Abstract
Fluid restriction and diuretic management are mainstays in the postoperative management of cardiac patients, at risk of volume overload and its deleterious effects on primary cardiac function and multi-organ systems. The importance of fluid homeostasis is further emphasized among orthotopic heart transplant recipients (OHT). We sought to investigate the relationship between postoperative volume overload, mortality, and allograft dysfunction among pediatric OHT recipients within 1-year of transplantation. This is a retrospective cohort study from a single pediatric OHT center. Children under 21 years undergoing cardiac transplantation between 2010 and 2018 were included. Cumulative fluid overload (cFO) was assessed as percent fluid accumulation adjusted for preoperative body weight. Greater than 10% cFO defined those with postoperative cFO and a comparison of postoperative cFO vs. no postoperative cFO (< 5%) is reported. 102 pediatric OHT recipients were included. Early cFO at 72 h post-OHT occurred in 14% and overall cFO at 1-week post-OHT occurred in 23% of patients. Risk factors for cFO included younger age, lower weight, and postoperative ECMO. Early cFO was associated with postoperative mortality at 1-year, OR 8.6 (95% CI 1.4, 51.6), p = 0.04, independent of age and weight. There was no significant relationship between cFO and allograft dysfunction, measured by rates of clinical rejection and cardiopulmonary filling pressures within 1-year of transplant. Early postoperative volume overload is prevalent and associated with increased risk of death at 1-year among pediatric OHT recipients. It may be an important postoperative marker of transplant survival, and this relationship warrants further clinical investigation.
Similar content being viewed by others
Introduction
Fluid therapy is widely recognized as essential practice for the acute resuscitation of critically ill children [1, 2]. However, aggressive fluid resuscitation beyond the acute resuscitation period may lead to early fluid overload and worse clinical outcomes [3]. Early fluid overload has been further investigated both as an early prognostic marker of poor clinical outcomes and an important potential modifiable risk factor in pediatric critical illness, shaping current clinical standards of fluid-goal directed therapy [3,4,5,6,7,8,9,10,11].
Post-cardiac surgery patients are considered particularly vulnerable to both postoperative volume overload and its secondary complications that result in severe morbidity and mortality [12, 13]. The risk of volume overload is multifactorial and often compounded by the effect of preoperative heart failure, the physiological changes following cardiopulmonary bypass that include capillary leak and low cardiac output syndrome, and renal dysfunction [14,15,16,17,18]. Further, early fluid overload following postoperative low cardiac output syndrome reduces end-organ perfusion pressure and propagates a cycle of dysregulated pathophysiological mechanisms that exacerbate systemic organ failure, tissue edema, and volume overload [19]. Fluid restriction and aggressive diuretic support have been the mainstay of post-operative management of cardiac surgical patients, among adult and pediatric patients [12, 13, 20, 21].
In orthotopic heart transplant recipients (OHT), the importance of fluid homeostasis is further emphasized in clinical practice. A maladaptive neuroendocrine and impaired renal response to plasma volume expansion have been observed in postoperative adult OHT patients and associated with both fluid overload and cardiac dysfunction in the early postoperative period [22]. Although the causality of this relationship is not entirely clear, interruptions to key neural and humoral homeostatic mechanisms from graft denervation has been previously described [23, 24]. Among sparse pediatric literature, a recent small retrospective study reported a high incidence of postoperative volume overload among pediatric heart transplant recipients and an independent association with poor short-term outcomes including postoperative mortality in those with severe fluid overload [21].
In our study, we sought to describe the incidence of postoperative cumulative fluid overload (cFO) in a pediatric OHT population and the perioperative risk factors associated with fluid overload. Given a growing body of pediatric cardiac surgical literature reporting the association of both postoperative peak and cumulative fluid overload at 72 h and poor outcomes [13, 25], we assessed cFO across two time points: (1) early (at 72 h of transplantation) and (2) overall (at 1-week post-transplantation). Second, we aimed to explore the relationship between cFO and early and late post-transplant outcomes assessed within the first year of transplantation. Specifically, we investigated the relationship between cFO and allograft dysfunction within the first year of transplant. We hypothesized cFO to be associated with allograft dysfunction as measured by rates of empiric clinical rejection, graft loss/death, and elevated cardiopulmonary filling pressures/ventricular strain observed on routine cardiac catheterization within the first year of transplant.
