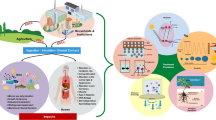
Abstract
Zebrafish (Danio rerio) is an increasingly popular vertebrate model used for assessing the toxicity of endocrine-disrupting chemicals (EDCs) on living beings. The zebrafish features high genetic homology to mammals, because of its rapid embryonic development, optical transparency of phenotypic screening embryos, high throughput genetic and chemical screening which make them a powerful toxicological model. This systematic review aimed to assess the recent literature on the use of zebrafish model in EDCs toxicity studies. We capture the data on the types of EDCs used, zebrafish life stages associated with the toxicity, and its effects on the alterations in neuroendocrine factors and cardiac hypoxia in zebrafish. A total of 17 articles published between 2010 and 2020 were curated. The information gathered highlighted the association of EDCs with cardiological outcomes and neurobehavioral effects and distorted expression of genes. The genes that were highlighted in the paper include bdnf, ntrk2a, grin2cb, VTG-1, HIF-1α, tnnt2, ntrk1, and pax6b. The effect of EDCs on cardiac hypoxia and neurodevelopmental and behavioral factors of zebrafish were described in all the papers chosen for this review. The involvement of EDCs in altered regulation of gene expression can be studied further to identify the potential EDC compounds on its toxicological and endocrine disruption function at the molecular level.

Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Brahimi-Horn MC, Pouysségur J (2009) HIF at a glance. J Cell Sci 122:1055–1057. https://doi.org/10.1242/jcs.035022
Cypher AD, Ickes JR, Bagatto B (2015) Bisphenol A alters the cardiovascular response to hypoxia in danio rerio embryos. Comp Biochem Physiol Part - C Toxicol Pharmacol 174–175:39–45. https://doi.org/10.1016/j.cbpc.2015.06.006
Dambal VY, Selvan KP, Lite C et al (2017) Developmental toxicity and induction of vitellogenin in embryo-larval stages of zebrafish (danio rerio) exposed to methyl Paraben. Ecotoxicol Environ Saf 141:113–118. https://doi.org/10.1016/j.ecoenv.2017.02.048
Darbre PD, Aljarrah A, Miller WR et al (2004) Concentrations of parabens in human breast tumours. J Appl Toxicol 24:5–13. https://doi.org/10.1002/jat.958
El Hussein S, Muret P, Berard M et al (2007) Assessment of principal parabens used in cosmetics after their passage through human epidermis? Dermis layers (ex-vivo study). Exp Dermatol 16:830–836. https://doi.org/10.1111/j.1600-0625.2007.00625.x
FAO/WHO WHO | Toxicological and health aspects of bisphenol A
Fenske L, Concato AC, Vanin AP et al (2020) 17-α-Ethinylestradiol modulates endocrine and behavioral responses to stress in zebrafish. Environ Sci Pollut Res 27:29341–29351. https://doi.org/10.1007/s11356-020-09318-7
Gao X, Wang H-S (2014) Impact of bisphenol A on the cardiovascular system — epidemiological and experimental evidence and molecular mechanisms. Int J Environ Res Public Health 11:8399–8413. https://doi.org/10.3390/ijerph110808399
Gyimah E, Dong X, Qiu W et al (2020) Sublethal concentrations of triclosan elicited oxidative stress, DNA damage, and histological alterations in the liver and brain of adult zebrafish. Environ Sci Pollut Res 27:17329–17338. https://doi.org/10.1007/s11356-020-08232-2
Horzmann KA, Reidenbach LS, Thanki DH et al (2018) Embryonic atrazine exposure elicits proteomic, behavioral, and brain abnormalities with developmental time specific gene expression signatures. J Proteomics 186:71–82. https://doi.org/10.1016/j.jprot.2018.07.006
Hu J, Xia M, Wang Y et al (2020) Paternal exposure to di-n-butyl-phthalate induced developmental toxicity in zebrafish (danio rerio). Birth Defects Res. https://doi.org/10.1002/bdr2.1812
Kim SKSS, Hwang KS, Yang JY et al (2020) Neurochemical and behavioral analysis by acute exposure to bisphenol A in zebrafish larvae model. Chemosphere. https://doi.org/10.1016/j.chemosphere.2019.124751
Kubo T, Maezawa N, Osada M et al (2004) Bisphenol A, an environmental endocrine-disrupting chemical, inhibits hypoxic response via degradation of hypoxia-inducible factor 1α (HIF-1α): Structural requirement of bisphenol A for degradation of HIF-1α. Biochem Biophys Res Commun 318:1006–1011. https://doi.org/10.1016/j.bbrc.2004.04.125
Luzeena Raja G, Divya Subhashree K, Lite C et al (2019) Transient exposure of methylparaben to zebrafish (danio rerio) embryos altered cortisol level, acetylcholinesterase activity and induced anxiety-like behaviour. Comp EndocrinoGenerallogy 279:53–59. https://doi.org/10.1016/j.ygcen.2018.11.001
MacIsaac JK, Gerona RR, Blanc PD et al (2014) Health care worker exposures to the antibacterial agent triclosan. J Occup Environ Med 56:834–839. https://doi.org/10.1097/JOM.0000000000000183
Negishi T, Kawasaki K, Suzaki S et al (2004) Behavioral alterations in response to fear-provoking stimuli and tranylcypromine induced by perinatal exposure to bisphenol A and nonylphenol in male rats. Environ Health Perspect 112:1159–1164. https://doi.org/10.1289/ehp.6961
