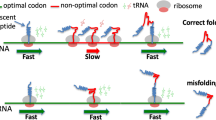
Abstract
Protein-coding DNA sequences can be translated into completely different amino acid sequences if the nucleotide triplets used are shifted by a non-triplet amount on the same DNA strand or by translating codons from the opposite strand. Such “alternative reading frames” of protein-coding genes are a major contributor to the evolution of novel protein products. Recent studies demonstrating this include examples across the three domains of cellular life and in viruses. These sequences increase the number of trials potentially available for the evolutionary invention of new genes and also have unusual properties which may facilitate gene origin. There is evidence that the structure of the standard genetic code contributes to the features and gene-likeness of some alternative frame sequences. These findings have important implications across diverse areas of molecular biology, including for genome annotation, structural biology, and evolutionary genomics.

Similar content being viewed by others
References
Affram Y, Zapata JC, Gholizadeh Z, Tolbert WD, Zhou W, Iglesias-Ussel MD, Pazgier M, Ray K, Latinovic OS, Romerio F (2019) The HIV-1 antisense protein ASP is a transmembrane protein of the cell surface and an integral protein of the viral envelope. J Virol. https://doi.org/10.1128/JVI.00574-19
Ardern Z (2022) Small proteins: overcoming size restrictions. Nat Rev Microbiol 20(2):65
Ardern Z, Neuhaus K, Scherer S (2020) Are antisense proteins in prokaryotes functional? Front Mol Biosci 7(August):187
Arendsee Z, Li J, Singh U, Bhandary P, Seetharam A, Wurtele ES (2019) Fagin: synteny-based phylostratigraphy and finer classification of young genes. BMC Bioinform 20(1):440
Axe DD (2004) Estimating the prevalence of protein sequences adopting functional enzyme folds. J Mol Biol 341(5):1295–1315
Barrell BG, Air GM, Hutchison CA (1976) Overlapping genes in bacteriophage φX174. Nature 264(5581):34–41
Bartonek L, Braun D, Zagrovic B (2020) Frameshifting preserves key physicochemical properties of proteins. Proc Natl Acad Sci USA 117(11):5907–5912
Biba D, Klink G, Bazykin G (2022) Pairs of mutually compensatory frameshifting mutations contribute to protein evolution. Mol Biol Evol. https://doi.org/10.1093/molbev/msac031
Blaabjerg LM, Kassem MM, Good LL, Jonsson N, Cagiada M, Johansson KE, Boomsma W, Stein A, Lindorff-Larsen K (2022) Rapid protein stability prediction using deep learning representations. bioRxiv. https://doi.org/10.1101/2022.07.14.500157
Blalock JE, Smith EM (1984) Hydropathic anti-complementarity of amino acids based on the genetic code. Biochem Biophys Res Commun 121(1):203–207
Blevins WR, Ruiz-Orera J, Messeguer X, Blasco-Moreno B, Villanueva-Cañas JL, Espinar L, Díez J, Carey LB, Mar Albà M (2021) Uncovering de novo gene birth in yeast using deep transcriptomics. Nat Commun 12(1):604
Boldogköi Z (2000) Coding in the noncoding DNA strand: A novel mechanism of gene evolution? J Mol Evol 51(6):600–606
Bordin N, Sillitoe I, Lees JG, Orengo C (2021) Tracing evolution through protein structures: nature captured in a few thousand folds. Front Mol Biosci 8(May):668184
Bordin N, Sillitoe I, Nallapareddy V, Rauer C, Lam SD, Waman VP, Sen N et al (2022) AlphaFold2 reveals commonalities and novelties in protein structure space for 21 model organisms. bioRxiv. https://doi.org/10.1101/2022.06.02.494367