Patients and Methods
Setting and Study Design
Children who were between the ages of 0 and 21 years of age undergoing cardiac transplantation between January 2010 and May 2018 were included in our analysis. This was a retrospective cohort study from a single-center large volume pediatric heart transplant center in a large metropolitan city, UCLA Mattel Children’s Hospital. The study was approved by the Institutional Review Board at the University of California, Los Angeles. There were no patients who underwent more than one cardiac transplantation during the time period and all indications of transplant, including repeat transplantation for graft failure, were included in the study.
Data Collection
A retrospective chart review was performed with demographic, preoperative, intraoperative, and post-operative variables that were all identified prior to data collection initiation. Data was obtained from electronic medical record. Demographic variables included age, gender, and race. Additional preoperative variables included were last recorded preoperative weight and baseline serum creatinine (both measured up until 1 week prior to transplantation), presence of a cyanotic heart lesion, primary indication for cardiac transplantation, and the presence of mechanical circulatory support at time of transplantation. Perioperative variables included cardiopulmonary bypass time (CPB), aortic cross-clamp time, and donor ischemic time, and need for immediate mechanical circulatory support (MCS) due to primary graft failure following transplantation. All patients with postoperative graft failure needing MCS support was due to the inability to separate initially from cardiopulmonary bypass with primary transplantation. There were no patients needing late MCS support with the initial postoperative recovery. Vasoactive inotrope score (VIS) was calculated using variables recorded within the first 72 h of transplantation. The VIS equation used for the calculation was the following: (dopamine + dobutamine) + (10 × milrinone) + (100 x (epinephrine + norepinephrine + phenylephrine)) in units of mcg/kg/min [26].
Percent fluid accumulation was grouped into three categories, cumulative fluid balance (cFO) of < 5%, 5–10%, and > 10%, respectively. Percent fluid calculation was calculated using accepted standards of fluid overload classification, using the following equation: (total volume input—volume of fluid output)/admission weight (kg) × 100 [27]. Early fluid overload was defined as patients with cumulative fluid overload of > 10% at 72 h of transplantation and overall postoperative cumulative fluid overload was defined as > 10% at 7 days following transplantation. If a patient was discharged or died prior to 7 days of transplantation, the cumulative fluid balance calculated up until day of discharge was used for analysis.
Outcome Measures
Early postoperative outcomes included postoperative mortality, development of acute kidney injury (AKI) within the first week of transplantation, and the use of continuous renal replacement therapy (CRRT) during the initial postoperative hospitalization stay. Postoperative mortality was defined as death related to any-cause during the initial hospitalized postoperative period following cardiac transplantation. Acute kidney injury was defined by the KDIGO definition of acute AKI, stages 1–3 with both serum creatinine and urine output collected daily within the first 7 days of postoperative stay [28]. Baseline preoperative creatinine levels were collected for comparison. Renal failure was defined as those with KDIGO stage 3 AKI.
Longer term outcomes were limited to data collection within the first year of cardiac transplantation as all post-transplant follow-up which included routine cardiac catheterization surveillance and any hospitalizations were exclusively followed within the UCLA health system during this time period. Clinical rejection was defined as new onset heart failure or left ventricular systolic dysfunction that warranted empiric treatment for acute rejection within the first year of transplantation [29], irrespective of corresponding biopsy results. Severe clinical rejection was defined as treatment for acute rejection and the presence of hemodynamic compromise, requiring the use of inotropic and/or vasopressor use, initial IV fluid resuscitation, and/or mechanical circulatory support with the use of extracorporeal life support (ECLS) or ventricular assist device (VAD).
Routine cardiac catheterization surveillance occurred at strict routine time points following transplantation at 1–4 weeks, 3–6 months, and 1-year following initial transplantation. If more than one cardiac catheterization procedure occurred within the aforementioned time frame, the worst hemodynamic data from a single cardiac catheterization procedure, as measured by cardiopulmonary filling pressures, was collected. Data collection from cardiac catheterization was limited to the collection of right atrial pressure (RAP), pulmonary capillary wedge pressure (PCWP), and cardiac index (CI) at the time of the cardiac catheterization procedure.
