prospective Non-inferiority Study. Pediatr Cardiol (2023). https://doi.org/10.1007/s00246-023-03311-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00246-023-03311-w