Bornberg-Bauer E, Hlouchova K, Lange A (2021) Structure and function of naturally evolved de novo proteins. Curr Opin Struct Biol 68(June):175–183
Brockhurst MA, Harrison E, Hall JPJ, Richards T, McNally A, MacLean C (2019) The ecology and evolution of pangenomes. Curr Biol CB 29(20):R1094-1103
Cai J, Zhao R, Jiang H, Wang W (2008) De novo origination of a new protein-coding gene in saccharomyces cerevisiae. Genetics 179(1):487–496
Çakır U, Gabed N, Brunet M, Roucou X, Kryvoruchko I (2021) Mosaic translation hypothesis: chimeric polypeptides produced via multiple ribosomal frameshifting as a basis for adaptability. FEBS J. https://doi.org/10.1111/febs.16269
Carter CW Jr (2021) simultaneous codon usage, the origin of the proteome, and the emergence of de-novo proteins. Curr Opin Struct Biol 68(June):142–148
Carvunis A-R, Rolland T, Wapinski I, Calderwood MA, Yildirim MA, Simonis N, Charloteaux B et al (2012) Proto-genes and de novo gene birth. Nature 487(7407):370–374
Cassan E, Arigon-Chifolleau A-M, Mesnard J-M, Gross A, Gascuel O (2016) Concomitant emergence of the antisense protein gene of HIV-1 and of the pandemic. Proc Natl Acad Sci USA 113(41):11537–11542
Chamakura KR, Tran JS, O’Leary C, Lisciandro HG, Antillon SF, Garza KD, Tran E, Min L, Young Ry (2020) Rapid de novo evolution of lysis genes in single-stranded RNA phages. Nat Commun 11(1):6009
Cheng H, Dustin Schaeffer R, Liao Y, Kinch LN, Pei J, Shi S, Kim B-H, Grishin NV (2014) ECOD: an evolutionary classification of protein domains. PLoS Comput Biol 10(12):e1003926
Chothia C (1992) Proteins. One thousand families for the molecular biologist. Nature 357(6379):543–544
Contreras R, Ysebaert M, Jou WM, Fiers W (1973) Bacteriophage Ms2 RNA: nucleotide sequence of the end of the a protein gene and the intercistronic region. Nat New Biol 241(108):99–101
Coray DS, Sibaeva N, McGimpsey S, Gardner PP (2019) The genetic robustness of RNA and protein from evolutionary, structural and functional perspectives. bioRxiv. https://doi.org/10.1101/480087
Delaye L, Deluna A, Lazcano A, Becerra A (2008) The origin of a novel gene through overprinting in Escherichia coli. BMC Evol Biol 8(January):31
Durairaj J, Pereira J, Akdel M, Schwede T (2022) What is hidden in the darkness? Characterization of alphafold structural space. bioRxiv. https://doi.org/10.1101/2022.10.11.511548
Durand É, Gagnon-Arsenault I, Hallin J, Hatin I, Dubé AK, Nielly-Thibault L, Namy O, Landry CR (2019) Turnover of ribosome-associated transcripts from de novo ORFs produces gene-like characteristics available for de novo gene emergence in wild yeast populations. Genome Res 29(6):932–943
Fellner L, Bechtel N, Witting MA, Simon S, Schmitt-Kopplin P, Keim D, Scherer S, Neuhaus K (2014) Phenotype of htgA (mbiA), a recently evolved orphan gene of Escherichia coli and Shigella, completely overlapping in antisense to yaaW. FEMS Microbiol Lett 350(1):57–64
Feltens R, Gossringer M, Willkomm DK, Urlaub H, Hartmann RK (2003) An unusual mechanism of bacterial gene expression revealed for the RNase P protein of thermus strains. Proc Natl Acad Sci USA 100(10):5724–5729
Firth AE (2014) Mapping overlapping functional elements embedded within the protein-coding regions of RNA viruses. Nucl Acids Res 42(20):12425–12439