Statistical Analysis
Student t-test and Chi-square test have been used in comparing continuous and categorical variables, respectively, between those with no fluid accumulation, (cFO) < 5% vs. cFO > 10% groups. Logistic regression models have been used for binary outcomes incorporating cFO group as a main study variable adjusting for age at transplant and admission weight. Binary outcomes we analyzed are mortality, acute kidney injury, rejections (clinical rejection, clinical or biopsy proven rejection, and severe clinical rejection), right atrial pressure (RAP) > 8 mmHg, pulmonary capillary wedge pressure (PCWP) > 12 mmHg, and low cardiac index (CI) as measured by a CI < 2 L/min/m2. The cFO (< 5% vs 10%) evaluated at 72 h and at 1 week were analyzed separately in all models. The frequency of hospitalizations within the first year of cardiac transplantation has been analyzed using general linear model with negative binomial link. Length of hospitalization was analyzed using general linear model. All tests were 2-sided and p-value ≤ 0.05 was considered statistically significant. SAS 9.4 (Cary, NC) has been used for all analyses.
Results
Risk Factors for Fluid Overload
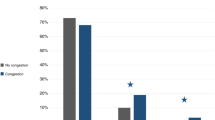
A comparison between baseline characteristics of those with early versus overall cFO, are presented in Tables 1 and 2, respectively. A total of 102 patients met study criteria for analysis during the time period. Early cFO occurred in 14% (14/102) of total cardiac transplant recipients and overall cFO occurred in 23% (23/102). Both time points for fluid overload showed similar findings. Overall postoperative cFO was most likely to occur in younger patients, with 60.9% (14/23) of patients with postoperative fluid overload (p < 0.001) were less than 1 year of age at time of transplantation compared to 12.3% (8/65) of patients without volume overload (< 5%). Corresponding to age, a lower preoperative weight (early, p = 0.01; overall, p < 0.001) was noted to be associated with fluid overload both within the early postoperative period and overall, at 1-week post-transplant. The presence of a cyanotic heart lesion (p = 0.05) was also noted to be a significant risk factor for volume overload in the early postoperative period.
There was no significant difference in the proportion of patients who required preoperative ECMO or VAD, or higher mean times for cardiopulmonary bypass or donor ischemic times among patients with early or overall postoperative volume overload compared to those without volume overload. Patients, however, who had immediate graft failure and required the use of postoperative ECMO, were noted to have higher incidence of both early and overall postoperative volume overload (early, p = 0.01; overall, p = 0.05) (Tables 1 and 2).
Fluid Overload and Early Clinical Outcomes
Both early and overall cFO was not significantly associated with in-hospital mortality (Table 3). Further, there was no significant relationship between early and overall cFO with acute kidney injury (early cFO, OR 1.3 [95% CI 0.4, 4.1], p = 0.69; overall cFO, OR 0.7 [95% CI 0.3, 1.7], p = 0.41), renal failure (early cFO, OR 1.3 [95% CI 0.4, 4.1], p = 0.69; overall cFO, OR 0.7 [95% CI 0.3, 1.7], p = 0.41), or use of CRRT (early cFO, OR 2.3 [95% CI 0.4, 14.0]; overall, cFO, OR 0.5 [95% CI 0.1, 4.9], p = 0.59).
Fluid Overload and Late Clinical Outcomes
Early cFO was noted to be associated with an increase in odds of mortality, OR 9.9 (95% CI 1.8, 54.7, p = 0.01) at 1 year. This association held true, following a multivariate analysis adjusting for age and weight, OR 8.6 (95% CI 1.4, 51.6, p = 0.04) (Table 3). We further assessed whether fluid overload is associated with higher rates of clinical rejection within the first year of transplantation. Although we observed a higher odds risk of overall clinical rejection with early fluid overload (OR 2.0, 95% CI 0.6–6.7; p = 0.3), and severe clinical rejection (OR 2.0 95% CI 0.5–9.1; p = 0.36), these findings were not statistically significant. This relationship was also not significant with overall cFO compared to those with no cFO. We also found no association between fluid overload and the rate of hospitalizations within the first year of cardiac transplantation (Est 1.6, SE 1.6; p = 0.34, by negative binomial regression model).