Nesan D, Sewell LC, Kurrasch DM (2018) Opening the black box of endocrine disruption of brain development: Lessons from the characterization of bisphenol A. Horm Behav 101:50–58
NTP (2008) NTP-CERHR monograph on the potential human reproductive and developmental effects of bisphenol A
Olaniyan LWB, Mkwetshana N, Okoh AI (2016) Triclosan in water, implications for human and environmental health. Springerplus. https://doi.org/10.1186/s40064-016-3287-x
Efsa Draft Scientific Opinion on the risks to public health related to the presence of bisphenol A in foodstuffs-part exposure assessment
Palanza P, Gioiosa L, vom Saal FS, Parmigiani S (2008) Effects of developmental exposure to bisphenol A on brain and behavior in mice. Environ Res 108:150–157
Pant J, Ranjan P, Deshpande SB (2011) Bisphenol A decreases atrial contractility involving NO-dependent G-cyclase signaling pathway. J Appl Toxicol 31:698–702. https://doi.org/10.1002/jat.1647
Papandreou I, Cairns RA, Fontana L et al (2006) HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab 3:187–197. https://doi.org/10.1016/j.cmet.2006.01.012
Parenti CC, Ghilardi A, Della Torre C et al (2019) Environmental concentrations of triclosan activate cellular defence mechanism and generate cytotoxicity on zebrafish (Danio rerio) embryos. Sci Total Environ 650:1752–1758. https://doi.org/10.1016/j.scitotenv.2018.09.283
Pelch K, Wignall JA, Goldstone AE et al (2019) A scoping review of the health and toxicological activity of bisphenol A (BPA) structural analogues and functional alternatives. Toxicology. https://doi.org/10.1016/j.tox.2019.06.006
Pizzino G, Irrera N, Cucinotta M et al (2017) Oxidative Stress: harms and benefits for human health. Oxid Med Cell Longev. https://doi.org/10.1155/2017/8416763
Saili KS, Corvi MM, Weber DN et al (2012) Neurodevelopmental low-dose bisphenol A exposure leads to early life-stage hyperactivity and learning deficits in adult zebrafish. Toxicology 291:83–92. https://doi.org/10.1016/j.tox.2011.11.001
Salahinejad A, Attaran A, Naderi M et al (2021) Chronic exposure to bisphenol S induces oxidative stress, abnormal anxiety, and fear responses in adult zebrafish (danio rerio). Sci Total Environ 750:141633. https://doi.org/10.1016/j.scitotenv.2020.141633
Sárria MP, Soares J, Vieira MN et al (2011) Rapid-behaviour responses as a reliable indicator of estrogenic chemical toxicity in zebrafish juveniles. Chemosphere 85:1543–1547. https://doi.org/10.1016/j.chemosphere.2011.07.048
Song Q, Li J (2014) A systematic review of the human body burden of e-waste exposure in China. Environ Int 68:82–93
Thakkar S, Seetharaman B, Ramasamy V (2021) Impact of chronic sub-lethal methylparaben exposure on cardiac hypoxia and alterations in neuroendocrine factors in zebrafish model. Mol Biol Rep. https://doi.org/10.1007/s11033-021-06878-w
Wirbisky SE, Weber GJ, Schlotman KE et al (2016) Embryonic atrazine exposure alters zebrafish and human miRNAs associated with angiogenesis, cancer, and neurodevelopment. Food Chem Toxicol 98:25–33. https://doi.org/10.1016/j.fct.2016.03.027
Xu S, Zhang H, Pao PC et al (2020) Exposure to phthalates impaired neurodevelopment through estrogenic effects and induced DNA damage in neurons. Aquat Toxicol. https://doi.org/10.1016/j.aquatox.2020.105469
Zhao J, Zhang Q, Zhang B et al (2020) Developmental exposure to lead at environmentally relevant concentrations impaired neurobehavior and NMDAR-dependent BDNF signaling in zebrafish larvae. Environ Pollut 257:113627. https://doi.org/10.1016/j.envpol.2019.113627
Zhu L, Shao Y, Xiao H et al (2018) Electrochemical simulation of triclosan metabolism and toxicological evaluation. Sci Total Environ 622–623:1193–1201. https://doi.org/10.1016/j.scitotenv.2017.11.317
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
ST performed literature search and data analysis, BS drafted and critically revised the work, HS performed literature search, RV had the idea for the article, supervised the work.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there is no potential conflict of interest concerning the research, authorship, and/or publication of this article. All authors read and approved the final manuscript.
Human or Animal Rights
This article does not contain any studies involving human participants and/or animals performed by any of the authors.
Informed Consent
Informed consent was obtained from all authors included in the manuscript.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thakkar, S., Seetharaman, B., Kumar, H. et al. Endocrine-Disrupting Chemicals Exposure Alter Neuroendocrine Factors, Disrupt Cardiac Functions and Provokes Hypoxia Conditions in Zebrafish Model. Arch Environ Contam Toxicol 83, 201–213 (2022). https://doi.org/10.1007/s00244-022-00955-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-022-00955-2