Firth AE (2020) A putative new SARS-CoV protein, 3c, encoded in an ORF overlapping ORF3a. J Gen Virol 101(10):1085–1089
Forsdyke DR (1995) Sense in antisense? J Mol Evol 41(5):582–586
Gelsinger DR, Dallon E, Reddy R, Mohammad F, Buskirk AR, DiRuggiero J (2020) Ribosome profiling in archaea reveals leaderless translation, novel translational initiation sites, and ribosome pausing at single codon resolution. Nucl Acids Res 48(10):5201–5216
Geyer R, Mamlouk AM (2018) On the efficiency of the genetic code after frameshift mutations. PeerJ 6(May):e4825
Gontijo AM, Miguela V, Whiting MF, Woodruff RC, Dominguez M (2011) intron retention in the drosophila melanogaster rieske iron sulphur protein gene generated a new protein. Nat Commun 2:323
Grassé PP (1973) L’évolution Du Vivant, Materiaux Pour Une Nouvelle Théorie Transformiste, Pierre Grassé. Sciences 1:1
Grassé P-P (1977) Evolution of living organisms: evidence for a new theory of transformation. Academic Press, London
Guan Y, Liu Li, Wang Q, Zhao J, Li P, Jinyong Hu, Yang Z, Running MP, Sun H, Huang J (2018) Gene refashioning through innovative shifting of reading frames in mosses. Nat Commun 9(1):1555
Heames B, Buchel F, Aubel M, Tretyachenko V, Loginov D, Novák P, Lange A, Bornberg-Bauer E, Hlouchová K (2023) Experimental characterization of de novo proteins and their unevolved random-sequence counterparts. Nat Ecol Evol 7(4):570–580
Hecht MH, Das A, Go A, Bradley LH, Wei Y (2004) De novo proteins from designed combinatorial libraries. Prot Sci A Publ Prot Soc 13(7):1711–1723
Hernandez IB, Yeo J, Jänes J, Wein T, Varadi M, Velankar S, Beltrao P, Steinegger M (2023) Clustering predicted structures at the scale of the known protein universe. bioRxiv. https://doi.org/10.1101/2023.03.09.531927
Hücker SM, Vanderhaeghen S, Abellan-Schneyder I, Scherer S, Neuhaus K (2018) The novel anaerobiosis-responsive overlapping gene ano is overlapping antisense to the annotated gene ECs2385 of Escherichia coli O157:H7 sakai. Front Microbiol 9(May):931
Ingolia NT, Brar GA, Stern-Ginossar N, Harris MS, Talhouarne GJS, Jackson SE, Wills MR, Weissman JS (2014) Ribosome profiling reveals pervasive translation outside of annotated protein-coding genes. Cell Rep 8(5):1365–1379
Itzkovitz S, Alon U (2007) The genetic code is nearly optimal for allowing additional information within protein-coding sequences. Genome Res 17(4):405–412
Jacob F (1977) Evolution and tinkering. Science. https://www.jstor.org/stable/1744610?casa_token=915UyeD8Q7AAAAAA:pDqkNrpyNa6H6enkNB0j0StEF5x6tlONkC1H6hbOUoviRxBFJSq6HKAczRkCljySNCUVAc21iIhoUP3Cp-KjxiGivK9wWOnjq-2drxZ7hwwfTbSMWQY
Jumper J, Evans R, Pritzel A, Green T, Figurnov M, Ronneberger O, Tunyasuvunakool K et al (2021) Highly accurate protein structure prediction with alphafold. Nature 596(7873):583–589
Jungreis I, Nelson CW, Ardern Z, Finkel Y, Krogan NJ, Sato K, Ziebuhr J et al (2021) Conflicting and ambiguous names of overlapping ORFs in the SARS-CoV-2 genome: a homology-based resolution. Virology 558(June):145–151
Kamijo T, Zindy F, Roussel MF, Quelle DE, Downing JR, Ashmun RA, Grosveld G, Sherr CJ (1997) Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19ARF. Cell 91(5):649–659
Keese PK, Gibbs A (1992) Origins of genes: ‘Big Bang’ or continuous creation? Of the National Academy of Sciences. https://www.pnas.org/content/89/20/9489.short