We also assessed hemodynamic surrogates of primary graft function by evaluating the relationship with fluid overload and cardiopulmonary filling pressures at serial routine time points following cardiac transplantation (Table 4). Although we found early and overall cFO associated with a higher odds risk of elevated right atrial pressure (RAP) > 8 mmHg, OR 2, 95% CI (0.4, 10.8), p = 0.42 and OR 1.2, 95% CI (0.3, 4.3), p = 0.8, respectively, measured within the first month of transplantation, these results were not statistically significant. Further, overall cFO was associated with a lower odds risk of elevated cardiopulmonary filling pressures, measured as elevated RAP > 8 mmHg, OR 0.1 95%CI (0.01, 0.5), p = 0.01 and pulmonary capillary wedge pressure (PCWP) > 12 mmHg, OR 0.1 95%CI (0.01, 0.6), p = 0.01 (Table 4), assessed at 3–6 months post-transplant. There was no significant relationship between both early and overall cFO and low cardiac index (CI), as measured by a cardiac index < 2 L/min/m2, at all routine time points of cardiac catheterization within the first year of cardiac transplantation.
Discussion
The primary results of this study can be summarized as follows. First, consistent with a recent pediatric heart transplant study [21], the incidence of postoperative fluid overload was noted to be higher than what has been previously reported for non-transplant cardiac surgery patients at 23% [13, 30]. Younger age, lower weight, and presence of immediate graft failure needing the use of postoperative ECMO are significant risk factors for both early and overall cumulative postoperative fluid overload. In addition, the presence of cyanotic heart disease was a significant risk factor for early fluid overload. There was no significant relationship between postoperative cFO and immediate post-transplant outcomes. However, at 1-year, early cFO was significantly associated with post-transplant mortality. This relationship was independent of age and weight with a logistic regressions model.
Our findings are consistent with previous data on early volume overload and poor outcomes with pediatric cardiac surgery and post-solid organ transplant [13, 21, 25, 31, 32]. Early fluid overload within 72 h of pediatric cardiac surgery is a known independent predictor of in-hospital mortality and poor cardiac function marked with low cardiac output syndrome [13]. However, we are to our knowledge the first study to show the association of early fluid overload with poor long-term clinical outcomes at 1-year in pediatric heart transplant recipients, suggesting a possible pathophysiologic relationship that extends beyond the immediate perioperative period. ICU-related studies have shown an independent relationship between cumulative fluid balance and long-term mortality among critically ill patients [33, 34]. Mechanistic studies have suggested prolonged fluid overload can lead to direct adverse effects that are inclusive of microvascular injury associated with tissue edema, endothelial dysfunction and shedding of the endothelial glycocalyx, and dysregulated immunity [4, 35,36,37]. Early perioperative fluid overload is also a known risk factor for early graft failure in a wide spectrum of solid organ transplants [31, 32, 38] and well described, poor predictor of long-term survival in allogeneic hematopoietic stem cell transplantation due to multi-end organ failure [39, 40]. In addition, heart transplant recipients are known to be especially vulnerable to the risk of perioperative volume overload due to abnormal cardiorenal neuroendocrine reflexes from cardiac denervation of the transplanted heart [23, 24]. Further, this abnormal neuroendocrine response in OHT recipients has been found to be positively associated with postoperative volume overload, post-transplant systemic hypertension, and transient early allograft dysfunction in adult studies [22,23,24]. This may in part suggest a biological plausibility to the association we observed between early cFO and death at 1 year among pediatric heart transplant recipients.
When further examining the relationship of volume overload with mortality, it is important to note that although not statistically significant, there was a trend toward a positive association between rates of empiric clinical rejection within the first year of transplant and early volume overload that was not observed with cumulative volume overload. Our study may have been statistically underpowered to assess this relationship. Further, a significant proportion of pediatric heart transplant recipients with primary allograft dysfunction are known to present as sudden death [41], and there may be an important relationship between early volume overload and allograft survival that are limited by our primary clinical outcomes of measure. Determinants of allograft dysfunction with routine cardiac catheterization are even also limited in assessment, as they lack longitudinal trajectory, prone to positive selection, and often further exclude small children and infants that are limited in size and vascular access for frequent surveillance post-transplant cardiac catheterization protocols. This study did not demonstrate an association with early fluid overload and elevated filling pressures at early selected time points of cardiac catheterization despite prior adult OHT demonstrating this association [22], possibly due to these study limitations listed above. The association between early fluid overload and poor late outcomes may also be driven by proposed mechanisms of surrounding tissue and endothelial damage, leading not only to primary allograft failure but multi-system end-organ damage as well.
This study also confirms that early volume overload among pediatric OHT patient is highly prevalent, with those at greatest risk being those smaller in size and younger. Irrespective if truly rooted by a pathobiological effect or simply a mere epiphenomenon, early postoperative volume overload may represent an early marker of post-transplant death within the first year of OHT transplantation and this finding warrants further investigation. Our study highlights the question whether closer surveillance of those that sustained early volume overload is indicated and if traditional bedside practices, especially among those that are younger and with low body weight, are sensitive to accurate postoperative volume assessment.
Limitations to our study include we did not assess probable or confirmed causes of death in our study as it relates to volume overload. In addition, other early postoperative outcomes such as duration of mechanical ventilation, duration of inotropic support, intensive care unit length of stay were not evaluated in this study, as we felt at the time they were often influenced by a multitude of other factors, not simply by fluid status. In addition, this was a single-center retrospective cohort study and validation of our findings among a larger multi-center, prospective cohort is warranted. The small sample size of our study limited statistical power and increased probability of type ii error with respect to the clinical outcomes of measure and our multivariate analysis. While we employed statistical analyses to adjust for known predictors of poor outcomes in the transplant population such as age and weight, the small retrospective nature of this study cannot fully account for potential confounders that can influence the statistical associations found in this study. Although our results are further supported by a growing body of literature suggesting a pathobiological role of postoperative volume overload and organ dysfunction in both the cardiac surgical and post-transplant population, any causal inferences made with volume overload and late-transplant outcomes in this study is conjectural and cannot be fully understood with this study alone. In addition, the pediatric heart transplant population is a heterogenous population compared to adult OHT recipients, further complicating the interpretation of our results and with data limited in assessing significant test interactions among subgroups.
This study however has several strengths. Among sparse literature, to date, this is the largest study relating postoperative volume overload and clinical outcomes among pediatric heart transplant recipients. It is the first study to assess the relationship with early volume overload and both clinical outcomes and surrogate markers of allograft dysfunction, extended to the first year of transplant. We assessed volume overload using validated cutoff values of volume overload as reported in the literature and further assessed volume overload at two time points in the postoperative period. Finally, we assessed all clinical and surrogate outcomes to standardized criteria parameters for data collection and outcome reporting.
In conclusion, early postoperative volume overload is highly prevalent and correlates with an increased risk of post-transplant mortality among pediatric heart transplant recipients. Our study indicates the relationship between early postoperative volume overload and mortality is beyond the perioperative period and may be an early predictor of mortality at 1-year following transplantation. Larger prospective studies are warranted to better explore this relationship and further evaluate if changes to goal direct fluid therapy in the postoperative period is needed among this vulnerable population.
Data availability
ML and MF had full access to all of the data in the study and take responsibility for the integrity of the data and accuracy of the data analysis.
Abbreviations
- FO:
-
Fluid overload
- OHT:
-
Orthotopic heart transplant
- CPB:
-
Cardiopulmonary bypass time
- VIS:
-
Vasoactive inotrope score
- Cfo:
-
Cumulative fluid overload
- AKI:
-
Acute kidney injury