Khan YA, Jungreis I, Wright JC, Mudge JM, Choudhary JS, Firth AE, Kellis M (2020) Evidence for a novel overlapping coding sequence in POLG initiated at a CUG start codon. BMC Genet 21(1):25
Kohl MP, Kompatscher M, Clementi N, Holl L, Erlacher MD (2023) Initiation at AUGUG and GUGUG sequences can lead to translation of overlapping reading frames in E. coli. Nucl Acids Res 51(1):271–289
Kolodny R, Nepomnyachiy S, Tawfik DS, Ben-Tal N (2021) Bridging themes: short protein segments found in different architectures. Mol Biol Evol 38(6):2191–2208
Konecny J, Eckert M, Schöniger M, Hofacker GL (1993) Neutral adaptation of the genetic code to double-strand coding. J Mol Evol 36(5):407–416
Koonin EV, Novozhilov AS (2017) Origin and evolution of the universal genetic code. Annu Rev Genet 51(November):45–62
Korniy N, Samatova E, Anokhina MM, Peske F, Rodnina MV (2019) Mechanisms and biomedical implications of − 1 programmed ribosome frameshifting on viral and bacterial mRNAs. FEBS Lett 593(13):1468–1482
Kosinski LJ, Masel J (2020) Readthrough errors purge deleterious cryptic sequences, facilitating the birth of coding sequences. Mol Biol Evol 37(6):1761–1774
Kosinski L, Aviles N, Gomez K, Masel J (2022) Random peptides rich in small and disorder-promoting amino acids are less likely to be harmful. Genome Biol Evol. https://doi.org/10.1093/gbe/evac085
Kreitmeier M, Ardern Z, Abele M, Ludwig C, Scherer S, Neuhaus K (2022) Spotlight on alternative frame coding: two long overlapping genes in pseudomonas aeruginosa are translated and under purifying selection. iScience 25(2):103844
Kubatova N, Pyper DJ, Jonker HRA, Saxena K, Remmel L, Richter C, Brantl S et al (2020) Rapid biophysical characterization and NMR spectroscopy structural analysis of small proteins from bacteria and archaea. Chembiochem A Eur J Chem Biol 21(8):1178–1187
Li Ke, Kong J, Zhang S, Zhao T, Qian W (2022) Distance-dependent inhibition of translation initiation by downstream out-of-frame AUGs is consistent with a brownian ratchet process of ribosome scanning. Genome Biol 23(1):254
Lin Z, Akin H, Rao R, Hie B, Zhu Z, Lu W, Smetanin N et al (2022) Evolutionary-scale prediction of atomic level protein structure with a language model. bioRxiv. https://doi.org/10.1101/2022.07.20.500902
Loughran G, Zhdanov AV, Mikhaylova MS, Rozov FN, Datskevich PN, Kovalchuk SI, Serebryakova MV et al (2020) Unusually efficient CUG initiation of an overlapping reading frame in POLG mRNA yields novel protein POLGARF. Proc Natl Acad Sci USA 117(40):24936–24946
Louis AA (2016) Contingency, convergence and hyper-astronomical numbers in biological evolution. Stud Hist Philos Biol Biomed Sci 58(August):107–116
Miller RH (1988) Human immunodeficiency virus may encode a novel protein on the genomic DNA plus strand. Science 239(4846):1420–1422
Mudge JM, Ruiz-Orera J, Prensner JR, Brunet MA, Calvet F, Jungreis I, Gonzalez JM et al (2022) Standardized annotation of translated open reading frames. Nat Biotechnol 40(7):994–999
Negoro S, Ohki T, Shibata N, Sasa K, Hayashi H, Nakano H, Yasuhira K, Kato D-I, Takeo M, Higuchi Y (2007) Nylon-oligomer degrading enzyme/substrate complex: catalytic mechanism of 6-aminohexanoate-dimer hydrolase. J Mol Biol 370(1):142–156