- CRRT:
-
Continuous renal replacement therapy
- KDIGO:
-
Kidney Disease Improving Global Outcomes
- ISHLT:
-
International Society for Heart and Lung Transplantation
- ACR:
-
Acute cellular rejection
- AMR:
-
Antibody-mediated rejection
- ECLS:
-
Extracorporeal life support
- VAD:
-
Ventricular assist device
- RAP:
-
Right atrial pressure
- PCWP:
-
Pulmonary capillary wedge pressure
- CI:
-
Cardiac index
- ICU:
-
Intensive care unit
References
Davis AL, Carcillo JA, Aneja RK, Deymann AJ, Lin JC, Nguyen TC, Okhuysen-Cawley RS, Relvas MS, Rozenfeld RA, Skippen PW, Stojadinovic BJ, Williams EA, Yeh TS, Balamuth F, Brierley J, de Caen AR, Cheifetz IM, Choong K, Conway E, Cornell T, Doctor A, Dugas MA, Feldman JD, Fitzgerald JC, Flori HR, Fortenberry JD, Graciano AL, Greenwald BM, Hall MW, Han YY, Hernan LJ, Irazuzta JE, Iselin E, van der Jagt EW, Jeffries HE, Kache S, Katyal C, Kissoon N, Kon AA, Kutko MC, MacLaren G, Maul T, Mehta R, Odetola F, Parbuoni K, Paul R, Peters MJ, Ranjit S, Reuter-Rice KE, Schnitzler EJ, Scott HF, Torres A, Weingarten-Arams J, Weiss SL, Zimmerman JJ, Zuckerberg AL (2017) American college of critical care medicine clinical practice parameters for hemodynamic support of pediatric and neonatal septic shock. Crit Care Med 45(6):1061–1093. https://doi.org/10.1097/CCM.0000000000002425
Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart K, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb SA, Beale RJ, Vincent JL, Moreno R, Subgroup SSCGCitP (2013) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 41(2):580–637. https://doi.org/10.1097/CCM.0b013e31827e83af
Alobaidi R, Morgan C, Basu RK, Stenson E, Featherstone R, Majumdar SR, Bagshaw SM (2018) Association between fluid balance and outcomes in critically ill children: a systematic review and meta-analysis. JAMA Pediatr 172(3):257–268. https://doi.org/10.1001/jamapediatrics.2017.4540
Bellomo R, Raman J, Ronco C (2001) Intensive care unit management of the critically ill patient with fluid overload after open heart surgery. Cardiology 96(3–4):169–176. https://doi.org/10.1159/000047400
Gillespie RS, Seidel K, Symons JM (2004) Effect of fluid overload and dose of replacement fluid on survival in hemofiltration. Pediatr Nephrol 19(12):1394–1399. https://doi.org/10.1007/s00467-004-1655-1
Hayes LW, Oster RA, Tofil NM, Tolwani AJ (2009) Outcomes of critically ill children requiring continuous renal replacement therapy. J Crit Care 24(3):394–400. https://doi.org/10.1016/j.jcrc.2008.12.017
Payen D, de Pont AC, Sakr Y, Spies C, Reinhart K, Vincent JL, Investigators SOiAIPS (2008) A positive fluid balance is associated with a worse outcome in patients with acute renal failure. Crit Care 12(3):R74. https://doi.org/10.1186/cc6916
Sinitsky L, Walls D, Nadel S, Inwald DP (2015) Fluid overload at 48 hours is associated with respiratory morbidity but not mortality in a general PICU: retrospective cohort study. Pediatr Crit Care Med 16(3):205–209. https://doi.org/10.1097/PCC.0000000000000318
Sutherland SM, Zappitelli M, Alexander SR, Chua AN, Brophy PD, Bunchman TE, Hackbarth R, Somers MJ, Baum M, Symons JM, Flores FX, Benfield M, Askenazi D, Chand D, Fortenberry JD, Mahan JD, McBryde K, Blowey D, Goldstein SL (2010) Fluid overload and mortality in children receiving continuous renal replacement therapy: the prospective pediatric continuous renal replacement therapy registry. Am J Kidney Dis 55(2):316–325. https://doi.org/10.1053/j.ajkd.2009.10.048
Zinter MS, Spicer AC, Liu KD, Orwoll BE, Alkhouli MF, Brakeman PR, Calfee CS, Matthay MA, Sapru A (2019) Positive cumulative fluid balance is associated with mortality in pediatric acute respiratory distress syndrome in the setting of acute kidney injury. Pediatr Crit Care Med 20(4):323–331. https://doi.org/10.1097/PCC.0000000000001845
Malbrain ML, Marik PE, Witters I, Cordemans C, Kirkpatrick AW, Roberts DJ, Van Regenmortel N (2014) Fluid overload, de-resuscitation, and outcomes in critically ill or injured patients: a systematic review with suggestions for clinical practice. Anaesthesiol Intensive Ther 46(5):361–380. https://doi.org/10.5603/AIT.2014.0060
Hazle MA, Gajarski RJ, Yu S, Donohue J, Blatt NB (2013) Fluid overload in infants following congenital heart surgery. Pediatr Crit Care Med 14(1):44–49. https://doi.org/10.1097/PCC.0b013e3182712799
Lex DJ, Tóth R, Czobor NR, Alexander SI, Breuer T, Sápi E, Szatmári A, Székely E, Gál J, Székely A (2016) Fluid overload is associated with higher mortality and morbidity in pediatric patients undergoing cardiac surgery. Pediatr Crit Care Med 17(4):307–314. https://doi.org/10.1097/PCC.0000000000000659
Shotan A, Dacca S, Shochat M, Kazatsker M, Blondheim DS, Meisel S (2005) Fluid overload contributing to heart failure. Nephrol Dial Transplant 20(Suppl 7):24–27. https://doi.org/10.1093/ndt/gfh1103
Butler J, Rocker GM, Westaby S (1993) Inflammatory response to cardiopulmonary bypass. Ann Thorac Surg 55(2):552–559. https://doi.org/10.1016/0003-4975(93)91048-r
Hirleman E, Larson DF (2008) Cardiopulmonary bypass and edema: physiology and pathophysiology. Perfusion 23(6):311–322. https://doi.org/10.1177/0267659109105079
Seghaye MC, Duchateau J, Grabitz RG, Faymonville ML, Messmer BJ, Buro-Rathsmann K, von Bernuth G (1993) Complement activation during cardiopulmonary bypass in infants and children. Relation to postoperative multiple system organ failure. J Thorac Cardiovasc Surg 106(6):978–987
Seghaye MC, Grabitz RG, Duchateau J, Busse S, Däbritz S, Koch D, Alzen G, Hörnchen H, Messmer BJ, Von Bernuth G (1996) Inflammatory reaction and capillary leak syndrome related to cardiopulmonary bypass in neonates undergoing cardiac operations. J Thorac Cardiovasc Surg 112(3):687–697. https://doi.org/10.1016/s0022-5223(96)70053-3
Sonntag J, Dähnert I, Stiller B, Hetzer R, Lange PE (1998) Complement and contact activation during cardiovascular operations in infants. Ann Thorac Surg 65(2):525–531. https://doi.org/10.1016/s0003-4975(97)01340-4
Ricci Z (2016) Fluid overload after neonatal cardiac surgery is bad: keep the bottles on the shelf, squeeze the patients…or both? Pediatr Crit Care Med 17(5):463–465. https://doi.org/10.1097/PCC.0000000000000720
Anderson NM, Bond GY, Joffe AR, MacDonald C, Robertson C, Urschel S, Morgan CJ, Group WCCPTPF-u (2021) Post-operative fluid overload as a predictor of hospital and long-term outcomes in a pediatric heart transplant population. Pediatr Transplant 25(3):e13897. https://doi.org/10.1111/petr.13897
Masters RG, Davies RA, Keon WJ, Walley VM, Koshal A, de Bold AJ (1993) Neuroendocrine response to cardiac transplantation. Can J Cardiol 9(7):609–617
Braith RW, Mills RM, Wilcox CS, Convertino VA, Davis GL, Limacher MC, Wood CE (1996) Fluid homeostasis after heart transplantation: the role of cardiac denervation. J Heart Lung Transplant 15(9):872–880
Braith RW, Mills RM, Wilcox CS, Davis GL, Wood CE (1996) Breakdown of blood pressure and body fluid homeostasis in heart transplant recipients. J Am Coll Cardiol 27(2):375–383. https://doi.org/10.1016/0735-1097(95)00467-x
Wilder NS, Yu S, Donohue JE, Goldberg CS, Blatt NB (2016) Fluid Overload is associated with late poor outcomes in neonates following cardiac surgery. Pediatr Crit Care Med 17(5):420–427. https://doi.org/10.1097/PCC.0000000000000715
Gaies MG, Gurney JG, Yen AH, Napoli ML, Gajarski RJ, Ohye RG, Charpie JR, Hirsch JC (2010) Vasoactive-inotropic score as a predictor of morbidity and mortality in infants after cardiopulmonary bypass. Pediatr Crit Care Med 11(2):234–238. https://doi.org/10.1097/PCC.0b013e3181b806fc
Goldstein SL, Currier H, Graf Cd, Cosio CC, Brewer ED, Sachdeva R (2001) Outcome in children receiving continuous venovenous hemofiltration. Pediatrics 107(6):1309–1312. https://doi.org/10.1542/peds.107.6.1309
Khwaja A (2012) KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 120(4):c179-184. https://doi.org/10.1159/000339789
Asimacopoulos EP, Garbern JC, Gauvreau K, Blume ED, Daly KP, Singh TP (2020) Association of clinical rejection versus rejection on protocol biopsy with cardiac allograft vasculopathy in pediatric heart transplant recipients. Transplantation 104(1):e31–e37. https://doi.org/10.1097/TP.0000000000002983