Nekrutenko A, Wadhawan S, Goetting-Minesky P, Makova KD (2005) Oscillating evolution of a mammalian locus with overlapping reading frames: an XLalphas/ALEX relay. PLoS Genet 1(2):e18
Nelson CW, Ardern Z, Goldberg TL, Meng C, Kuo C-H, Ludwig C, Kolokotronis S-O, Wei X (2020a) Dynamically evolving novel overlapping gene as a factor in the SARS-CoV-2 pandemic. Elife. https://doi.org/10.7554/eLife.59633
Nelson CW, Ardern Z, Wei X (2020b) OLGenie: estimating natural selection to predict functional overlapping genes. Mol Biol Evol 37(8):2440–2449
Ohno S (1970) Evolution by gene duplication. Springer, Berlin
Ohno S (1984) Birth of a unique enzyme from an alternative reading frame of the preexisted, internally repetitious coding sequence. Proc Natl Acad Sci USA 81(8):2421–2425
Okamura K, Feuk L, Marquès-Bonet T, Navarro A, Scherer SW (2006) Frequent appearance of novel protein-coding sequences by frameshift translation. Genomics 88(6):690–697
Ouspenskaia T, Law T, Clauser KR, Klaeger S, Sarkizova S, Aguet F, Li Bo et al (2022) Unannotated proteins expand the MHC-I-restricted immunopeptidome in cancer. Nat Biotechnol 40(2):209–217
Pan F, Zhang Y, Liu X, Zhang J (2021) Estimating the designability of protein structures. bioRxiv. https://doi.org/10.1101/2021.11.03.467111
Papadopoulos C, Callebaut I, Gelly J-C, Hatin I, Namy O, Renard M, Lespinet O, Lopes A (2021) Intergenic ORFs as elementary structural modules of de novo gene birth and protein evolution. Genome Res. https://doi.org/10.1101/gr.275638.121
Pavesi A (2019) Asymmetric evolution in viral overlapping genes is a source of selective protein adaptation. Virology 532(June):39–47
“Plutarch, Theseus” (2022) Accessed November 23, 2022. https://www.perseus.tufts.edu/hopper/text?doc=Perseus%3Atext%3A2008.01.0067%3Achapter%3D23%3Asection%3D1
Raes J, Van de Peer Y (2005) Functional divergence of proteins through frameshift mutations. Trends Genet TIG 21(8):428–431
Rancurel C, Mahvash Khosravi A, Dunker K, Romero PR, Karlin D (2009) Overlapping genes produce proteins with unusual sequence properties and offer insight into de novo protein creation. J Virol 83(20):10719–10736
Ruiz-Orera J, Verdaguer-Grau P, Villanueva-Cañas JL, Messeguer X, Mar Albà M (2018) Translation of neutrally evolving peptides provides a basis for de novo gene evolution. Nat Ecol Evol 2(5):890–896
Sabath N, Wagner A, Karlin D (2012) Evolution of viral proteins originated de novo by overprinting. Mol Biol Evol 29(12):3767–3780
Salisbury FB (1969) Natural selection and the complexity of the gene. Nature 224(5217):342–343
Schmitz JF, Bornberg-Bauer E (2017) Fact or fiction: updates on how protein-coding genes might emerge de novo from previously non-coding DNA. F1000Research 6(January):57
Schmitz JF, Ullrich KK, Bornberg-Bauer E (2018) Incipient de novo genes can evolve from frozen accidents that escaped rapid transcript turnover. Nat Ecol Evol 2(10):1626–1632
Sealfon RS, Lin MF, Jungreis I, Wolf MY, Kellis M, Sabeti PC (2015) FRESCo: finding regions of excess synonymous constraint in diverse viruses. Genome Biol 16(February):38
Sillitoe I, Bordin N, Dawson N, Waman VP, Ashford P, Scholes HM, Pang CSM et al (2021) CATH: increased structural coverage of functional space. Nucl Acids Res 49(D1):D266–D273
Smith JM (1970) Natural selection and the concept of a protein space. Nature 225(5232):563–564