Castañuela-Sánchez V, Hernández-Suárez A, García-Benítez L, Díaz-García L, Martínez-Jasso G, Macedo-Quenot AP (2022) Fluid overload as a predictor of morbidity and mortality in pediatric patients following congenital heart surgery. Bol Med Hosp Infant Mex 79(3):187–192. https://doi.org/10.2485/BMHIM.21000183
Jeong HW, Jung KW, Kim SO, Kwon HM, Moon YJ, Jun IG, Song JG, Hwang GS (2019) Early postoperative weight gain is associated with increased risk of graft failure in living donor liver transplant recipients. Sci Rep 9(1):20096. https://doi.org/10.1038/s41598-019-56543-3
Geube MA, Perez-Protto SE, McGrath TL, Yang D, Sessler DI, Budev MM, Kurz A, McCurry KR, Duncan AE (2016) Increased intraoperative fluid administration is associated with severe primary graft dysfunction after lung transplantation. Anesth Analg 122(4):1081–1088. https://doi.org/10.1213/ANE.0000000000001163
Wang TJ, Pai KC, Huang CT, Wong LT, Wang MS, Lai CM, Chen CH, Wu CL, Chao WC (2022) A positive fluid balance in the first week was associated with increased long-term mortality in critically ill patients: a retrospective cohort study. Front Med (Lausanne) 9:727103. https://doi.org/10.3389/fmed.2022.727103
Balakumar V, Murugan R, Sileanu FE, Palevsky P, Clermont G, Kellum JA (2017) Both positive and negative fluid balance may be associated with reduced long-term survival in the critically Ill. Crit Care Med 45(8):e749–e757. https://doi.org/10.1097/CCM.0000000000002372
Bellomo R, Kellum JA, Ronco C, Wald R, Martensson J, Maiden M, Bagshaw SM, Glassford NJ, Lankadeva Y, Vaara ST, Schneider A (2017) Acute kidney injury in sepsis. Intensive Care Med 43(6):816–828. https://doi.org/10.1007/s00134-017-4755-7
Juffermans NP, van den Brom CE, Kleinveld DJB (2020) Targeting endothelial dysfunction in acute critical illness to reduce organ failure. Anesth Analg 131(6):1708–1720. https://doi.org/10.1213/ANE.0000000000005023
Jaffee W, Hodgins S, McGee WT (2018) Tissue edema, fluid balance, and patient outcomes in severe sepsis: an organ systems review. J Intensive Care Med 33(9):502–509. https://doi.org/10.1177/0885066617742832
Calixto Fernandes MH, Schricker T, Magder S, Hatzakorzian R (2018) Perioperative fluid management in kidney transplantation: a black box. Crit Care 22(1):14. https://doi.org/10.1186/s13054-017-1928-2
Hingorani SR (2017) Fluid: too much or too little-transplant mortality may hang in the balance. Biol Blood Marrow Transplant 23(12):2020–2022. https://doi.org/10.1016/j.bbmt.2017.10.032
Rondon-Clavo C, Scordo M, Hilden P, Shah GL, Cho C, Maloy MA, Papadopoulos EB, Jakubowski AA, O’Reilly RJ, Gyurkocza B, Castro-Malaspina H, Tamari R, Shaffer BC, Perales MA, Jaimes EA, Giralt SA (2018) Early fluid overload is associated with an increased risk of nonrelapse mortality after ex vivo CD34-selected allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 24(12):2517–2522. https://doi.org/10.1016/j.bbmt.2018.07.031
Daly KP, Chakravarti SB, Tresler M, Naftel DC, Blume ED, Dipchand AI, Almond CS, Investigators PHTS (2011) Sudden death after pediatric heart transplantation: analysis of data from the pediatric Heart Transplant Study Group. J Heart Lung Transplant 30(12):1395–1402. https://doi.org/10.1016/j.healun.2011.08.015
Funding
This work was supported by the UCLA CTSI Biostatistics Grant (UL1TR001881) that provided funding for statistical consultation and data analysis.
Author information
Authors and Affiliations
Contributions
L, A, F: study concept and design. L: acquisition of data. L, S, A, F: analysis and interpretation of data. L: drafting of the manuscript. L, S, A, F: critical revision of the manuscript for important intellectual content. S, P: statistical analysis. L: administrative, technical, or material support. F: study supervision. L, S, P, A, F: approval of final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no other disclosures to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lim, M.J., Sim, MS., Pan, S. et al. Early Postoperative Volume Overload is a Predictor of 1-Year Post-Transplant Mortality in Pediatric Heart Transplant Recipients. Pediatr Cardiol 44, 1014–1022 (2023). https://doi.org/10.1007/s00246-023-03134-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-023-03134-9