Smith C, Canestrari JG, Wang AJ, Champion MM, Derbyshire KM, Gray TA, Wade JT (2022) Pervasive translation in mycobacterium tuberculosis. Elife. https://doi.org/10.7554/eLife.73980
Soler-Camargo NC, Silva-Pereira TT, Zimpel CK, Camacho MF, Zelanis A, Aono AH, Patané JS, Dos Santos AP, Guimarães AMS (2022) The rate and role of pseudogenes of the mycobacterium tuberculosis complex. Microb Genomics. https://doi.org/10.1099/mgen.0.000876
Stewart H, Lu Y, O’Keefe S, Valpadashi A, Cruz-Zaragoza LD, Michel HA, Nguyen SK et al (2022) The SARS-CoV-2 protein ORF3c is a mitochondrial modulator of innate immunity. bioRxiv. https://doi.org/10.1101/2022.11.15.516323
Storz G, Wolf YI, Ramamurthi KS (2014) Small proteins can no longer be ignored. Annu Rev Biochem 83(March):753–777
Tassios E, Nikolaou C, Vakirlis N (2023) Intergenic regions of saccharomycotina yeasts are enriched in potential to encode transmembrane domains. Mol Biol Evol. https://doi.org/10.1093/molbev/msad059
Tian P, Best RB (2017) How many protein sequences fold to a given structure? A coevolutionary analysis. Biophys J 113(8):1719–1730
Tokuriki N, Stricher F, Schymkowitz J, Serrano L, Tawfik DS (2007) The stability effects of protein mutations appear to be universally distributed. J Mol Biol 369(5):1318–1332
Vakirlis N, Hebert AS, Opulente DA, Achaz G, Hittinger CT, Fischer G, Coon JJ, Lafontaine I (2018) A molecular portrait of de novo genes in yeasts. Mol Biol Evol 35(3):631–645
Vakirlis N, Acar O, Hsu B, Coelho NC, Branden Van Oss S, Wacholder A, Medetgul-Ernar K et al (2020a) De novo emergence of adaptive membrane proteins from thymine-rich genomic sequences. Nat Commun 11(1):781
Vakirlis N, Carvunis A-R, McLysaght A (2020b) Synteny-based analyses indicate that sequence divergence is not the main source of orphan genes. Elife. https://doi.org/10.7554/eLife.53500
Van de Voorde A, Contreras R, Rogiers R, Fiers W (1976) The initiation region of the SV40 VP1 gene. Cell 9(1):117–120
Van Oss SB, Branden S, Carvunis A-R (2019) De novo gene birth. PLoS Genet 15(5):e1008160
Vanderhaeghen S, Zehentner B, Scherer S, Neuhaus K, Ardern Z (2018) The novel EHEC gene asa overlaps the TEGT transporter gene in antisense and is regulated by NaCl and growth phase. Sci Rep 8(1):17875
Wacholder A, Parikh SB, Coelho NC, Acar O, Houghton C, Chou L, Carvunis A-R (2023) A vast evolutionarily transient translatome contributes to phenotype and fitness. bioRxiv. https://doi.org/10.1101/2021.07.17.452746
Wang X, Dong Q, Chen G, Zhang J, Liu Y, Cai Y (2022) Frameshift and wild-type proteins are often highly similar because the genetic code and genomes were optimized for frameshift tolerance. BMC Genomics. https://doi.org/10.1186/s12864-022-08435-6
Watson AK, Lopez P, Bapteste E (2022) Hundreds of out-of-frame remodeled gene families in the Escherichia coli pangenome. Mol Biol Evol. https://doi.org/10.1093/molbev/msab329
Weidmann L, Dijkstra T, Kohlbacher O, Lupas AN (2021) Minor deviations from randomness have huge repercussions on the functional structuring of sequence space. bioRxiv. https://doi.org/10.1101/706119
Weisman CM (2022) The origins and functions of de novo genes: Against all odds? J Mol Evol 90(3–4):244–257
Weisman CM, Murray AW, Eddy SR (2020) Many, but not all, lineage-specific genes can be explained by homology detection failure. PLoS Biol 18(11):e3000862
Weisman CM, Murray AW, Eddy SR (2022) Mixing genome annotation methods in a comparative analysis inflates the apparent number of lineage-specific genes. Curr Biol CB 32(12):2632–39.e2
Weissenow K, Heinzinger M, Rost B (2022) Protein language-model embeddings for fast, accurate, and alignment-free protein structure prediction. Structure. https://doi.org/10.1016/j.str.2022.05.001
Whitehead DJ, Wilke CO, Vernazobres D, Bornberg-Bauer E (2008) The look-ahead effect of phenotypic mutations. Biol Direct 3(May):18
Wichmann S, Ardern Z (2019) Optimality in the standard genetic code is robust with respect to comparison code sets. Biosystems 185(November):104023
Wichmann S, Ardern Z (2023) Highly similar average collateral effect of synonymous mutations across alternative reading frames: a potential role in evolvability. BioCosmos 3(1):1–11
Wichmann S, Scherer S, Ardern Z (2021) Biological factors in the synthetic construction of overlapping genes. BMC Genomics 22(1):888
Willis S, Masel J (2018) Gene birth contributes to structural disorder encoded by overlapping genes. Genetics 210(1):303–313
Wilson BA, Foy SG, Neme R, Masel J (2017) Young genes are highly disordered as predicted by the preadaptation hypothesis of de novo gene birth. Nat Ecol Evol 1(6):0146–0146
Wnętrzak M, Błażej P, Mackiewicz P (2019) Optimization of the standard genetic code in terms of two mutation types: point mutations and frameshifts. Biosystems 181(July):44–50
Wright BW, Molloy MP, Jaschke PR (2022a) Overlapping genes in natural and engineered genomes. Nat Rev Genet 23(3):154–168
Wright BW, Yi Z, Weissman JS, Chen J (2022b) The dark proteome: translation from noncanonical open reading frames. Trends Cell Biol 32(3):243–258
Xu H, Zhang J (2021) On the origin of frameshift-robustness of the standard genetic code. Mol Biol Evol 38(10):4301–4309
Zehentner B, Ardern Z, Kreitmeier M, Scherer S, Neuhaus K (2020a) A novel pH-regulated, unusual 603 Bp overlapping protein coding gene pop is encoded antisense to ompA in Escherichia coli O157:H7 (EHEC). Front Microbiol 11(March):377
Zehentner B, Ardern Z, Kreitmeier M, Scherer S, Neuhaus K (2020b) Evidence for numerous embedded antisense overlapping genes in diverse E. coli strains. bioRxiv. https://doi.org/10.1101/2020.11.18.388249
Zile K, Dessimoz C, Wurm Y, Masel J (2020) Only a single taxonomically restricted gene family in the drosophila melanogaster subgroup can be identified with high confidence. Genome Biol Evol 12(8):1355–1366
Zull JE, Smith SK (1990) Is genetic code redundancy related to retention of structural information in both DNA strands? Trends Biochem Sci 15(7):257–261
Acknowledgements
Thanks to Md. Hassan uz-Zaman for detailed discussion and to Avril Coghlan, Alisson Gontijo, Cosmin Saveanu, Arlin Stoltzfus, for helpful comments on the manuscript.
Funding
This work, conducted at the Wellcome Sanger Institute, was supported by the Wellcome Trust, Grant No. [220540/Z/20/A].
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Erich Bornberg-Bauer.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ardern, Z. Alternative Reading Frames are an Underappreciated Source of Protein Sequence Novelty. J Mol Evol 91, 570–580 (2023). https://doi.org/10.1007/s00239-023-10122-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-023-10122-3